Abstract
Background
Various studies reported a higher incidence of allergic disorders, with an overreactivity of type 2 helper T-cell (Th2) immune mechanisms, in children with idiopathic steroid-sensitive nephrotic syndrome (ISSNS). However, Th2 predominance in ISSNS has not been definitively identified. To determine whether Th2 was predominant in children with ISSNS, we used paired samples to measure the type 1 helper T-cell (Th1)/Th2 ratios and serum cytokine levels secreted by Th1 and Th2.
Methods
We measured the Th1/Th2 ratios and levels of Th1- or Th2-secreted cytokines in paired samples. Fourteen children met the inclusion criteria: (1) ISSNS; (2) selectivity index < 0.1; (3) sera obtained in at least two disease phases; (4) no infection; (5) no immunosuppressants. Two control groups (group B, normal urinalysis; group C, nephrotic syndrome other than ISSNS) were included for cytokine level comparisons. Th1 and Th2 numbers were counted by three-color flow cytometry. Cytokine levels were measured by bead-based assay.
Results
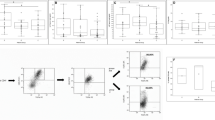
The Th1/Th2 ratio was lower in group A-1 [nephrotic-phase before steroid treatment (STx)] than in groups A-2 (remission-phase with STx) and A-3 (remission-phase without STx). Th2-secreted interleukin-5 (IL-5) levels were higher in group A-1 than in groups A-2 and A-3. There were no differences in IL-5 levels between groups A-1 and C and between groups A-3 and B.
Conclusion
Our results suggest that Th2 played a predominant role both in the Th1/Th2 ratio and in the serum IL-5 level in children with ISSNS in the nephrotic phase.


Similar content being viewed by others
References
The primary nephrotic syndrome in children. Identification of patients with minimal change nephrotic syndrome from initial response to prednisone. A report of the International Study of Kidney Disease in Children. J Pediatr. 1981;98:561–4.
Wittig HJ, Goldman AS. Nephrotic syndrome associated with inhaled allergens. Lancet. 1970;1:542–3.
Reeves WG, Cameron JS, Johansson SG, Ogg CS, Peters DK, Weller RO. Seasonal nephrotic syndrome. Description and immunological findings. Clin Allergy. 1975;5:121–37.
Meadow SR, Sarsfield JK. Steroid-responsive and nephrotic syndrome and allergy: clinical studies. Arch Dis Child. 1981;56:509–16.
Meadow SR, Sarsfield JK, Scott DG, Rajah SM. Steroid-responsive nephrotic syndrome and allergy: immunological studies. Arch Dis Child. 1981;56:517–24.
Lin CY, Lee BH, Lin CC, Chen WP. A study of the relationship between childhood nephrotic syndrome and allergic diseases. Chest. 1990;97:1408–11.
Yap HK, Yip WC, Lee BW, Ho TF, Teo J, Aw SE, et al. The incidence of atopy in steroid-responsive nephrotic syndrome: clinical and immunological parameters. Ann Allergy. 1983;51:590–4.
Hopkin JM. Atopy, asthma, and the mycobacteria. Thorax. 2000;55:443–5.
Araya CE, Wasserfall CH, Brusko TM, Mu W, Segal MS, Johnson RJ, et al. A case of unfulfilled expectations. Cytokines in idiopathic minimal lesion nephrotic syndrome. Pediatr Nephrol. 2006;21:603–10.
Kaneko K, Tuchiya K, Fujinaga S, Kawamura R, Ohtomo Y, Shimizu T, et al. Th1/Th2 balance in childhood idiopathic nephrotic syndrome. Clin Nephrol. 2002;58:393–7.
Gasparoni A, Ciardelli L, Avanzini A, Castellazzi AM, Carini R, Rondini G, et al. Age-related changes in intracellular TH1/TH2 cytokine production, immunoproliferative T lymphocyte response and natural killer cell activity in newborns, children and adults. Biol Neonate. 2003;84:297–303.
Han SN, Adolfsson O, Lee CK, Prolla TA, Ordovas J, Meydani SN. Age and vitamin E-induced changes in gene expression profiles of T cells. J Immunol. 2006;177:6052–61.
Stachowski J, Barth C, Michalkiewicz J, Krynicki T, Jarmolinski T, Runowski D, et al. Th1/Th2 balance and CD45-positive T cell subsets in primary nephrotic syndrome. Pediatr Nephrol. 2000;14:779–85.
White RH. Clinical applications of selectivity of proteinuria. Contrib Nephrol. 1981;24:63–71.
Hogg RJ, Portman RJ, Milliner D, Lemley KV, Eddy A, Ingelfinger J. Evaluation and management of proteinuria and nephrotic syndrome in children: recommendations from a pediatric nephrology panel established at the National Kidney Foundation conference on proteinuria, albuminuria, risk, assessment, detection, and elimination (PARADE). Pediatrics. 2000;105:1242–9.
MacDonald NE, Wolfish N, McLaine P, Phipps P, Rossier E. Role of respiratory viruses in exacerbations of primary nephrotic syndrome. J Pediatr. 1986;108:378–82.
Nephrotic syndrome in children: prediction of histopathology from clinical and laboratory characteristics at time of diagnosis. A report of the International Study of Kidney Disease in Children. Kidney Int 1978;13:159–65.
Rebien W, Muller-Wiefel DE, Wahn U, Scharer K. IgE mediated hypersensitivity in children with idiopathic nephrotic syndrome. Int J Pediatr Nephrol. 1981;2:23–8.
Kanai T, Yamagata T, Momoi MY. Macrophage inflammatory protein-1beta and interleukin-8 associated with idiopathic steroid-sensitive nephrotic syndrome. Pediatr Int. 2009;51:443–7.
Salsano ME, Graziano L, Luongo I, Pilla P, Giordano M, Lama G. Atopy in childhood idiopathic nephrotic syndrome. Acta Paediatr. 2007;96:561–6.
Ohtomo Y, Fujinaga S, Hattori M. Suplatast tosilate dimethylsulfonium treatment for steroid-dependent nephrotic syndrome. Pediatr Int. 2005;47:230–1.
Bossios A, Sjostrand M, Dahlborn AK, Samitas K, Malmhall C, Gaga M, et al. IL-5 expression and release from human CD34 cells in vitro; ex vivo evidence from cases of asthma and Churg-Strauss syndrome. Allergy. 2010;65(7):831–9.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kanai, T., Shiraishi, H., Yamagata, T. et al. Th2 cells predominate in idiopathic steroid-sensitive nephrotic syndrome. Clin Exp Nephrol 14, 578–583 (2010). https://doi.org/10.1007/s10157-010-0330-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-010-0330-z