Abstract
Background
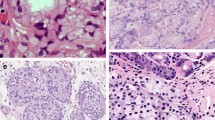
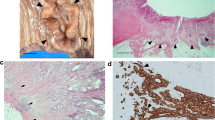
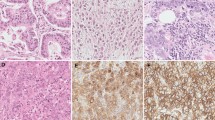
Recent studies have identified loss of stromal caveolin-1 (Cav-1) expression as a new prognostic histological characteristic in various types of human cancers. However, the clinical and pathological significance of stromal Cav-1 expression in esophageal squamous cell carcinoma (ESCC) remains largely unknown. We examined Cav-1 expression in both tumor and stromal cells in ESCC tissue by immunohistochemical analysis to evaluate its clinicopathological significance and prognostic value.
Methods
A total of 110 patients with ESCC who underwent surgical resection were included in this study. The expression of Cav-1 in both tumor and stromal cells in esophageal tumor tissues was examined immunohistochemically.
Results
Cav-1 expression was found in the cytoplasm of both tumor and stromal cells. Tumor Cav-1 overexpression was observed in 37.3 % tumors, which correlated to deeper tumor invasion (p = 0.038). Down-regulation of stromal Cav-1 expression was observed in 40.9 % tumors. The stromal Cav-1 down-regulation group had more lymph node metastases and more locoregional recurrences than those with higher expression (p = 0.020 and p = 0.002, respectively). In addition, down-regulation of stromal Cav-1 expression was associated with shorter disease-free survival (p < 0.001) and overall survival (p < 0.001). Multivariate analysis revealed that down-regulation of stromal Cav-1 expression was an independent prognostic factor for both disease-free survival (p = 0.028) and overall survival (p = 0.007).
Conclusions
Down-regulation of stromal Cav-1 expression in ESCC had high malignant potential. It predicts high-risk of lymph node metastases and locoregional recurrence, and it could be a powerful prognostic marker for patients with ESCC.


Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Bonnetain F, Bouche O, Michel P, et al. A comparative longitudinal quality of life study using the Spitzer quality of life index in a randomized multicenter phase III trial (FFCD 9102): chemoradiation followed by surgery compared with chemoradiation alone in locally advanced squamous resectable thoracic esophageal cancer. Ann Oncol. 2006;17:827–34.
Rice TW, Rusch VW, Ishwaran H, Blackstone EH. Cancer of the esophagus and esophagogastric junction: data-driven staging for the seventh edition of the American Joint Committee on Cancer/International Union Against Cancer Cancer staging manuals. Cancer. 2010;116:3763–73.
Chen SB, Weng HR, Wang G, et al. Prognostic factors and outcome for patients with esophageal squamous cell carcinoma underwent surgical resection alone: evaluation of the seventh edition of the american joint committee on cancer staging system for esophageal squamous cell carcinoma. J Thorac Oncol. 2013;8:495–501.
Hou X, Wei JC, Xu Y, et al. The positive lymph node ratio predicts long-term survival in patients with operable thoracic esophageal squamous cell carcinoma in China. Ann Surg Oncol. 2013;20:1653–59.
Liu Q, Tan Z, Lin P, et al. Impact of the number of resected lymph nodes on postoperative survival of patients with node-negative oesophageal squamous cell carcinoma. Eur J Cardiothorac Surg. 2013. doi:10.1093/ejcts/ezt097.
Mercier I, Jasmin JF, Pavlides S, et al. Clinical and translational implications of the caveolin gene family: lessons from mouse models and human genetic disorders. Lab Invest. 2009;89:614–23.
Rajab A, Straub V, McCann LJ, et al. Fatal cardiac arrhythmia and long-QT syndrome in a new form of congenital generalized lipodystrophy with muscle rippling (CGL4) due to PTRF-CAVIN mutations. PLoS Genet. 2010;6:e1000874.
Sotgia F, Del Galdo F, Casimiro MC, et al. Caveolin-1−/− null mammary stromal fibroblasts share characteristics with human breast cancer-associated fibroblasts. Am J Pathol. 2009;174:746–61.
Goetz JG, Lajoie P, Wiseman SM, Nabi IR. Caveolin-1 in tumor progression: the good, the bad and the ugly. Cancer Metastasis Rev. 2008;27:715–35.
Di Vizio D, Morello M, Sotgia F, Pestell RG, Freeman MR, Lisanti MP. An absence of stromal caveolin-1 is associated with advanced prostate cancer, metastatic disease and epithelial Akt activation. Cell Cycle. 2009;8:2420–4.
Joo HJ, Oh DK, Kim YS, Lee KB, Kim SJ. Increased expression of caveolin-1 and microvessel density correlates with metastasis and poor prognosis in clear cell renal cell carcinoma. BJU Int. 2004;93:291–6.
Kato K, Hida Y, Miyamoto M, et al. Overexpression of caveolin-1 in esophageal squamous cell carcinoma correlates with lymph node metastasis and pathologic stage. Cancer. 2002;94:929–33.
Steffens S, Schrader AJ, Blasig H, et al. Caveolin 1 protein expression in renal cell carcinoma predicts survival. BMC Urol. 2011;11:25.
Wiechen K, Diatchenko L, Agoulnik A, et al. Caveolin-1 is down-regulated in human ovarian carcinoma and acts as a candidate tumor suppressor gene. Am J Pathol. 2001;159:1635–43.
Witkiewicz AK, Dasgupta A, Sotgia F, et al. An absence of stromal caveolin-1 expression predicts early tumor recurrence and poor clinical outcome in human breast cancers. Am J Pathol. 2009;174:2023–34.
Wu KN, Queenan M, Brody JR, Potoczek M, Sotgia F, Lisanti MP, Witkiewicz AK. Loss of stromal caveolin-1 expression in malignant melanoma metastases predicts poor survival. Cell Cycle. 2011;10:4250–5.
Sotgia F, Martinez-Outschoorn UE, Howell A, Pestell RG, Pavlides S, Lisanti MP. Caveolin-1 and cancer metabolism in the tumor microenvironment: markers, models, and mechanisms. Annu Rev Pathol. 2012;7:423–67.
Zhao X, He Y, Gao J, Fan L, Li Z, Yang G, Chen H. Caveolin-1 expression level in cancer associated fibroblasts predicts outcome in gastric cancer. PLoS One. 2013;8:e59102.
Amatangelo MD, Bassi DE, Klein-Szanto AJ, Cukierman E. Stroma-derived three-dimensional matrices are necessary and sufficient to promote desmoplastic differentiation of normal fibroblasts. Am J Pathol. 2005;167:475–88.
Bremnes RM, Al-Shibli K, Donnem T, et al. The role of tumor-infiltrating immune cells and chronic inflammation at the tumor site on cancer development, progression, and prognosis: emphasis on non–small cell lung cancer. J Thorac Oncol. 2011;6:824–33.
Erkan M, Reiser-Erkan C, Michalski CW, Kong B, Esposito I, Friess H, Kleeff J. The impact of the activated stroma on pancreatic ductal adenocarcinoma biology and therapy resistance. Curr Mol Med. 2012;12:288–303.
Kong X, Li L, Li Z, Xie K. Targeted destruction of the orchestration of the pancreatic stroma and tumor cells in pancreatic cancer cases: molecular basis for therapeutic implications. Cytokine Growth Factor Rev. 2012;23:343–56.
Onishi T, Hayashi N, Theriault RL, Hortobagyi GN, Ueno NT. Future directions of bone-targeted therapy for metastatic breast cancer. Nat Rev Clin Oncol. 2010;7:641–51.
Wang K, Ma W, Wang J, et al. Tumor-stroma ratio is an independent predictor for survival in esophageal squamous cell carcinoma. J Thorac Oncol. 2012;7:1457–61.
Simpkins SA, Hanby AM, Holliday DL, Speirs V. Clinical and functional significance of loss of caveolin-1 expression in breast cancer-associated fibroblasts. J Pathol. 2012;227:490–8.
Razani B, Zhang XL, Bitzer M, von Gersdorff G, Bottinger EP, Lisanti MP. Caveolin-1 regulates transforming growth factor (TGF)-beta/SMAD signaling through an interaction with the TGF-beta type I receptor. J Biol Chem. 2001;276:6727–38.
Xia H, Khalil W, Kahm J, Jessurun J, Kleidon J, Henke CA. Pathologic caveolin-1 regulation of PTEN in idiopathic pulmonary fibrosis. Am J Pathol. 2010;176:2626–37.
Goetz JG, Minguet S, Navarro-Lerida I, et al. Biomechanical remodeling of the microenvironment by stromal caveolin-1 favors tumor invasion and metastasis. Cell. 2011;146:148–63.
Salem AF, Whitaker-Menezes D, Lin Z, et al. Two-compartment tumor metabolism: autophagy in the tumor microenvironment and oxidative mitochondrial metabolism (OXPHOS) in cancer cells. Cell Cycle. 2012;11:2545–56.
Witkiewicz AK, Kline J, Queenan M, et al. Molecular profiling of a lethal tumor microenvironment, as defined by stromal caveolin-1 status in breast cancers. Cell Cycle. 2011;10:1794–809.
Tang W, Chang SB, Hemler ME. Links between CD147 function, glycosylation, and caveolin-1. Mol Biol Cell. 2004;15:4043–50.
Du ZM, Hu CF, Shao Q, et al. Upregulation of caveolin-1 and CD147 expression in nasopharyngeal carcinoma enhanced tumor cell migration and correlated with poor prognosis of the patients. Int J Cancer. 2009;125:1832–41.
Kim HN, Chung HS. Caveolin-1 inhibits membrane-type 1 matrix metalloproteinase activity. BMB Rep. 2008;41:858–62.
Williams TM, Medina F, Badano I, et al. Caveolin-1 gene disruption promotes mammary tumorigenesis and dramatically enhances lung metastasis in vivo. Role of Cav-1 in cell invasiveness and matrix metalloproteinase (MMP-2/9) secretion. J Biol Chem. 2004;279:51630–46.
Acknowledgment
Our research was supported by the Shandong Province Science and Technology Key Project Fund (2012GSF11852). We thank Dr. T. G. Zhang (Department of Pathology, Qilu Hospital of Shandong University, China) and Dr. J. H. Zhen (Department of Pathology, Qilu Hospital of Shandong University, China) for their expert suggestions and technical assistance.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jia, Y., Wang, N., Wang, J. et al. Down-Regulation of Stromal Caveolin-1 Expression in Esophageal Squamous Cell Carcinoma: A Potent Predictor of Lymph Node Metastases, Early Tumor Recurrence, and Poor Prognosis. Ann Surg Oncol 21, 329–336 (2014). https://doi.org/10.1245/s10434-013-3225-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-3225-x