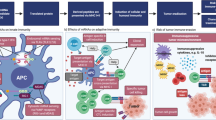
Abstract
Recent advances in molecular biology have led to dramatic enhancement of the stability of in vitro transcribed (IVT) messenger RNA (mRNA) and improvement in its translational efficacy. Nowadays, mRNA-based vaccines represent a promising approach in the field of anticancer immunotherapy, gaining attention over the earlier-established bacteria-, virus-, or cell-based vaccination approaches. Here, we present the experimental procedures employed in our laboratory to induce anticancer immune responses in different murine tumor models using IVT mRNA encoding for immune activation signals and antigens of interest.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Palucka K, Banchereau J (2013) Dendritic-cell-based therapeutic cancer vaccines. Immunity 39:38–48
Constantino J, Gomes C, Falcão A et al (2015) Antitumor dendritic cell-based vaccines: lessons from 20 years of clinical trials and future perspectives. Transl Res. doi:10.1016/j.trsl.2015.07.008
Van Lint S, Wilgenhof S, Heirman C et al (2014) Optimized dendritic cell-based immunotherapy for melanoma: the TriMix-formula. Cancer Immunol Immunother 63:959–967
Van Lint S, Goyvaerts C, Maenhout S et al (2012) Preclinical evaluation of TriMix and antigen mRNA-based antitumor therapy. Cancer Res 72:1661–1671
Van der Jeught K, Bialkowski L, Daszkiewicz L et al (2015) Targeting the tumor microenvironment to enhance antitumor immune responses. Oncotarget 6:1359–1381
Van der Jeught K, Van Lint S, Thielemans K, Breckpot K (2015) Intratumoral delivery of mRNA: overcoming obstacles for effective immunotherapy. Oncoimmunology 4:e1005504
Van Lint S, Renmans D, Broos K et al (2015) Intratumoral delivery of TriMix mRNA results in T-cell activation by cross-presenting dendritic cells. Cancer Immunol Res 4:146–156. doi:10.1158/2326-6066.CIR-15-0163
Tuyaerts S, Van Meirvenne S, Bonehill A et al (2007) Expression of human GITRL on myeloid dendritic cells enhances their immunostimulatory function but does not abrogate the suppressive effect of CD4 + CD25+ regulatory T cells. J Leukoc Biol 82:93–105
Pen JJ, Keersmaecker BD, Heirman C et al (2014) Interference with PD-L1/PD-1 co-stimulation during antigen presentation enhances the multifunctionality of antigen-specific T cells. Gene Ther 21:1–10
De Keersmaecker B, Heirman C, Corthals J et al (2011) The combination of 4-1BBL and CD40L strongly enhances the capacity of dendritic cells to stimulate HIV-specific T cell responses. J Leukoc Biol 89:989–999
Bonehill A, Van Nuffel AMT, Corthals J et al (2009) Single-step antigen loading and activation of dendritic cells by mRNA electroporation for the purpose of therapeutic vaccination in melanoma patients. Clin Cancer Res 15:3366–3375
Aerts-Toegaert C, Heirman C, Tuyaerts S et al (2007) CD83 expression on dendritic cells and T cells: correlation with effective immune responses. Eur J Immunol 37:686–695
Van Nuffel AMT, Benteyn D, Wilgenhof S et al (2012) Intravenous and intradermal TriMix-dendritic cell therapy results in a broad T-cell response and durable tumor response in a chemorefractory stage IV-M1c melanoma patient. Cancer Immunol Immunother 61:1033–1043
Wilgenhof S, Corthals J, Van Nuffel AMT et al (2015) Long-term clinical outcome of melanoma patients treated with messenger RNA-electroporated dendritic cell therapy following complete resection of metastases. Cancer Immunol Immunother 64:381–388
Wilgenhof S, Van Nuffel AMT, Benteyn D et al (2013) A phase IB study on intravenous synthetic mRNA electroporated dendritic cell immunotherapy in pretreated advanced melanoma patients. Ann Oncol 24:2686–2693
Johansen P, Häffner AC, Koch F et al (2005) Direct intralymphatic injection of peptide vaccines enhances immunogenicity. Eur J Immunol 35:568–574
Van Lint S, Heirman C, Thielemans K, Breckpot K (2013) mRNA: from a chemical blueprint for protein production to an off-the-shelf therapeutic. Hum Vaccin Immunother 9:265–274
Joyce JA, Fearon DT (2015) T cell exclusion, immune privilege, and the tumor microenvironment. Science 348:74–80
Keyaerts M, Verschueren J, Bos TJ et al (2008) Dynamic bioluminescence imaging for quantitative tumour burden assessment using IV or IP administration of D: luciferin: effect on intensity, time kinetics and repeatability of photon emission. Eur J Nucl Med Mol Imaging 35:999–1007
Acknowledgements
This work was supported by the Agency for Innovation by Science and Technology (IWT-Vlaanderen), Interuniversity Attraction Poles Program, the National Cancer Plan, the Stichting Tegen Kanker, the Kom op tegen Kanker, the Fonds voor Wetenschappelijk Onderzoek Vlaanderen (FWO-Vlaanderen), the Hercules Foundation Flanders (Middelzware onderzoeksinfrastructuur), and the EU FP7-cancer immunotherapy program. Marleen Keyaerts is a Senior Clinical Investigator of FWO-Vlaanderen.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media New York
About this protocol
Cite this protocol
Bialkowski, L. et al. (2017). Adjuvant-Enhanced mRNA Vaccines. In: Kramps, T., Elbers, K. (eds) RNA Vaccines. Methods in Molecular Biology, vol 1499. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6481-9_11
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6481-9_11
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6479-6
Online ISBN: 978-1-4939-6481-9
eBook Packages: Springer Protocols