Abstract
Background
Although several non-randomized studies comparing robotic pancreaticoduodenectomy (RPD) and open pancreaticoduodenectomy (OPD) recently demonstrated that the two operative techniques could be equivalent in terms of safety outcomes and short-term oncologic efficacy, no definitive answer has arrived yet to the question as to whether robotic assistance can contribute to reducing the high rate of postoperative morbidity.
Methods
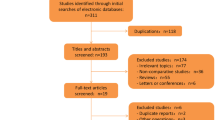
Systematic literature search was performed using MEDLINE, the Cochrane Central Register of Controlled Trials, and EMBASE databases. Prospective and retrospective studies comparing RPD and OPD as surgical treatment for periampullary benign and malignant lesions were included in the systematic review and meta-analysis with no limits of language or year of publication.
Results
18 non-randomized studies were included for quantitative synthesis with 13,639 patients allocated to RPD (n = 1593) or OPD (n = 12,046). RPD and OPD showed equivalent results in terms of mortality (3.3% vs 2.8%; P = 0.84), morbidity (64.4% vs 68.1%; P = 0.12), pancreatic fistula (17.9% vs 15.9%; P = 0.81), delayed gastric emptying (16.8% vs 16.1%; P = 0.98), hemorrhage (11% vs 14.6%; P = 0.43), and bile leak (5.1% vs 3.5%; P = 0.35). Estimated intra-operative blood loss was significantly lower in the RPD group (352.1 ± 174.1 vs 588.4 ± 219.4; P = 0.0003), whereas operative time was significantly longer for RPD compared to OPD (461.1 ± 84 vs 384.2 ± 73.8; P = 0.0004). RPD and OPD showed equivalent results in terms of retrieved lymph nodes (19.1 ± 9.9 vs 17.3 ± 9.9; P = 0.22) and positive margin status (13.3% vs 16.1%; P = 0.32).
Conclusions
RPD is safe and feasible as surgical treatment for malignant or benign disease of the pancreatic head and the periampullary region. Equivalency in terms of surgical radicality including R0 curative resection and number of harvested lymph nodes between the two groups confirmed the reliability of RPD from an oncologic point of view.






Similar content being viewed by others
References
Acharya A, Markar SR, Sodergren MH et al (2017) Meta-analysis of adjuvant therapy following curative surgery for periampullary adenocarcinoma. Br J Surg 104:814–822
Gagner M, Pomp A (1994) Laparoscopic pylorus-preserving pancreatoduodenectomy. Surg Endosc 8:408–410
Nickel F, Haney CM, Kowalewski KF et al (2019) Laparoscopic versus open pancreaticoduodenectomy: a systematic review and meta-analysis of randomized controlled trials. Ann Surg. https://doi.org/10.1097/SLA.0000000000003309
Boggi U, Amorese G, Vistoli F et al (2015) Laparoscopic pancreaticoduodenectomy: a systematic literature review. Surg Endosc 29:9–23
Boggi U, Palladino S, Massimetti G et al (2015) Laparoscopic robot-assisted versus open total pancreatectomy: a case-matched study. Surg Endosc 29:1425–1432
Peng L, Lin S, Li Y et al (2017) Systematic review and meta-analysis of robotic versus open pancreaticoduodenectomy. Surg Endosc 31:3085–3097
Ricci C, Casadei R, Taffurelli G et al (2018) Minimally invasive pancreaticoduodenectomy: what is the best "Choice"? A systematic review and network meta-analysis of non-randomized comparative studies. World J Surg 42:788–805
Shin SH, Kim YJ, Song KB et al (2017) Totally laparoscopic or robot-assisted pancreaticoduodenectomy versus open surgery for periampullary neoplasms: separate systematic reviews and meta-analyses. Surg Endosc 31:3459–3474
Zhang J, Wu WM, You L et al (2013) Robotic versus open pancreatectomy: a systematic review and meta-analysis. Ann Surg Oncol 20:1774–1780
Klompmaker S, van Hilst J, Wellner UF et al (2018) Outcomes after minimally-invasive versus open pancreatoduodenectomy: a pan-European propensity score matched study. Ann Surg. https://doi.org/10.1097/SLA.0000000000002850
Zimmerman AM, Roye DG, Charpentier KP (2018) A comparison of outcomes between open, laparoscopic and robotic pancreaticoduodenectomy. HPB (Oxford) 20:364–369
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700
Stroup DF, Berlin JA, Morton SC et al (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA 283:2008–2012
Higgins JPT, Green S (eds). Cochrane handbook for systematic reviews of interventions version 5.3.5. The cochrane collaboration, 2014. www.handbook.cochrane.org. Accessed April 2018.
Sterne JA, Herna’n MA, Reeves BC et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919
Balshem H, Helfand M, Schünemann HJ et al (2011) GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol 64:401–406
Bassi C, Dervenis C, Butturini G et al (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 138:8–13
Bassi C, Marchegiani G, Dervenis C et al (2017) The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery 161:584–591
Koch M, Garden OJ, Padbury R et al (2011) Bile leakage after hepatobiliary and pancreatic surgery: a definition and grading of severity by the International Study Group of Liver Surgery. Surgery 149:680–688
Wente MN, Bassi C, Dervenis C et al (2007) Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 142:761–768
Wente MN, Veit JA, Bassi C et al (2007) Postpancreatectomy hemorrhage (PPH): an International Study Group of Pancreatic Surgery (ISGPS) definition. Surgery 142:20–25
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5:13
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Boggi U, Napoli N, Costa F et al (2016) Robotic-assisted pancreatic resections. World J Surg 40:2497–2506
Walsh M, Chalikonda S, Saavedra JRA (2011) Laparoscopic robotic assisted Whipple: early results of a novel technique and comparison with the standard open procedure. Surg Endosc 25:S221
Kim H, Kim JR, Han Y et al (2017) Early experience of laparoscopic and robotic hybrid pancreaticoduodenectomy. Int J Med Robot. https://doi.org/10.1002/rcs.1814
Torphy RJ, Friedman C, Halpern A et al (2018) Comparing short-term and oncologic outcomes of minimally invasive versus open pancreaticoduodenectomy across low and high volume centers. Ann Surg. https://doi.org/10.1097/SLA.0000000000002810
Croome KP, Farnell MB, Que FG et al (2014) Total laparoscopic pancreaticoduodenectomy for pancreatic ductal adenocarcinoma: oncologic advantages over open approaches? Ann Surg 260:633–638
Adam MA, Choudhury K, Dinan MA et al (2015) Minimally invasive versus open pancreaticoduodenectomy for cancer: practice patterns and short-term outcomes among 7061 patients. Ann Surg 262:372–377
Hammill C, Cassera M, Swanstrom L et al (2010) Robotic assistance may provide the technical capability to perform a safe, minimally invasive pancreaticoduodenectomy. HPB (Oxford) 12:198
McMillan MT, Zureikat AH, Hogg ME et al (2017) A propensity score-matched analysis of robotic vs open pancreatoduodenectomy on incidence of pancreatic fistula. JAMA Surg 152:327–335
Zureikat AH, Postlewait LM, Liu Y et al (2016) A multi-institutional comparison of perioperative outcomes of robotic and open pancreaticoduodenectomy. Ann Surg 264:640–649
Kauffmann EF, Napoli N, Menonna F et al (2019) A propensity score-matched analysis of robotic versus open pancreatoduodenectomy for pancreatic cancer based on margin status. Surg Endosc 33:234–242
Girgis MD, Zenati MS, Steve J et al (2017) Robotic approach mitigates perioperative morbidity in obese patients following pancreaticoduodenectomy. HPB (Oxford) 19:93–98
Varley PR, Zenati MS, Klobuka A et al (2018) Does robotic pancreaticoduodenectomy improve outcomes in patients with high risk morphometric features compared to the open approach. HPB (Oxford). https://doi.org/10.1016/j.hpb.2018.10.016
Napoli N, Kauffmann EF, Menonna F et al (2018) Robotic versus open pancreatoduodenectomy: a propensity score-matched analysis based on factors predictive of postoperative pancreatic fistula. Surg Endosc 32:1234–1247
Baker EH, Ross SW, Seshadri R et al (2016) Robotic pancreaticoduodenectomy: comparison of complications and cost to the open approach. Int J Med Robot 12:554–560
Bao PQ, Mazirka PO, Watkins KT (2014) Retrospective comparison of robot-assisted minimally invasive versus open pancreaticoduodenectomy for periampullary neoplasms. J Gastrointest Surg 18:682–689
Buchs NC, Addeo P, Bianco FM et al (2011) Robotic versus open pancreaticoduodenectomy: a comparative study at a single institution. World J Surg 35:2739–2746
Zhou NX, Chen JZ, Liu Q et al (2011) Outcomes of pancreatoduodenectomy with robotic surgery versus open surgery. Int J Med Robot 7:131–137
Kim HS, Han Y, Kang JS et al (2018) Comparison of surgical outcomes between open and robot-assisted minimally invasive pancreaticoduodenectomy. J Hepatobiliary Pancreat Sci 25:142–149
Wang SE, Shyr BU, Chen SC et al (2018) Comparison between robotic and open pancreaticoduodenectomy with modified Blumgart pancreaticojejunostomy: a propensity score-matched study. Surgery 164:1162–1167
Chen S, Chen JZ, Zhan Q et al (2015) Robot-assisted laparoscopic versus open pancreaticoduodenectomy: a prospective, matched, mid-term follow-up study. Surg Endosc 29:3698–3711
Chalikonda S, Aguilar-Saavedra JR, Walsh RM (2012) Laparoscopic robotic-assisted pancreaticoduodenectomy: a case-matched comparison with open resection. Surg Endosc 26:2397–2402
Lai EC, Yang GP, Tang CN (2012) Robot-assisted laparoscopic pancreaticoduodenectomy versus open pancreaticoduodenectomy–a comparative study. Int J Surg 10:475–479
Addeo P, Delpero JR, Paye F et al (2014) Pancreatic fistula after a pancreaticoduodenectomy for ductal adenocarcinoma and its association with morbidity: a multicentre study of the French Surgical Association. HPB (Oxford) 16:46–55
Kimura W, Miyata H, Gotoh M et al (2014) A pancreaticoduodenectomy risk model derived from 8575 cases from a national single-race population (Japanese) using a web-based data entry system: the 30-day and in-hospital mortality rates for pancreaticoduodenectomy. Ann Surg 259:773–780
Kornaropoulos M, Moris D, Beal EW et al (2017) Total robotic pancreaticoduodenectomy: a systematic review of the literature. Surg Endosc 31:4382–4392
Lei P, Wei B, Guo W et al (2014) Minimally invasive surgical approach compared with open pancreaticoduodenectomy: a systematic review and meta-analysis on the feasibility and safety. Surg Laparosc Endosc Percutan Tech 24:296–305
Marino MV, Podda M, Gomez Ruiz M et al (2019) Robotic-assisted versus open pancreaticoduodenectomy: the results of a case-matched comparison. J Robot Surg. https://doi.org/10.1007/s11701-019-01018-w
Kazanjian KK, Hines OJ, Duffy JP et al (2008) Improved survival following pancreaticoduodenectomy to treat adenocarcinoma of the pancreas: the influence of operative blood loss. Arch Surg 143:1166–1171
Podda M, Thompson J, Kulli CTG et al (2017) Vascular resection in pancreaticoduodenectomy for periampullary cancers. A 10 year retrospective cohort study. Int J Surg 39:37–44
Kulemann B, Hoeppner J, Wittel U et al (2015) Perioperative and long-term outcome after standard pancreaticoduodenectomy, additional portal vein and multivisceral resection for pancreatic head cancer. J Gastrointest Surg 19:438–444
Ravikumar R, Sabin C, Abu Hilal M et al (2014) Portal vein resection in borderline resectable pancreatic cancer: a United Kingdom multicenter study. J Am Coll Surg 218:401–411
Shyr BU, Chen SC, Shyr YM et al (2019) Surgical, survival, and oncological outcomes after vascular resection in robotic and open pancreaticoduodenectomy. Surg Endosc. https://doi.org/10.1007/s00464-019-06779-x
Author information
Authors and Affiliations
Contributions
MP: Study conception and design, literature search, acquisition, interpretation and analysis of data; drafting and critically revising the article for important intellectual content; and final approval of the version to be published. CG: Study conception and design, literature search, acquisition, interpretation and analysis of data (statistical expertise); drafting and critically revising the article for important intellectual content; and final approval of the version to be published. SDS: Study conception and design, interpretation and analysis of data; drafting and critically revising the article for important intellectual content; editing and revising the English for the final version to be published; and final approval of the version to be published. MVM: Interpretation and analysis of data; drafting and critically revising the article for important intellectual content; and final approval of the version to be published. RJD: Interpretation and analysis of data; drafting and critically revising the article for important intellectual content; editing and revising the English for the final version to be published; and final approval of the version to be published. GP: Interpretation and analysis of data; drafting and critically revising the article for important intellectual content; and final approval of the version to be published. AP: Study conception and design, literature search, acquisition, interpretation and analysis of data; drafting and critically revising the article for important intellectual content; and final approval of the version to be published.
Corresponding author
Ethics declarations
Disclosures
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Mauro Podda, Chiara Gerardi, Salomone Di Saverio, Marco Vito Marino, R Justin Davies, Gianluca Pellino, and Adolfo Pisanu have no conflict of interest of finantial ties to disclose.
Ethical approval
No ethical approval was required for this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
PROSPERO ID: CRD42019133843.
Electronic supplementary material
Below is the link to the electronic supplementary material.
464_2020_7460_MOESM2_ESM.doc
Supplemental Digital Content Table 2. General Characteristics of the Studies Included for the Systematic Review and Meta-analysis (DOC 46 kb)
464_2020_7460_MOESM3_ESM.doc
Supplemental Digital Content Table 3. Risk of Bias in the Published Studies (By the Risk Of Bias In Non-Randomised Studies – of Interventions - ROBINS-I Tool) (DOC 31 kb)
464_2020_7460_MOESM4_ESM.doc
Supplemental Digital Content Table 4. Summary of Outcomes_1: Clinical Outcomes (Mortality, Morbidity, Overall POPF, Grade B POPF, Grade C POPF, Overall DGE, Overall PPH, Bile Leak, SSI, Clavien-Dindo Grade I-II Complications, Clavien-Dindo Grade III-V Complications) (DOC 73 kb)
464_2020_7460_MOESM5_ESM.doc
Supplemental Digital Content Table 5. Summary of Outcomes_2: Operative Outcomes (Conversion, Estimated Blood Loss, Operative Time); Post-operative Outcomes (Length of Hospital Stay, Reoperation); Costs (Operative Costs, Total Costs) (DOC 48 kb)
464_2020_7460_MOESM6_ESM.doc
Supplemental Digital Content Table 6. Summary of Pathology Outcomes (Tumor Size, Retrieved Lymph Nodes, Positive Margin, Pancreatic Adenocarcinoma, Distal Common Bile Duct Adenocarcinoma, Duodenal Adenocarcinoma, Ampullary Adenocarcinoma, Malignant Neuroendocrine Neoplasia, IPMN, Others Benign, Others Malignant) (DOC 73 kb)
464_2020_7460_MOESM7_ESM.doc
Supplemental Digital Content Table 7. Summary results of the meta-analyses comparing Totally RPD and Open PD (Subgroup Analysis) (DOC 42 kb)
464_2020_7460_MOESM8_ESM.tiff
Supplemental Digital Content. Fig. 1. Funnel plots of Mortality [A]. Morbidity [B]. Post-Operative Pancreatic Fistula [C]. Delayed Gastric Emptying [D]. Post-Pancreatectomy Hemorrhage [E]. Surgical Site Infection [F]. Operative Time [G]. Estimated Blood Loss [H]. and Length of Hospital Stay [I] (TIFF 122 kb)
464_2020_7460_MOESM9_ESM.tiff
Supplemental Digital Content. Fig. 2. Meta-analyses of Patients Characteristics: Charlson Comorbidity Index [A]. Age [B]. BMI [C]. Neoadjuvant Chemotherapy [D]. Vascular Resection [E]. Male Sex [F]. Subgroup Analyses: Totally Robotic and Hybrid RPD (TIFF 460 kb)
464_2020_7460_MOESM10_ESM.tiff
Supplemental Digital Content. Fig. 3. Meta-analyses of Pathology Outcomes: Tumor Size [A]. Pancreatic Adenocarcinoma [B]. Distal Common Bile Duct Adenocarcinoma [C]. Duodenal Adenocarcinoma [D]. Ampullary Adenocarcinoma [E]. Malignant Neuroendocrine Neoplasia [F]. Others Benign [G]. IPMN [H]. Subgroup Analyses: Totally Robotic and Hybrid RPD (TIFF 412 kb)
464_2020_7460_MOESM11_ESM.tiff
Supplemental Digital Content. Fig. 4. Sensitivity analyses excluding large registry studies: Morbidity [A]. Overall POPF [B]. Overall PPH [C]. SSI [D]. Operative Time [E]. Estimated Blood Loss [F]. Subgroup Analyses: Totally Robotic and Hybrid RPD (TIFF 417 kb)
Rights and permissions
About this article
Cite this article
Podda, M., Gerardi, C., Di Saverio, S. et al. Robotic-assisted versus open pancreaticoduodenectomy for patients with benign and malignant periampullary disease: a systematic review and meta-analysis of short-term outcomes. Surg Endosc 34, 2390–2409 (2020). https://doi.org/10.1007/s00464-020-07460-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-07460-4