Abstract
Pain caused by dysfunction or damage to the peripheral or central nervous system is typified by the symptoms described by patients with painful diabetic neuropathy, post-herpetic neuralgia and central poststroke pain. All these conditions are more common in the elderly.
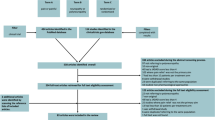
Neuropathic pain has long been recognised as one of the more difficult types of pain to treat; however, with the current emphasis on providing a multidisciplinary assessment and approach to management, more patients will be offered relief of their symptoms and an improved quality of life.
Despite the use of combination drug therapy, adequate pain relief in the elderly is difficult to achieve without adverse effects. In an attempt to minimise these it is important to include non-drug treatment options in the management plan. Lifestyle changes and environmental modification, together with encouragement to adopt an appropriate exercise programme and an emphasis on maintaining mobility and independence should always be considered.
Interventional therapy ranging from simple nerve blocks to intrathecal drug delivery can be of value.
Drug treatment remains the mainstay of therapy. Tricyclic antidepressants such as amitriptyline, while having significant adverse effects in the elderly, have a number needed to treat (NNT) of 3.5 for 50% pain relief in diabetic neuropathy and 2.1 for 50% pain relief in postherpetic neuralgia.
The newer antiepileptic drugs, such as gabapentin, appear to have a better adverse effect profile and provide similar efficacy with the NNT for treating painful diabetic neuropathy being 3.7 and 3.2 for treating pain in postherpetic neuralgia.
As our understanding of the complexities of the pain processes increases, we are becoming more able to appropriately combine treatments to achieve not only improved pain relief but also improved function.




Similar content being viewed by others
References
Merskey H, Bogduk N. Classification of chronic pain. 2nd ed. Seattle (WA): IASP Press, 1994
Woolf CJ, Mannion RJ. Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet 1999; 353(9168): 1959–64
National Health & Medical Research Council. Acute pain management: scientific evidence. Australia: Commonwealth of Australia, 1999
Cherny NI, Portenoy RK. In: Wall P, Melzack R, editors. Text book of pain. 4th ed. Philadelphia (PA): W B Saunders Co., 1999: 1049–51
Nolan L, O’Malley K. Prescribing for the elderly: part II. Prescribing patterns: differences due to age. J Am Geriatr Soc 1988; 36(3): 245–54
Ferrell BA. Overview of aging and pain. In: Ferrell BA, Ferrell BR, editors. Pain in the elderly. Seattle (WA): IASP Press, 1996: 1–10
Crook J, Rideout E, Browne G. The prevalence of pain complaints in a general population. Pain 1984; 18(3): 299–314
Ferrell BA, Ferrell BR, Osterweil D. Pain in the nursing home. J Am Geriatr Soc 1990; 38(4): 409–14
Ferrell BA, Ferrell BR, Rivera L. Pain in cognitively impaired nursing home patients. J Pain Symptom Manage 1995; 10(8): 591–8
Gibson SJ, Farrell MJ, Katz B. Multidisciplinary management of chronic nonmalignant pain in older adults. In: Ferrell BR, Ferrell BA, editors. Pain in the elderly. Seattle (WA): IASP Press, 1996: 91–100
Ferrell BR. Patient education and nondrug interventions. In: Ferrell BR, Ferrell BA, editors. Pain in the elderly. Seattle (WA): IASP Press, 1996: 35–44
Hoffman J. Placebo analgesia. In: Ramamurthy S, Rogers JN, editors. Decision making in pain management. St Louis (MO): Mosby-Year Book, 1993: 204
Winnie AP, Candido KD. Differential neural blockade for the diagnosis of pain. In: Waldman SD, editor. Interventional pain management. Philadelphia (PA): WB Saunders, 2001: 162–73
Peters G, Nurmikko TJ. Peripheral and gasserian ganglion-level procedures for the treatment of trigeminal neuralgia. Clin J Pain 2002; 18: 28–34
Elias WJ, Burchiel KJ. Microvascular decompression. Clin J Pain 2002; 18: 35–41
Choi YK, Liu J. The use of 5% lidocaine for prolonged analgesia in chronic pain patients: a new technique. Reg Anesth Pain Med 1998; 23(1): 96–100
Saberski LR, Ahmad M, Fitzgerald JF. Cryoneurolysis and radiofrequency lesioning. In: Raj PP, editor. Practical management of pain. St Louis (MO): Mosby, 2000: 753–67
Bennett G, Serafini M, Burchiel K, et al. Evidence-based review of the literature on intrathecal delivery of pain medication. J Pain Symptom Manage 2000; 20(2): S12–36
Anderson VC, Burchiel KJ. A prospective study of long-term intrathecal morphine in the management of chronic non-malignant pain. Neurosurgery 1999; 44(2): 289–300; discussion 300–1
Lund C, Mogensen T, Hjortso NC, et al. Systemic morphine enhances spread of sensory analgesia during postoperative epidural bupivacaine infusion. Lancet 1985; 2(8465): 1156–7
Dahl JB, Rosenberg J, Hansen BL, et al. Differential analgesic effects of low-dose epidural morphine and morphine-bupivacaine at rest and during mobilization after major abdominal surgery. Anesth Analg 1992; 74(3): 362–5
Etches RC, Gammer TL, Cornish R. Patient-controlled epidural analgesia after thoracotomy: a comparison of meperidine with and without bupivacaine. Anesth Analg 1996; 83(1): 81–6
Sjoberg M, Karlsson PA, Nordberg C, et al. Neuropathologic findings after long term intrathecal infusion of morphine and Bupivacaine for ain treatment in cancer patients. Anaesthesiology 1992; 76: 173–86
Wagemans MF, van der Valk P, Spoelder EM, et al. Neurohistopathological findings after continuous intrathecal administration of morphine or a morphine/Bupivacaine mixture in cancer pain patients. Acta Anaesthesiol Scand 1997; 41: 103–8
Dahm P, Lundborg C, Janson M, et al. Comparison of 0.5% intrathecal bupivacaine with 0.5% intrathecal ropivacaine in the treatment of refractory cancer and noncancer pain conditions: results from a prospective, crossover, double-blind, randomized study. Reg Anesth Pain Med 2000; 25(5): 480–7
Eisenach JC, DuPen S, Dubois M, et al. Epidural clonidine analgesia for intractable cancer pain. The Epidural Clonidine Study Group. Pain 1995; 61(3): 391–9
Eisenach JC, Hood DD, Curry R. Intrathecal, but not intravenous, clonidine reduces experimental thermal or capsaicin-induced pain and hyperalgesia in normal volunteers. Anesth Analg 1998; 87(3): 591–6
Zuniga RE, Schlicht CR, Abram SE. Intrathecal baclofen is analgesic in patients with chronic pain. Anesthesiology 2000; 92(3): 876–80
Bowersox SS, Luther R. Pharmacotherapeutic potential of Omega-conotoxin MV2A (SNX-111), and N-type neuronal calcium channel blocker found in the venom of Conus Magus. Toxicon 1998; 36: 1651–8
Penn RD, Paice JA. Adverse effects associated with the intrathecal administration of ziconotide. Pain 2000; 85(1–2): 291–6
Lauretti GR, Reis MP, Prado WA, et al. Dose-response study of intrathecal morphine versus intrathecal neostigmine, their combination, or placebo for postoperative analgesia in patients undergoing anterior and posterior vaginoplasty. Anesth Analg 1996; 82(6): 1182–7
Hood DD, Mallak KA, Eisenach JC, et al. Interaction between intrathecal neostigmine and epidural clonidine in human volunteers. Anesthesiology 1996; 85(2): 315–25
Pellerin M, Hardy F, Abergel A, et al. Chronic refractory pain in cancer patients. Value of the spinal injection of lysine acetylsalicylate: 60 cases [in French]. Presse Med 1987; 16(30): 1465–8
Naji P, Farschtschian M, Wilder-Smith OH, et al. Epidural droperidol and morphine for postoperative pain. Anesth Analg 1990; 70(6): 583–8
Wilder-Smith CH, Wilder-Smith OH, Farschtschian M, et al. Epidural droperidol reduces the side effects and duration of analgesia of epidural sufentanil. Anesth Analg 1994; 79(1): 98–104
Bach V, Carl P, Ravlo O, et al. Potentiation of epidural opioids with epidural droperidol: a one year retrospective study. Anaesthesia 1986; 41(11): 1116–9
Grip G, Svensson BA, Gordh Jr T, et al. Histopathology and evaluation of potentiation of morphine-induced antinociception by intrathecal droperidol in the rat. Acta Anaesthesiol Scand 1992; 36(2): 145–52
Geurts JW, Kallewaard JW, Richardson J, et al. Targeted methylpredisolone acetate/hyaluronidase/clonidine injection after diagnostic epiduroscopy for chronic sciatica, one year follow up study. Reg Anesth Pain Med 2002; 27: 343–52
Richardson J, McGurgan P, Cheema S, et al. Spinal endoscopy in chronic low back pain with radiculopathy a prospective case series. Anaesthesia 2001; 56: 454–60
North RB, Kidd DH, Piantadosi S. Spinal cord stimulation versus reoperation for failed back surgery syndrome: a prospective, randomized study design. Acta Neurochir Suppl 1995; 64: 106–8
Harke H, Gretenkort P, Ladsleif HU, et al. Spinal cord stimulation in postherpetic neuralgia and in acute herpes zoster pain. Anesth Analg 2002; 94: 694–700
Spincemaille GH, de Vet HC, Ubbink DT, et al. The results of spinal cord stimulation in critical limb ischaemia: a review. Eur J Vasc Endovasc Surg 2002; 21: 99–105
Merry AF, Smith WM, Anderson DJ, et al. Cost effectiveness of spinal cord stimulation in patients with intractable angina. N Z Med J 2001; 114: 179–81
McQuay HJ, Moore RA. Using numerical results from systematic reviews in clinical practice. Ann Intern Med 1997; 126(9): 712–20
Collins SL, Moore RA, McQuay HJ, et al. Antidepressants and anticonvulsants for diabetic neuropathy and postherpetic neuralgia: a quantitative systematic review. J Pain Symptom Manage 2000; 20: 449–58
Kvinesdal B, Molin J, Froland A, et al. Imipramine treatment of painful diabetic neuropathy. JAMA 1984; 251(13): 1727–30
Sindrup SH, Ejlertsen B, Froland A, et al. Imipramine treatment in diabetic neuropathy: relief of subjective symptoms without changes in peripheral and autonomic nerve function. Eur J Clin Pharmacol 1989; 37(2): 151–3
Young RJ, Clarke BF. Pain relief in diabetic neuropathy; the effectiveness of imipramine and related drugs. Diabetic Med 1985; 2: 363–6
Sindrup SH, Gram LF, Brosen F, et al. The selective serotonin reuptake inhibitor paroxetine is effective in the treatment of diabetic neuropathy symptoms. Pain 1990; 42(2): 35–44
Sindrup SH, Tuxen C, Gram LF, et al. Lack of effect of mianserin on the symptoms of diabetic neuropathy. Eur J Clin Pharmacol 1992; 43: 251–5
Sindrup SH, Gram LF, Skjold T, et al. Clomipramine vs desi-pramine vs placebo in the treatment of diabetic neuropathy symptoms: a double-blind cross-over study. Br J Clin Pharmacol 1990; 30(5): 683–91
Max MB, Lynch SA, Muir J, et al. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic neuropathy. N Engl J Med 1992; 326(19): 1250–6
Vrethem M, Boivie J, Arnqvist H, et al. A comparison a amitriptyline and maprotiline in the treatment of painful polyneuropathy in diabetics and nondiabetics. Clin J Pain 1997; 13(4): 313–23
Sindrup SH, Bjerre U, Dejgaard A, et al. The selective serotonin re-uptake inhibitor citalopram relieves the symptoms of diabetic neuropathy. Clin Pharmacol Ther 1992; 52: 547–52
Mendel C, Klein R, Campbell D, et al. A trial of amitriptyline and fluphenazine in the treatment of painful diabetic neuropathy. JAMA 1986; 255: 637–9
Gomez-Perez FJ, Rull JA, Dies H, et al. Nortriptyline and fluphenazine in the symptomatic treatment of diabetic neuropathy: a double blind cross over study. Pain 1985; 23: 395–400
Watson CP, Evans RJ, Reed K, et al. Amitriptyline versus placebo in postherpetic neuralgia. Neurology 1982; 32(6): 671–3
Max MB, Culnane M, Schafer SC, et al. Amitriptyline relieves diabetic neuropathy pain in patients with normal or depressed mood. Neurology 1987; 37(4): 589–96
Kishore-Kumar R, Max RB, Schafer SC, et al. Desipramine relieves postherpetic neuralgia. Clin Pharmacol Ther 1990; 47: 305–12
Bowsher D. The management of central post stroke pain. Postgrad Med J 1995; 71: 598–604
Leijon G, Boivie J. Central post stroke pain — a controlled trial of amitriptyline and carbamazepine. Pain 1989; 36: 27–36
Bryson HM, Wilde MI. Amitriptyline. A review of its pharmacological properties and therapeutic uses in chronic pain states. Drugs Aging 1996; 8(6): 459–76
Watson CP, Vernich L, Chipman M, et al. Nortriptyline versus amitriptyline in postherpetic neuralgia, a randomised trial. Neurology 1998; 51: 1166–71
Sindrup S, Jensen T. Efficacy of pharmacological treatments of neuropathic pain: an update and effect related to mechanism of drug action. Pain 1999; 83: 389–400
Campbell FG, Graham JG, Zilkha KJ. Clinical trial of carbam-azepine (Tegretol) in trigeminal neuralgia. J Neurol Neurosurg Psychiatry 1966; 29(3): 265–7
Killian JM, Fromm GH. Carbamazepine in the treatment of neuralgia: use of side effects. Arch Neurol 1968; 19(2): 129–36
Nicol CF. A four year double-blind study of Tegretol in facial pain. Headache 1969; 9(1): 54–7
Rull JA, Quibrera R, Gonzales-Millan H, et al. Symptomatic treatment of peripheral diabetic neuropathy with carbamazepine (Tegretol): double blind crossover trial. Diabetologia 1969; 5(4): 215–8
Jensen TS. Anticonvulsants in neuropathic pain. Eur J Pain 2002; 6 Suppl. A: 61–8
Goodman LS, Limbird LE, Milinoff PB, et al., editors. 9th ed. Goodman and Gilman’s: The pharmacological basis of therapeutics. New York (NY): McGraw-Hill Professional, 1996
McQuay H, Carroll D, Jadad AR, et al. Anticonvulsant drugs for management of pain: a systematic review. BMJ 1995; 311(7012): 1047–52
Saudek CD, Werns S, Reidenberg MM. Phenytoin in the treatment of diabetic symmetrical polyneuropathy. Clin Pharmacol Ther 1977; 22(2): 196–9
Chadda VS, Mathur MS. Double blind study of the effects of diphenylhydantoin sodium on diabetic neuropathy. J Assoc Physicians India 1978; 26(5): 403–6
Backonja M, Beydoun A, Edwards KR, et al. Gabapentin for the symptomatic treatment of painful neuropathy in patients with diabetes mellitus: a randomized controlled trial. JAMA 1998; 280(21): 1831–6
Rowbotham M, Harden N, Stacey B, et al. Gabapentin for the treatment of postherpetic neuralgia: a randomized controlled trial. JAMA 1998; 280(21): 1837–42
Grorsen K, Schott C, Herman R, et al. Gabapentin in the treatment of painful diabetic neuropathy: a placebo controlled, double blind crossover trial. J Neurol Neurosurg Psychiatry 1999; 66: 251–2
Morello CM, Leckband SG, Stoner CB. Randomized double blind study comparing the efficacy of gabapentin with amitriptyline on diabetic peripheral neuropathy pain. Arch Intern Med 1999; 59: 1931–7
Perucca E. The clinical pharmacokinetics of the new antiepileptic drugs. Epilepsia 1999; 40 Suppl. 9: S7–S13
Gatti G, Bonomi I, Jannuzzi G, et al. The new antiepileptic drugs: pharmacological and clinical aspects. Current Pharmaceutical Design 2000; 6: 839–60
Canavero S, Bonicalz V. Lamotrigine control of central pain. Pain 1996; 68: 179–81
Harbison J, Dennehy F, Keating D. Lamotrigine for pain with hyperalgesia. Irish Med J 1997; 90: 56
Eisenberg E, Lurie Y, Braker C, et al. Lamotrigine reduces painful diabetic neuropathy: a randomized, controlled study. Neurology 2001; 57: 505–9
Vestergaard K, Andersen G, Gottrup H, et al. Lamotrigine for central post stroke pain: a randomized controlled trial. Neurology 2001; 56: 184–90
Finnerup NB, Sindrup SH, Bach FW, et al. Lamotrigine in spinal cord injury pain: a randomized controlled trial. Pain 2002; 96: 375–83
McCleane G. 200mg daily of lamotrigine has no analgesic effect on neuropathic pain: a randomized double-blind, placebo controlled trial. Pain 1999; 83: 105–7
Zakrzewska JM, Chaudhry Z, Nurmikko TJ, et al. Lamotrigine (Lamictal) in refractory trigeminal neuralgia: results from a double-blind placebo controlled crossover trial. Pain 1997; 73(2): 223–30
Drewes AM, Andreasen A, Poulson LH. Valproate for treatment of chronic central pain after spinal cord injury: a double blind cross over study. Paraplegia 1994; 32: 565–9
Mao J, Chen LL. Systemic lidocaine for neuropathic pain relief. Pain 2000; 87: 7–17
Baranowski P, De Courcey J, Bonello E. A trial of intravenous lidocaine on the pain and allodynia of postherpetic neuralgia. J Pain and Symptom Manage 1999; 17: 429–33
Brose WG, Cousins MJ. Subcutaneous lidocaine for treatment of neuropathic cancer pain. Pain 1991; 45: 145–8
Wright JM, Oki JC, Graves III L. Mexiletine in the symptomatic treatment of diabetic peripheral neuropathy. Ann Pharmacother 1997; 31(1): 29–34
Oskarsson P, Ljunggren JG, Lins PE. Efficacy and safety of mexiletine in the treatment of. painful diabetic neuropathy. The Mexiletine Study Group. Diabetes Care 1997; 20(10): 1594–7
Stracke H, Meyer UE, Schumacher HE, et al. Mexiletine in the treatment of diabetic neuropathy. Diabetes Care 1992; 15(11): 1550–5
Kalso E, Tramer MR, McQuay HJ, et al. Systemic local-anaesthetic-type drugs in chronic pain: a systematic review. Eur J Pain 1998; 2(1): 3–14
Raffa RB, Friderichs E, Reimann W, et al. Opioid and non-opioid components independently contribute to the mechanism of action of tramadol and ‘atypical opioid analgesics’. J Pharmacol and Exp Ther 1992; 260: 275–85
Harati Y, Gooch C, Swenson M, et al. Double-blind randomized trial of tramadol for the treatment of the pain of diabetic neuropathy. Neurology 1998; 50(6): 1842–6
Sindrup SH, Andersen G, Madsen C, et al. Tramadol relieves pain and allodynia in polyneuropathy: a randomised, doubleblind, controlled trial. Pain 1999; 83(1): 85–90
Therapeutic Goods Administration. Tramadol and serotonin syndrome. Australian Adverse Drug Reactions Bulletin [online]: 2001, 20(4). Available from URL: http://www.health.gov.au/tga/docs/html/aadrbltn/aadr0112.htm [Accessed 2002 Dec 3]
Shipton EA. Tramadol-present and future. Anaesth Intensive Care 2000; 28: 263–74
Arner S, Meyerson BA. Lack of analgesic effect of opioids on neuropathic and idiopathic forms of pain. Pain 1988; 33(1): 11–23
Davies HT, Crombie LK, Macrae WA. Polarised views on treating neurogenic pain. Pain 1993; 54(3): 341–6
Portenoy RK, Foley KM, Inturrisi CE. The nature of opioid responsiveness and its implications for neuropathic pain: new hypotheses derived from studies of opioid infusions. Pain 1990; 43(3): 273–86
Watson CP, Babul N. Efficacy of oxycodone in neuropathic pain: a randomized trial in postherpetic neuralgia. Neurology 1998; 50(6): 1837–41
Cheville A, Chen A, Oster G, et al. A randomised trial of controlled release oxycodone during inpatient rehabilitation following unilateral total knee arthroplasty. J Bone and Joint Surg 2001; 83-A: 572–6
The Capsaicin Study Group. Treatment of painful diabetic neuropathy with topical capsaicin: a multicenter, doubleblind, vehicle-controlled study. Arch Intern Med 1991; 151(11): 2225–9
Low PA, Opfer-Gehrking TL, Dyck PJ, et al. Double-blind, placebo-controlled study of the application of capsaicin cream in chronic distal painful polyneuropathy. Pain 1995; 62(2): 163–8
Bernstein JE, Korman NJ, Bickers DR, et al. Topical capsaicin treatment of chronic postherpetic neuralgia. J Am Acad Dermatol 1989; 21 (2 Pt 1): 265–70
Watson CP, Tyler KL, Bickers DR, et al. A randomized vehicle-controlled trial of topical capsaicin in the treatment of postherpetic neuralgia. Clin Ther 1993; 15(3): 510–26
Zhang WY, Li Wan Po A. The effectiveness of topically applied capsaicin: a meta-analysis. Eur J Clin Pharmacol 1994; 46: 517–22
Rowbotham MC, Davies PS, Fields HL. Topical lidocaine gel relieves postherpetic neuralgia. Ann Neurol 1995; 37(2): 246–53
Rowbotham MC, Davies PS, Verkempinck C, et al. Lidocaine patch: double-blind controlled study of a new treatment method for post-herpetic neuralgia. Pain 1996; 65(1): 39–44
Devers A, Galer BS. Topical lidocaine patch relieves a variety of neuropathic pain conditions: an open label study. Clin J Pain 2000; 16: 205–8
Graziotti PJ, Goucke CR. The use of oral opioids in patients with chronic non-cancer pain: management strategies. Med J Aust 1997; 167(1): 30–4
Acknowledgements
Dr Ahmad has received no funding and has no conflicts of interest directly relevant to this manuscript. Dr Goucke has also received no funding and has served on medical advisory boards for Pfizer, MundiPharma and CSL.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmad, M., Goucke, C.R. Management Strategies for the Treatment of Neuropathic Pain in the Elderly. Drugs Aging 19, 929–945 (2002). https://doi.org/10.2165/00002512-200219120-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-200219120-00004