Abstract
Background
Psoriasis is a chronic skin disease associated with increased morbidity and mortality. Effective and safe long term treatment options are required to manage the illness successfully. A number of systemic agents are available, however, each of them has potentially significant side effects. Fumaric acid esters (FAE) are used first line in Germany for the management of moderate to severe psoriasis, however, their use in Ireland is on an unlicensed basis (Clinical and Experimental Dermatology 37:786–801, 2012).
Objectives
The purpose of this literature review is to evaluate the efficacy and safety of FAEs in the management of moderate to severe psoriasis in adult patients. The reviewer intends to systematically review all available literature on the efficacy and/or safety of fumaric acid esters in the management of moderate to severe psoriasis in adult patients.
Methods
A systematic review of the literature was performed by one reviewer. The PubMed, TRIP, Embase, and Cochrane Collaboration databases were systematically interrogated to include randomised controlled trials, cohort studies and case studies evaluating the efficacy and/or safety of FAEs in the management of moderate to severe psoriasis in adult patients. Inclusion criteria were studies which included adults over 18 years of age, with a diagnosis of moderate to severe chronic plaque psoriasis, who were treated with FAEs and no other systemic anti-psoriatic agents concurrently. Exclusion criteria were studies involving children, mild psoriasis, studies which did not include patients with chronic plaque psoriasis, the use of FAE for the management of illnesses other than psoriasis, and patients treated with more than one systemic anti-psoriatic agent concurrently.
Results
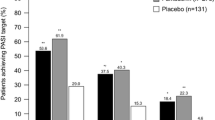
In total 19 articles were selected for review including 2 randomised placebo controlled trials, 1 non-randomised comparative study, 7 retrospective cohort studies, 2 prospective cohort studies and 7 case studies. The findings suggest that FAEs are a safe and effective treatment option for the management of moderate to severe psoriasis in adult patients. Gastrointestinal side effects may occur on treatment initiation and may be minimised by slow dose titration. Lymphocytopenia and eosinophilia are common, however, they are rarely of significance and there is no high level of evidence available to suggest a resultant increased risk of infection or malignancy. Rarely alterations of renal and hepatic function may occur, however, these are largely reversible on treatment withdrawal.
Conclusion
In conclusion, the use of FAE in the management of moderate to severe psoriasis is a promising treatment option, especially for those patients intolerant of, or unresponsive to other agents. If blood parameters are closely monitored during treatment as per the European Medicine Agencies guidelines (European Medicines Agency, ‘Updated recommendations to minimise the risk of the rare brain infection PML with Tecfidera’, http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2015/10/WC500196017.pdf, 2015) they may be safely used in practice. The licensing of FAEs in Ireland for the treatment of moderate to severe psoriasis would be desirable, increasing available treatment options.





Similar content being viewed by others
References
Altmeyer PJ, Matthes U, Pawlak F et al (1994) Antipsoriatic effect of fumaric acid derivatives: results of a multicenter double-blind study in 100 patients. J Am Acad Dermatol 30:977–981
Barth D, Simon JC, Wetzig T (2011) ‘Malignant melanoma during treatment with fumaric acid esters—coincidence or treatment related? JDDG. J der Deutschen Dermatologischen Gesellschaft 3:223–224
Brewer L, Rogers S (2007) Fumaric acid esters in the management of severe psoriasis. Clin Exp Dermatol 32:246–249
Carboni I, De Felice C, De Simoni I et al (2004) Fumaric acid esters in the treatment of psoriasis: an Italian experience. J Dermatol Treat 15:23–26
Ermis U, Weis J, Schulz JB (2013) PML in a patient treated with fumaric acid. N Engl J Med 368(17):1657–1658
Fairhurst DA, Ashcroft DM, Griffiths EM (2005) ‘Optimal Management of Severe Plaque Form of Psoriasis. Am J Clin Dermatol 6(5):283–294
Fika Z, Williams REA, Williamson DJ (2006) Fumaric acid esters in psoriasis. Br J Dermatol 154(3):567–568
Harries MJ, Chalmers RJG, Griffiths CEM (2005) Fumaric acid esters for severe psoriasis: a retrospective review of 58 cases. Br J Dermatol 153:549–551
Heelan K, Markham T (2012) Fumaric acid esters as a suitable first line treatment for severe psoriasis: an Irish experience. Clin Exp Dermatol 37:786–801
Hoefnagel JJ, Thio HB, Willemze R et al (2003) Long term safety aspects of systemic therapy with fumaric acid esters in severe psoriasis. Br J Dermatol 149:363–369
Ismail N, Collins P, Rogers S et al (2014) Drug survival of fumaric acid esters for psoriasis: a retrospective study. Br J Dermatol 171:397–402
Jennings L, Murphy GM (2009) Squamous cell carcinoma as a complication of fumaric acid ester immunosuppression. Eur Acad Dermatol Venereol 23:1445–1469
Kolbach DN, Niebor C (1992) ‘Fumaric acid therapy in psoriasis: Results and side effects of 2 years of treatment’. J Am Acad Dermatol 27(5 part 1): 769–771
Mrowietz U, Christophers E, Altmeyer P (1998) Treatment of severe psoriasis with fumaric acid esters: scientific background and guidelines for therapeutic use. Br J Dermatol 141:424–429
Naldi L, Griffiths CEM (2005) Traditional therapies in the management of moderate to severe chronic plaque psoriasis: an assessment of the benefits and risks. Br J Dermatol 152:597–615
Nugteren-Huying WM, van der Schroeff JG, Hermans J et al (1990) Fumaric acid therapy for psoriasis: a randomised, double blind, placebo controlled study. J Am Acad Dermatol 22(2):311–312
Ogilvie S, Lewis Jones S, Dawe R et al (2011) Proteinuria with fumaric acid ester treatment for psoriasis. Clin Exp Dermatol 36:632–634
Pathirana D, Ormerod AD, Saiag P et al (2009) ‘European S3- Guidelines on the systemic treatment of psoriasis vulgaris. Eur Acad Dermatol Venereol 23(2):5–70
Raschka C, Koch HJ (1999) Long term treatment of psoriasis using fumaric acid preparations can be associated with severe proximal tubular damage. Hum Exp Toxicol 18:738–739
Reich K, Thaci D, Mrowietz U et al (2009) Efficacy and safety of fumaric acid esters in the long term treatment of psoriasis- a retrospective study (FUTURE). JDDG. J der Deutschen Dermatologischen Gesellschaft 7:603–610
Reid C, Holian J, Kane D et al (2013) De Toni Fanconi Syndrome secondary to fumaric acid esters. Br J Dermatol 169(1):24
Schmitt J, Zhang Z, Wozel G et al (2008) Efficacy and tolerability of biologic and nonbiologic systemic treatments for moderate to severe psoriasis: meta-analysis of randomised controlled trials. Br J Dermatol 159:513–526
Sladden MJ, Osborne JE, Hutchinson PE (2006) Fumaric acid esters for severe psoriasis: the Leicestershire experience. Br J Dermatol 155:841–865
Thaçi D, Weisenseel P, Philipp S et al (2013) ‘Efficacy and safety of fumaric acid esters in patients with psoriasis on medication for comorbid conditions – a retrospective evaluation (FACTS). J Ger Soc Dermatol 1610–0379:439–445
Van Oosten BW, Killestein J, Barkhof F et al (2013) PML in a patient treated with dimethylfumarate from a compounding pharmacy. N Engl J Med 368(17):1658–1659
Wu Y, Mills D, Bala M (2008) Psoriasis: cardiovascular risk factors and other disease comorbidities. J Drugs Dermatol 7:373–377
European Medicines Agency (2015). ‘Updated recommendations to minimise the risk of the rare brain infection PML with Tecfidera’. Available at: http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2015/10/WC500196017.pdf. Accessed Mar 1, 2016
Acknowledgments
With special thanks to Dr Bart Ramsay, Consultant Dermatologist, University Hospital Limerick.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smith, D. Fumaric acid esters for psoriasis: a systematic review. Ir J Med Sci 186, 161–177 (2017). https://doi.org/10.1007/s11845-016-1470-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-016-1470-2