Abstract
Purpose
To define the role of surgery for metastatic/recurrent lesions after resection of primary gastrointestinal stromal tumors (GISTs).
Methods
Based on data obtained from the Kinki GIST registry, patients with recurrence or metastasis were divided into a surgical treatment group (ST group), comprised those treated with surgery in addition to tyrosine kinase inhibitor (TKI) therapy; and a drug treatment group (DT group), comprised those treated with TKI therapy alone. We compared the baseline characteristics and survival outcomes of the groups.
Results
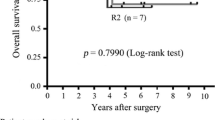
Metastasis or recurrence developed in 93 of the 737 patients with GISTs treated between 2003 and 2007, 50 (53.8 %) of whom were assigned to the ST group and 43 (46.2 %) to the DT group. In the ST group, the 5-year overall survival rate was significantly higher for patients who underwent R0/R1 resection than for those who underwent R2 resection (82.2 vs. 47.0 %, p = 0.018). Survival time after recurrence was correlated with the duration of total TKI therapy in both the ST and DT groups (r = 0.766 and r = 0.932, respectively, p < 0.001).
Conclusions
Continuous TKI therapy appears to be important primarily for the prognostic improvement of patients with recurrent/metastatic GISTs. R0/R1 resection may have benefits when combined with TKI therapy for patients with stable disease or disease responsive to TKI therapy, less than four metastatic lesions, and lesions <100 mm in total.


Similar content being viewed by others
References
Chung JC, Kim HC, Hur SM. Limited resection for duodenal gastrointestinal stromal tumors and their oncologic outcomes. Surg Today. 2016;46:110–6.
Demetri GD, von Mehren M, Blanke CD, Van den Abbeele AD, Eisenberg B, Roberts PJ, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002;347:472–80.
Demetri GD, van Oosterom AT, Garrett CR, Blackstein ME, Shah MH, Verweij J, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet. 2006;368:1329–38.
Demetri GD, Reichardt P, Kang YK, Blay JY, Rutkowski P, Gelderblom H, et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:295–302.
Rubió-Casadevall J, Martinez-Trufero J, Garcia-Albeniz X, Calabuig S, Lopez-Pousa A, Del Muro JG, et al. Role of surgery in patients with recurrent, metastatic, or unresectable locally advanced gastrointestinal stromal tumors sensitive to imatinib: a retrospective analysis of the Spanish Group for Research on Sarcoma (GEIS). Ann Surg Oncol. 2015;22:2948–57.
Gronchi A, Fiore M, Miselli F, Lagonigro MS, Coco P, Messina A, et al. Surgery of residual disease following molecular-targeted therapy with imatinib mesylate in advanced/metastatic GIST. Ann Surg. 2007;245:341–6.
Zaydfudim V, Okuno SH, Que FG, Nagorney DM, Donohue JH. Role of operative therapy in treatment of metastatic gastrointestinal stromal tumors. J Surg Res. 2012;177:248–54.
Hakimé A, Le Cesne A, Deschamps F, Farouil G, Boudabous S, Aupérin A, et al. A role for adjuvant RFA in managing hepatic metastases from gastrointestinal stromal tumors (GIST) after treatment with targeted systemic therapy using kinase inhibitors. Cardiovasc Intervent Radiol. 2014;37:132–9.
Vassos N, Agaimy A, Hohenberger W, Croner RS. Management of liver metastases of gastrointestinal stromal tumors (GIST). Ann Hepatol. 2015;14:531–9.
Du CY, Zhou Y, Song C, Wang YP, Jie ZG, He YL, et al. Is there a role of surgery in patients with recurrent or metastatic gastrointestinal stromal tumours responding to imatinib: a prospective randomised trial in China. Eur J Cancer. 2014;50:1772–8.
Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411–9.
DeMatteo RP, Maki RG, Singer S, Gonen M, Brennan MF, Antonescu CR. Results of tyrosine kinase inhibitor therapy followed by surgical resection for metastatic gastrointestinal stromal tumor. Ann Surg. 2007;245:347–52.
Bischof DA, Kim Y, Blazer DG 3rd, Behman R, Karanicolas PJ, Law CH, et al. Surgical management of advanced gastrointestinal stromal tumors: an international multi-institutional analysis of 158 patients. J Am Coll Surg. 2014;219:439–49.
Yeh CN, Chen TW, Tseng JH, Liu YY, Wang SY, Tsai CY, et al. Surgical management in metastatic gastrointestinal stromal tumor (GIST) patients after imatinib mesylate treatment. J Surg Oncol. 2010;102:599–603.
Park SJ, Ryu MH, Ryoo BY, Park YS, Sohn BS, Kim HJ, et al. The role of surgical resection following imatinib treatment in patients with recurrent or metastatic gastrointestinal stromal tumors: results of propensity score analyses. Ann Surg Oncol. 2014;21:4211–7.
DeMatteo RP, Shah A, Fong Y, Jarnagin WR, Blumgart LH, Brennan MF. Results of hepatic resection for sarcoma metastatic to liver. Ann Surg. 2001;234:540–7.
Bauer S, Rutkowski P, Hohenberger P, Miceli R, Fumagalli E, Siedlecki JA, et al. Long-term follow-up of patients with GIST undergoing metastasectomy in the era of imatinib—analysis of prognostic factors (EORTC-STBSG collaborative study). Eur J Surg Oncol. 2014;40:412–9.
Turley RS, Peng PD, Reddy SK, Barbas AS, Geller DA, Marsh JW, et al. Hepatic resection for metastatic gastrointestinal stromal tumors in the tyrosine kinase inhibitor era. Cancer. 2010;118:3571–8.
Hasegawa J, Kanda T, Hirota S, Fukuda M, Nishitani A, Takahashi T, et al. Surgical interventions for focal progression of advanced gastrointestinal stromal tumors during imatinib therapy. Int J Clin Oncol. 2007;12:212–7.
Nishida T, Hirota S, Yanagisawa A, Sugano Y, Minami M, Yamamura Y, et al. GIST guideline subcommittee. Clinical practice guidelines for gastrointestinal stromal tumor (GIST) in Japan: English version. Int J Clin Oncol. 2008;13:416–30.
Ng EH, Pollock RE, Romsdahl MM. Prognostic implications of patterns of failure for gastrointestinal leiomyosarcomas. Cancer. 1992;69:1334–41.
Acknowledgments
This study was performed by the Kinki GIST Study Group, which includes the following institutions: Osaka University Hospital, Osaka General Medical Center, Shizuoka General Hospital, Osaka National Hospital, Osaka Red Cross Hospital, Toyonaka Municipal Hospital, Tenri Hospital, Osaka City General Hospital, Osaka City University Hospital, Osaka Police Hospital, Sakai Municipal Hospital, Kanazawa Medical University Hospital, Kitano Hospital, Juntendo University Shizuoka Hospital, Osaka Rosai Hospital, Osaka Medical College Hospital, Matsushita Memorial Hospital, Kyoto City Hospital, Shiga Medical Center for Adults, Hyogo Cancer Center, Hoshigaoka Koseinenkin Hospital, Kinki University Hospital, Kansai Electric Power Hospital, National Hospital Organization Osaka Minami Medical Center, Tokyo Metropolitan Cancer and Infectious diseases Center Komagome Hospital, Yodogawa Christian Hospital, Osaka Medical Center for Cancer and Cardiovascular Diseases, Kansai Medical University Takii Hospital, Sumitomo Hospital, Suita Municipal Hospital, The Hospital of Hyogo College of Medicine, Osaka City Sumiyoshi Hospital, Kansai Medical University Hirakata Hospital, Nissei Hospital, Otemae Hospital, Bellland General Hospital, Minoh City Hospital, Osaka Ekisaikai Hospital, Higashiosaka City General Hospital, and Rinku General Medical Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Sato, S., Tsujinaka, T., Masuzawa, T. et al. Role of metastasectomy for recurrent/metastatic gastrointestinal stromal tumors based on an analysis of the Kinki GIST registry. Surg Today 47, 58–64 (2017). https://doi.org/10.1007/s00595-016-1351-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-016-1351-3