Abstract
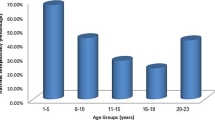
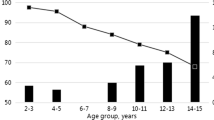
A nationwide hepatitis B virus (HBV) vaccination program for neonates was launched in Iran in 1993. Despite the success of this program, concern about its long-term success still remains, because breakthrough infections due to emergence of surface mutants have been reported in immunized children. We aimed to evaluate the seroprevalence of HBV and vaccine escape mutants among individuals born after the initiation of the nationwide vaccination program in Iran. This study included 1115 participants younger than 23 years old, with 223 in each age cohort. The presence of HBsAg, anti-HBs and anti-HBc was evaluated using an ELISA kit. HBV-DNA levels were measured in anti-HBc and/or HBsAg-positive subjects. PCR products were sequenced and mutations were identified. The overall HBsAg prevalence was 0.27 %. Anti-HBs and anti-HBc positive rates were 48 % and 0.18 %, respectively. Two individuals were positive for anti-HBc, one of whom was also positive for HBsAg, and the other was positive for anti-HBc only. HBV DNA was detected in three out of four anti-HBc-and /or HBsAg-positive subjects. An I195M mutation within the S gene was detected in two of the three HBV-DNA-positive cases. A very low prevalence of HBsAg and isolated anti-HBc were found in this study. The I195M mutation found in the surface gene could have been induced by immune pressure. Although the number of ‘‘vaccine escape’’ mutants found in this cohort was low, ongoing surveillance of breakthrough infections and escape mutants is still needed.

Similar content being viewed by others
References
Cha C, Dematteo RP (2005) Molecular mechanisms in hepatocellular carcinoma development. Best Pract Res Clin Gastroenterol 19:25–37
van der Sande MA, Waight PA, Mendy M, Zaman S, Kaye S, Sam O, Kahn A, Jeffries D, Akum AA, Hall AJ, Bah E, McConkey SJ, Hainaut P, Whittle HC (2007) 0 Long-term protection against HBV chronic carriage of Gambian adolescents vaccinated in infancy and immune response in HBV booster trial in adolescence. PLoS One 2(8):e753
Poovorawan Y, Chongsrisawat V, Tangkijvanich P (2001) Problems and prevention of viral hepatitis in Thailand. J Med Assoc Thai 84(Suppl 1):S18–S25
Chen DS (2009) Hepatitis B vaccination: the key towards elimination and eradication of hepatitis B. J Hepatol 50:805–816
Farzadegan H, Shamszad M, Noori-Arya K (1980) Epidemiology of viral hepatitis among Iranian population a viral marker study. Ann Acad Med Singapore 9:144–148
Merat S, Malekzadeh R, Rezvan H, Khatibian M (2000) Hepatitis B in Iran. Arch Iranian Med 3:192–201
Kabir A, Alavian SM, Ahanchi N, Malekzadeh R (2006) Combined passive and active immunoprophylaxis for preventing perinatal transmission of the hepatitis B virus in infants born to HBsAg positive mothers in comparison with vaccine alone. Hepatol Res. 36(4):265–271
Adibi P, Ghassemian R, Alavian SM, Ranjbar M, Mohammadalizadeh AH, Nematizadeh F, Mamani M, Rezazadeh M, Keramat F, Ardalan A, Esmaeili A, Zali MR (2004) Effectiveness of hepatitis B vaccination in children of chronic hepatitis B mothers. Saudi Med J. 25:1414–1418
Esteghamati A, Keshtkar AA, Nadjafi L, Gouya MM, Salaramoli M, Roshandel G, Yaghini F (2011) Hepatitis B vaccination coverage among Iranian children aged 15-26 months in 2006. East Mediterr Health J 17(2):93–100
Alavian SM, Fallahian F, Lankarani KB (2007) The changing epidemiology of viral hepatitis B in Iran. J Gastrointestin Liver Dis. 16(4):403–406
Alavian SM, Hajariazdeh B, Ahmadzad Asl M, Kabir A, Bagheri Lankarani KB (2008) Hepatitis B virus infection in Iran: a systematic review. Hepat Mon. 8(4):281–294
Alavian SM, Lankarani K (2012) Hepatitis B virus infection; a vanishing disease in Iranian children. J Compr Ped 3(1):1–2
McMahon BJ, Bruden DL, Petersen KM, Bulkow LR, Parkinson AJ, Nainan O, Khristova M, Zanis C, Peters H, Margolis HS (2005) Antibody levels and protection after hepatitis B vaccination: results of a 15-year follow up. Ann Intern Med 142:333–341
Chang MH (2010) Breakthrough HBV infection in vaccinated children in Taiwan: surveillance for HBV mutants. Antivir Ther 15:463–469
Ngui SL, Andrews NJ, Underhill GS, Heptonstall J, Teo CG (1998) Failed postnatal immunoprophylaxis for hepatitis B: characteristics of maternal hepatitis B virus as risk factors. Clin Infect Dis 27:100–106
del Canho R, Grosheide PM, Schalm SW, de Vries RR, Heijtink RA (1994) Failure of neonatal hepatitis B vaccination: the role of HBV-DNA levels in hepatitis B carrier mothers and HLA antigens in neonates. J Hepatol 20:483–486
Hsu HY, Chang MH, Ni YH, Lin HH, Wang SM, Chen DS (1997) Surface gene mutants of hepatitis B virus in infants who develop acute or chronic infections despite immunoprophylaxis. Hepatology 26:786–791
Hsu HY, Chang MH, Liaw SH, Ni YH, Chen HL (1999) Changes of hepatitis B surface antigen variants in carrier children before and after universal vaccination in Taiwan. Hepatology 30:1312–1317
Hsu HY, Chang MH, Ni YH (2004) Chen HL (2004) Survey of hepatitis B surface variant infection in children 15 years after a nationwide vaccination programme in Taiwan. Gut 53:1499–1503
Weber B (2005) Genetic variability of the S gene of hepatitis B virus: clinical and diagnostic impact. J Clin Virol. 32(2):102–112
Coleman PF (2006) Surveillance for hepatitis B surface antigen mutants. J Med Virol. 78(Suppl 1):S56–S58
Zuckerman JN, Zuckerman AJ (2003) Mutations of the surface protein of hepatitis B virus. Antiviral Res. 60(2):75–78
Said ZN (2011) An overview of occult hepatitis B virus infection. World J Gastroenterol. 17(15):1927–1938
Carman WF, Zanetti AR, Karayiannis P, Waters J, Manzillo G, Tanzi E, Zuckerman AJ, Thomas HC (1990) Vaccine induced escape mutant of hepatitis B virus. Lancet 336:325–329
Coleman PF (2006) Detecting hepatitis B surface antigen mutants. Emerg Infect Dis 6:198–203
Cooreman MP, Leroux-Roels G, Paulij WP (2001) Vaccine and hepatitis B immune globulin-induced escape mutations of hepatitis B virus surface antigen. J Biomed Sci 8:237–247
Liu CJ, Lo SC, Kao JH, Tseng PT, Lai MY, Ni YH, Yeh SH, Chen PJ, Chen DS (2006) Transmission of occult hepatitis B virus by transfusion to adult and pediatric recipients in Taiwan. J Hepatol 44:39–46
Su WJ, Ho MC, Ni YH, Wu JF, Jeng YM, Chen HL, Wu YM, Hu RH, Chang MH, Lee PH (2010) Clinical course of de novo hepatitis B infection after pediatric liver transplantation. Liver Transpl 16:215–221
WHO Publication (2010) Hepatitis B vaccines: WHO position paper—Recommendations. Vaccine 28(3):589–590
Sun Z, Ming L, Zhu X, Lu J (2002) Prevention and control of hepatitis B in China. J Med Virol 67:447–450
Samandari T, Fiore AE, Negus S, Williams JL, Kuhnert W, McMahon BJ, Bell BP (2007) Waning immunity to plasma-derived hepatitis B vaccine and the need for boosters 15 years after neonatal vaccination. Pediatrics 120:e373–e381
Lu CY, Ni YH, Chiang BL, Chen PJ, Chang MH, Chang LY, Su IJ, Kuo HS, Huang LM, Chen DS, Lee CY (2008) Humoral and cellular immune responses to a hepatitis B vaccine booster 15-18 years after neonatal immunization. J Infect Dis 197:1419–1426
Jan CF, Huang KC, Chien YC, Davies HD, Chiu TY, Huang LM, Chen CJ, Chen DS (2010) Determination of immune memory to hepatitis B vaccination through early booster response in college students. Hepatology 51:1547–1554
West DJ, Calandra GB (1996) Vaccine induced immunologic memory for hepatitis B surface antigen: implications for policy on booster vaccination. Vaccine 14(11):1019–1027
John TJ, Cooksley G (2005) Hepatitis B vaccine boosters: is there a clinical need in high endemicity populations? J Gastroenterol Hepatol 201:5–10
Lai MW, Lin TY, Tsao KC, Huang CG, Hsiao MJ, Liang KH, Yeh CT (2012) Increased seroprevalence of HBV DNA with mutations in the s gene among individuals greater than 18 years old after complete vaccination. Gastroenterology 143(2):400–407
Ni YH, Chang MH, Wu JF, Hsu HY, Chen HL, Chen DS (2012) Minimization of hepatitis B infection by a 25-year universal vaccination program. J Hepatol 57(4):730–735
Hsu HY, Chang MH, Ni YH, Jeng YM, Chiang CL, Chen HL, Wu JF, Chen PJ (2013) Long-term follow-up of children with postnatal immunoprophylaxis failure who were infected with hepatitis B virus surface antigen gene mutant. J Infect Dis 207:1047–1057
Zanetti AR, Tanzi E, Manzillo G, Sbreglia C, Caporaso N, Thomas H, Zuckerman AJ (1988) Hepatitis B variant in Europe. Lancet 2:1132–1133
Teo CG, Locarnini S (2010) Potential threat of drug-resistant and vaccine-escape HBV mutants to public health. Antivir Ther 15:445–449
Nainan OV, Khristova ML, Byun K, Xia G, Taylor PE, Stevens CE, Margolis HS (2002) Genetic variation of hepatitis B surface antigen coding region among infants with chronic hepatitis B virus infection. J Med Virol 68:319–327
Lazarevic I (2014) Clinical implications of hepatitis B virus mutations: recent advances. World J Gastroenterol 20(24):7653–7664
Fernández-Galindo DA, Sánchez-Ávila F, Bobadilla-Morales L, Gómez-Quiróz P, Bueno-Topete M, Armendáriz-Borunda J, Sánchez-Orozco LV (2015) New amino acid changes in drug resistance sites and HBsAg in hepatitis B virus genotype H. J Med Virol 87(6):985–992
Perazzo P, Eguibar N, González RH, Nusblat AD, Cuestas ML (2015) Hepatitis B virus (HBV) and S-escape mutants: From the beginning until now. J Hum Virol Retrovirol 2(3):00046
Hsu HY, Chang MH, Ni YH, Chiang CL, Chen HL, Wu JF, Chen PJ (2010) No increase in prevalence of hepatitis B surface antigen mutant in a population of children and adolescents who were fully covered by universal infant immunization. J Infect Dis 201:1192–1200
Torbenson M, Thomas DL (2002) Occult hepatitis B. Lancet Infect Dis 2:479–486
Huang C-H, Yuan Q, Chen PJ, Zhang YL, Chen CR, Zheng QB, Yeh SH, Yu H, Xue Y, Chen YX, Liu PG, Ge SX, Zhang J, Xia NS (2012) Influence of mutations in hepatitis B virus surface protein on viral antigenicity and phenotype in occult HBV strains from blood donors. J Hepatol. 57(4):720–729
Acknowledgements
The authors are grateful to Pasteur Institute of Iran for financial support of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Pasteur Institute of Iran (grant number: 767).
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Pasteur Institute of Iran ethics committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Aghasadeghi, M.R., Velayati, A.A., Mamishi, S. et al. Low prevalence of hepatitis B vaccine escape mutants among individuals born after the initiation of a nationwide vaccination program in Iran. Arch Virol 161, 3405–3411 (2016). https://doi.org/10.1007/s00705-016-3050-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-016-3050-1