Abstract
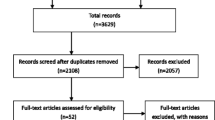
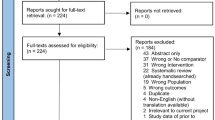
The aim of this review is to assess the role of hepatic lymphadenectomy in patients undergoing liver resection for colorectal metastases. Meta-analysis of trials identified by a systematic literature search of the Medline, Embase and Central databases was performed. There were no randomized controlled trials which assessed the survival benefit to patients undergoing liver resection for colorectal metastases of either a routine or a ‘selective’ lymphadenectomy. The prevalence of nodal metastases after lymphadenectomy was 8.4 %. The overall 3-year and 5-year survival rates in patients with hepatic lymph node metastases undergoing hepatectomy were 21.8 % (63 of 288 patients) and 8.5 % (27 of 315 patients), respectively, compared with 58.2 % (1,366 of 2,346 patients) and 47.5 % (1,717 of 3,609 patients) in patients undergoing hepatectomy who had no hepatic nodal metastases. The odds ratios for 3-year and 5-year mortality in node positive disease compared with node negative disease were 4.54 (95 % confidence interval 3.15–6.54) and 6.33 (95 % confidence interval 4.28–9.36), respectively. In conclusion, long-term survival rates are low in patients undergoing hepatectomy with hepatic lymph node metastases. The poor outcome is irrespective of whether the nodal metastases are discovered following routine lymphadenectomy or are detected because of pathological enlargement. Further trials in this patient group are required.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Rodgers MS, McCall JL. Surgery for colorectal liver metastases with hepatic lymph node involvement: a systematic review. Br J Surg. 2000;87(9):1142–55.
Poston GJ, Adam R, Alberts S, et al. OncoSurge: a strategy for improving resectability with curative intent in metastatic colorectal cancer. J Clin Oncol. 2005;23(28):7125–34.
Jaeck D, Nakano H, Bachellier P, et al. Significance of hepatic pedicle lymph node involvement in patients with colorectal liver metastases: a prospective study. Ann Surg Oncol. 2002;9(5):430–8.
Beckurts KT, Holscher AH, Thorban S, et al. Significance of lymph node involvement at the hepatic hilum in the resection of colorectal liver metastases. Br J Surg. 1997;84(8):1081–4.
Laurent C, Sa Cunha A, Rullier E, et al. Impact of microscopic hepatic lymph node involvement on survival after resection of colorectal liver metastasis. J Am Coll Surg. 2004;198(6):884–91.
• Alberts SR, Wagman LD. Chemotherapy for colorectal cancer liver metastases. Oncologist. 2008;13(10):1063–73. The authors reviewed past and present chemotherapy regimens for metastatic colorectal cancer.
• Adam R, de Haas RJ, Wicherts DA, et al. Is hepatic resection justified after chemotherapy in patients with colorectal liver metastases and lymph node involvement? J Clin Oncol. 2008;26(22):3672–80. The authors demonstrated 5-year survival in a highly selected group of patients.
• Pulitanò C, Bodingbauer M, Aldrighetti L, et al. Liver resection for colorectal metastases in presence of extrahepatic disease: results from an international multi-institutional analysis. Ann Surg Oncol. 2011;18(5):1380–8. This is a report of a relatively large series where the authors reviewed their data on nodal involvement in metastatic colorectal cancer patients.
• Carpizo DR, Are C, Jarnagin W, et al. Liver resection for metastatic colorectal cancer in patients with concurrent extrahepatic disease: results in 127 patients treated at a single center. Ann Surg Oncol. 2009;16(8):2138–46. This is a report of a relatively large series where the authors reviewed their data on concurrent extrahepatic disease in metastatic colorectal cancer patients.
Lupinacci RM, Coelho FF, Kruger J, et al. Hilar lymph node involvement in colorectal cancer liver metastases – an overview. J Gastrointest Digest Sys. 2011;S6:002. doi:10.4172/jgds.S6-002.
Bennett JJ, Schmidt CR, Klimstra DS, et al. Perihepatic lymph node micrometastases impact outcome after partial hepatectomy for colorectal metastases. Ann Surg Oncol. 2008;15(4):1130–6.
Gurusamy KS, Imber C, Davidson BR. Management of the hepatic lymph nodes during resection of liver metastases from colorectal cancer: a systematic review. HPB Surg. 2008;2008:684150. doi:10.1155/2008/684150.
Review Manager version 5.2. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration; 2011.
Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med. 1998;17(24):2815–34.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177–88.
Demets DL. Methods for combining randomized clinical trials: strengths andlimitations. Stat Med. 1987;6(3):341–50.
Ambiru S, Miyazaki M, Isono T, et al. Hepatic resection for colorectal metastases: analysis of prognostic factors. Dis Colon Rectum. 1999;42(5):632–9.
Kokudo N, Seki M, Ohta H, et al. Effects of systemic and regional chemotherapy after hepatic resection for colorectal metastases. Ann Surg Oncol. 1998;5(8):706–12.
Nakamura S, Yokoi Y, Suzuki S, et al. Results of extensive surgery for liver metastases in colorectal carcinoma. Br J Surg. 1992;79(1):35–8.
Rosen CB, Nagorney DM, Taswell HF, et al. Perioperative blood transfusion and determinants of survival after liver resection for metastatic colorectal carcinoma. Ann Surg. 1992;216(4):493–504.
Harms J, Obst T, Thorban S, et al. The role of surgery in the treatment of liver metastases for colorectal cancer patients. Hepatogastroenterology. 1999;46(28):2321–8.
Yasui K, Hirai T, Kato T, et al. Major anatomical hepatic resection with regional lymph node dissection for liver metastases from colorectal cancer. J Hepatobiliary Pancreat Surg. 1995;2:103–7.
Sanchez-Cespedes M, Esteller M, Hibi K, et al. Molecular detection of neoplastic cells in lymph nodes of metastatic colorectal cancer patients predicts recurrence. Clin Cancer Res. 1999;5(9):2450–4.
Minagawa M, Makuuchi M, Torzilli G, et al. Extension of the frontiers of surgical indications in the treatment of liver metastases from colorectal cancer- long term results. Ann Surg. 2000;231(4):487–99.
Aoki T, Umekita N, Tanaka S, et al. Prognostic value of concomitant resection of extrahepatic disease in patients with liver metastases of colorectal origin. 2008;143(6):706–14.
Oussoultzoglou E, Rosso E, Fuchshuber P, et al. Perioperative carcinoembryonic antigen measurements to predict curability after liver resection for colorectal metastases. Arch Surg. 2008;143(12):1150–8.
Jonas S, Thelen A, Benckert C, et al. Extended resections of liver metastases from colorectal cancer. World J Surg. 2007;31(3):511–21.
Settmacher U, Dittmar Y, Knösel T, et al. Predictors of long-term survival in patients with colorectal liver metastases: a single center study and review of the literature. Int J Color Dis. 2011;26(8):967–81.
Ishida H, Ishibashi K, Ohsawa T, et al. Significance of hepatic lymph node metastasis in patients with unresectable synchronous liver metastasis of colorectal cancer. Int Surg. 2011;96(4):291–9.
Lupinacci RM, Herman P, Coelho FC, et al. Diagnosis and impact of hilar lymph node micrometastases on the outcome of resected colorectal liver metastasis. Hepatogastroenterology. 2013.
Grobmyer SR, Wang L, Gonen M, et al. Perihepatic lymph node assessment in patients undergoing partial hepatectomy for malignancy. Ann Surg. 2006;244(2):260–4.
Conflict of Interest
Rahul S. Koti declares he has no conflict of interest.
Constantinos Simillis declares he has no conflict of interest.
Kurinchi S. Gurusamy is supported by grants from the Wellcome Trust and the National Institute for Health Research (NIHR), and he has also received payment for preparation of a manuscript from NIHR.
Michael Jacovides declares he has no conflict of interest.
Brian R. Davidson declares he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koti, R.S., Simillis, C., Gurusamy, K.S. et al. Management of the Hepatic Lymph Nodes During Resection of Liver Metastases from Colorectal Cancer: A Systematic Review. Curr Colorectal Cancer Rep 9, 203–212 (2013). https://doi.org/10.1007/s11888-013-0165-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-013-0165-6