Abstract
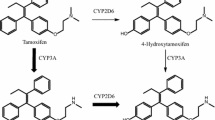
The antiestrogen tamoxifen is extensively metabolized in patients to form a series of compounds with altered affinity for estrogen receptors (ERs), the primary target of this drug. Furthermore, these metabolites exhibit a range of partial agonist and antagonist activities for ER mediated effects that do not depend directly on their absolute affinity for ERs. Thus, clinical response to tamoxifen therapy is likely to depend on the aggregate effect of these different metabolites resulting from their abundance in the patient, their affinity for the receptors, and their agonist/antagonist profile. A recent study has shown that plasma concentrations of the tamoxifen metabolite 4-hydroxy-N-desmethyl tamoxifen (endoxifen), in patents undergoing tamoxifen therapy, are dependent on the cytochrome P450 (CYP) 206 genotype of the patient and that medications commonly prescribed to patients on tamoxifen therapy can also inhibit endoxifen production. In this study we characterized the properties of this metabolite with respect to binding to ERs, ability to inhibit estrogen stimulated breast cancer cell proliferation and the regulation of estrogen responsive genes. We demonstrate that endoxifen has essentially equivalent activity to the potent metabolite 4-hydroxy tamoxifen (4-OH-tam) often described as the active metabolite of this drug. Since plasma levels of endoxifen in patients with functional CYP2D6 frequently exceed the levels of 4-OH-tam, it seems likely that endoxifen is at least as important as 4-OH-tam to the overall activity of this drug and suggests that CYP2D6 status and concomitant administration of drugs that inhibit CYP2D6 activity have the potential to affect response to tamoxifen therapy.
Similar content being viewed by others
References
American Cancer Society: Cancer Facts and Figures, 2002
Osborne CK: Tamoxifen in the treatment of breast cancer. N Engl J Med 339: 1609–1618, 1998
Furr BJ, Jordan VC: The pharmacology and clinical uses of tamoxifen. Pharmacol Ther 25: 127–205, 1984
Stearns V, Johnson MD, Rae JM et al.: Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J Natl Cancer Inst 95: 1758–1764, 2003
Jeppesen U, Gram LF, Vistisen K, Loft S, Poulsen HE, Brosen K: Dose-dependent inhibition of CYP1A2, CYP2C19 and CYP2D6 by citalopram, fluoxetine, fluvoxamine and paroxetine. Eur J Clin Pharmacol 51: 73–78, 1996
Leonessa F, Jacobson M, Boyle B, Lippman J, McGarvey M, Clarke R: Effect of tamoxifen on the multidrug-resistant phenotype in human breast cancer cells: isobologram, drug accumulation, andM(r) 170,000 glycoprotein (gp170) binding studies. Cancer Res 54: 441–447, 1994
Edwards DP, McGuire WL: 17 alpha-estradiol is a biologically active estrogen in human breast cancer cells in tissue culture. Endocrinology 107: 884–891, 1980
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254, 1976
Johnson MD, Westley BR, May FE: Oestrogenic activity of tamoxifen and its metabolites on gene regulation and cell proliferation in MCF-7 breast cancer cells. Br J Cancer 59: 727–738, 1989
Jordan VC: Metabolites of tamoxifen in animals and man: identification, pharmacology, and significance. Breast Cancer Res Treat 2: 123–138, 1982
Patterson JS, Settatree RS, Adam AK, Kemp JV: Serum concentrations of tamoxifen and major metabolites during longterm Nolvadex therapy, correlated with clinical response. In: Mouridsen HT, Palshoff T (eds) Breast Cancer: Experimental and Clinical Aspects. Pergamon Press, Oxford, UK, 1980, p 89
Desta Z, Ward BA, Soukhova N, Flockhart DA: Comprehensive kinetic characterization of tamoxifen primary demethylation and hydroxylation by the human cytochrome P450s. Drug Metab Rev 25(Suppl. 2): 189 (abstract #377), 2003
Fisher B, Costantino JP, Wickerham DL et al.: Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst 90: 1371–1388, 1998
Stearns V, Ullmer L, Lopez JF, Smith Y, Isaacs C, Hayes D: Hot flushes. Lancet 360: 1851–1861, 2002
Stearns V, Beebe KL, Iyengar M, Dube E: Paroxetine controlled release in the treatment of menopausal hot flashes: a randomized controlled trial. JAMA 289: 2827–2834, 2003
Lien EA, Solheim E, Kvinnsland S, Ueland PM: Identification of 4-hydroxy-N-desmethyltamoxifen as a metabolite of tamoxifen in human bile. Cancer Res 48: 2304–2308, 1988
Lien EA, Solheim E, Lea OA, Lundgren S, Kvinnsland S, Ueland PM: Distribution of 4-hydroxy-N-desmethyltamoxifen and other tamoxifen metabolites in human biological fluids during tamoxifen treatment. Cancer Res 49: 2175–2183, 1989
Shiau AK, Barstad D, Radek JT et al.: Structural characterization of a subtype-selective ligand reveals a novel mode of estrogen receptor antagonism. Nat Struct Biol 9: 359–364, 2002
Lee KH, Ward BA, Desta Z, Flockhart DA, Jones DR: Quantification of tamoxifen and three metabolites in plasma by high-performance liquid chromatography with fluorescence detection: application to a clinical trial. J Chromatogr B Analyt Technol Biomed Life Sci 791: 245–253, 2003
Marez D, Legrand M, Sabbagh N et al.: Polymorphism of the cytochrome P450 CYP2D6 gene in a European population: characterization of 48 mutations and 53 alleles, their frequencies and evolution. Pharmacogenetics 7: 193–202, 1997
Broly F, Gaedigk A, Heim M, Eichelbaum M, Morike K, Meyer UA: Debrisoquine/sparteine hydroxylation genotype and phenotype: analysis of common mutations and alleles of CYP2D6 in a European population. DNA Cell Biol 10: 545–558, 1991
Rae JM, Cordero KE, Scheys JO, Lippman ME, Flockhart DA, Johnson MD: Genotyping for polymorphic drug metabolizing enzymes from paraffin-embedded and immunohistochemically stained tumor samples. Pharmacogenetics 13: 501–507, 2003
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Johnson, M.D., Zuo, H., Lee, KH. et al. Pharmacological Characterization of 4-hydroxy-N-desmethyl Tamoxifen, a Novel Active Metabolite of Tamoxifen. Breast Cancer Res Treat 85, 151–159 (2004). https://doi.org/10.1023/B:BREA.0000025406.31193.e8
Issue Date:
DOI: https://doi.org/10.1023/B:BREA.0000025406.31193.e8