Abstract
Background
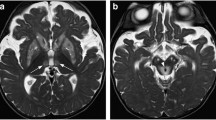
Neuronal intranuclear inclusion disease (NIID) is a neurodegenerative disease characterized by eosinophilic hyaline intranuclear inclusions in multiple organs. On conventional MRI, high signals on diffused weight image (DWI) along the corticomedullary junction have demonstrated great diagnostic values for adult-onset NIID. However, changes of contrast MRI in the acute period of the encephalopathy-like episode have rarely been investigated.
Methods
Patients with enhanced lesions were retrieved in our database including 35 patients with adult-onset NIID between October 2017 and December 2019. Conventional and contrast MRI were conducted in all patients. Standard procedures of skin biopsy were performed in all patients. Repeat-primed PCR and amplicon length PCR were used to screen the GGC expansion in the 5′UTR of the NOTCH2NLC gene.
Results
Four of 35 patients (11.4%) were identified to have a cortical enhancement in this study. The enhanced lesions were selectively spread along the surface of posterior cortex and were clinically associated with encephalopathy-like episodes. These patients had a younger age of onset, shorter duration of disease, and a higher incidence of a headache than those without enhancement. Typical p62-postive intranuclear inclusions were observed in all patients, while patient 1 simultaneously had many nuclei full of abnormal substance immunopositive to p62, as well as short-curly filament materials on electron microscopy. All patients were identified to have GGC repeat expansion in the NOTCH2NLC gene.
Conclusion
Post-contrast MRI should be routinely performed in the adult-onset NIID patients. Some patients with adult-onset NIID showed cortical enhancement and edema along the surface of posterior cortex, indicating that dehydrate and anti-inflammatory drugs might be potential therapies for these patients.







Similar content being viewed by others
Data availability
All relevant data are described in the paper. Deidentified data can be requested. Data can be requested by all interested researchers, who can be contacted via the corresponding author.
References
Sone J, Mori K, Inagaki T, Katsumata R, Takagi S, Yokoi S, Araki K, Kato T, Nakamura T, Koike H, Takashima H, Hashiguchi A, Kohno Y, Kurashige T, Kuriyama M, Takiyama Y, Tsuchiya M, Kitagawa N, Kawamoto M, Yoshimura H, Suto Y, Nakayasu H, Uehara N, Sugiyama H, Takahashi M, Kokubun N, Konno T, Katsuno M, Tanaka F, Iwasaki Y, Yoshida M, Sobue G (2016) Clinicopathological features of adult-onset neuronal intranuclear inclusion disease. Brain 139(Pt 12):3170–3186. https://doi.org/10.1093/brain/aww249
Wang Y, Wang B, Wang L, Yao S, Zhao J, Zhong S, Cong L, Liu L, Zhang J, Zhang J, Hong D (2020) Diagnostic indicators for adult-onset neuronal intranuclear inclusion disease. Clin Neuropathol 39(1):7–18. https://doi.org/10.5414/NP301203
Deng J, Gu M, Miao Y, Yao S, Zhu M, Fang P, Yu X, Li P, Su Y, Huang J, Zhang J, Yu J, Li F, Bai J, Sun W, Huang Y, Yuan Y, Hong D, Wang Z (2019) Long-read sequencing identified repeat expansions in the 5'UTR of the NOTCH2NLC gene from Chinese patients with neuronal intranuclear inclusion disease. J Med Genet 56(11):758–764. https://doi.org/10.1136/jmedgenet-2019-106268
Tian Y, Wang JL, Huang W, Zeng S, Jiao B, Liu Z, Chen Z, Li Y, Wang Y, Min HX, Wang XJ, You Y, Zhang RX, Chen XY, Yi F, Zhou YF, Long HY, Zhou CJ, Hou X, Wang JP, Xie B, Liang F, Yang ZY, Sun QY, Allen EG, Shafik AM, Kong HE, Guo JF, Yan XX, Hu ZM, Xia K, Jiang H, Xu HW, Duan RH, Jin P, Tang BS, Shen L (2019) Expansion of human-specific GGC repeat in neuronal intranuclear inclusion disease-related disorders. Am J Hum Genet 105(1):166–176. https://doi.org/10.1016/j.ajhg.2019.05.013
Ishiura H, Shibata S, Yoshimura J, Suzuki Y, Qu W, Doi K, Almansour MA, Kikuchi JK, Taira M, Mitsui J, Takahashi Y, Ichikawa Y, Mano T, Iwata A, Harigaya Y, Matsukawa MK, Matsukawa T, Tanaka M, Shirota Y, Ohtomo R, Kowa H, Date H, Mitsue A, Hatsuta H, Morimoto S, Murayama S, Shiio Y, Saito Y, Mitsutake A, Kawai M, Sasaki T, Sugiyama Y, Hamada M, Ohtomo G, Terao Y, Nakazato Y, Takeda A, Sakiyama Y, Umeda-Kameyama Y, Shinmi J, Ogata K, Kohno Y, Lim SY, Tan AH, Shimizu J, Goto J, Nishino I, Toda T, Morishita S, Tsuji S (2019) Noncoding CGG repeat expansions in neuronal intranuclear inclusion disease, oculopharyngodistal myopathy and an overlapping disease. Nat Genet 51(8):1222–1232. https://doi.org/10.1038/s41588-019-0458-z
Sone J, Mitsuhashi S, Fujita A, Mizuguchi T, Hamanaka K, Mori K, Koike H, Hashiguchi A, Takashima H, Sugiyama H, Kohno Y, Takiyama Y, Maeda K, Doi H, Koyano S, Takeuchi H, Kawamoto M, Kohara N, Ando T, Ieda T, Kita Y, Kokubun N, Tsuboi Y, Katoh K, Kino Y, Katsuno M, Iwasaki Y, Yoshida M, Tanaka F, Suzuki IK, Frith MC, Matsumoto N, Sobue G (2019) Long-read sequencing identifies GGC repeat expansions in NOTCH2NLC associated with neuronal intranuclear inclusion disease. Nat Genet 51(8):1215–1221. https://doi.org/10.1038/s41588-019-0459-y
Okubo M, Doi H, Fukai R, Fujita A, Mitsuhashi S, Hashiguchi S, Kishida H, Ueda N, Morihara K, Ogasawara A, Kawamoto Y, Takahashi T, Takahashi K, Nakamura H, Kunii M, Tada M, Katsumoto A, Fukuda H, Mizuguchi T, Miyatake S, Miyake N, Suzuki J, Ito Y, Sone J, Sobue G, Takeuchi H, Matsumoto N, Tanaka F (2019) GGC repeat expansion of NOTCH2NLC in adult patients with leukoencephalopathy. Ann Neurol 86(6):962–968. https://doi.org/10.1002/ana.25586
Sun QY, Xu Q, Tian Y, Hu ZM, Qin LX, Yang JX, Huang W, Xue J, Li JC, Zeng S, Wang Y, Min HX, Chen XY, Wang JP, Xie B, Liang F, Zhang HN, Wang CY, Lei LF, Yan XX, Xu HW, Duan RH, Xia K, Liu JY, Jiang H, Shen L, Guo JF, Tang BS (2020) Expansion of GGC repeat in the human-specific NOTCH2NLC gene is associated with essential tremor. Brain 143(1):222–233. https://doi.org/10.1093/brain/awz372
Fang P, Yu Y, Yao S, Chen S, Zhu M, Chen Y, Zou K, Wang L, Wang H, Xin L, Hong T, Hong D (2020) Repeat expansion scanning of the NOTCH2NLC gene in patients with multiple system atrophy. Ann Clin Transl Neurol 7(4):517–526. https://doi.org/10.1002/acn3.51021(Epub ahead of print)
Chen L, Wu L, Li S, Huang Q, Xiong J, Hong D, Zeng X (2018) A long time radiological follow-up of neuronal intranuclear inclusion disease: two case reports. Medicine 97(49):e13544. https://doi.org/10.1097/MD.0000000000013544
Langford LA (1994) Demyelinative process associated with atypical intranuclear glial inclusions. Ultrastruct Pathol 18(1–2):15–18. https://doi.org/10.3109/01913129409016268
Wang R, Nie X, Xu S, Zhang M, Dong Z, Yu S (2020) Interrelated pathogenesis? Neuronal intranuclear inclusion disease combining with hemiplegic migraine. Headache 60(2):382–395. https://doi.org/10.1111/head.13687
Han X, Han M, Liu N, Xu J, Zhang Y, Zhang Y, Hong D, Zhang W (2019) Adult-onset neuronal intranuclear inclusion disease presenting with typical MRI changes. Brain Behav 9(12):e01477. https://doi.org/10.1002/brb3.1477
Fujita K, Osaki Y, Miyamoto R, Shimatani Y, Abe T, Sumikura H, Murayama S, Izumi Y, Kaji R (2017) Neurologic attack and dynamic perfusion abnormality in neuronal intranuclear inclusion disease. Neurol Clin Pract 7(6):e39–e42. https://doi.org/10.1212/CPJ.0000000000000389
Abe K, Fujita M (2017) Over 10 years MRI observation of a patient with neuronal intranuclear inclusion disease. BMJ Case Rep pii:bcr2016218790. https://doi.org/10.1136/bcr-2016-218790
Kitagawa N, Sone J, Sobue G, Kuroda M, Sakurai M (2014) Neuronal intranuclear inclusion disease presenting with resting tremor. Case Rep Neurol 6(2):176–180. https://doi.org/10.1159/000363687
Nakamura M, Ueki S, Kubo M, Yagi H, Sasaki R, Okada Y, Akiguchi I, Kusaka H, Kondo T (2018) Two cases of sporadic adult-onset neuronal intranuclear inclusion disease preceded by urinary disturbance for many years. J Neurol Sci 392:89–93. https://doi.org/10.1016/j.jns.2018.07.012
Araki K, Sone J, Fujioka Y, Masuda M, Ohdake R, Tanaka Y, Nakamura T, Watanabe H, Sobue G (2016) Memory loss and frontal cognitive dysfunction in a patient with adult-onset neuronal intranuclear inclusion disease. Intern Med 55(16):2281–2284. https://doi.org/10.2169/internalmedicine.55.5544
Liu Y, Lu J, Li K, Zhao H, Feng Y, Zhang Z, Hu L, Li G, Shao Y, Wang Y (2019) A multimodal imaging features of the brain in adult-onset neuronal intranuclear inclusion disease. Neurol Sci 40(4):905. https://doi.org/10.1007/s10072-019-03787-6
Kawarabayashi T, Nakamura T, Seino Y, Hirohata M, Mori F, Wakabayashi K, Ono S, Harigaya Y, Shoji M (2018) Disappearance of MRI imaging signals in a patient with neuronal intranuclear inclusion disease. J Neurol Sci 388:1–3. https://doi.org/10.1016/j.jns.2018.02.038
Black DF, Kung S, Sola CL, Bostwick MJ, Swanson J (2004) Familial hemiplegic migraine, neuropsychiatric symptoms, and Erdheim-Chester disease. Headache 44:911–915. https://doi.org/10.1111/j.1526-4610.2004.04174.x
Bhatia H, Babtain F (2011) Sporadic hemiplegic migraine with seizures and transient MRI abnormalities. Case Rep Neurol Med 2011:258372. https://doi.org/10.1155/2011/258372
Mateo I, Foncea N, Vicente I, Beldarrain M, García-Moncó J (2004) Migraine associated seizures with recurrent and reversible magnetic resonance imaging abnormalities. Headache 44:265–270. https://doi.org/10.1111/j.1526-4610.2004.04059.x
Zhao D, Hong D, Zhang W, Yao S, Qi X, Lv H, Zheng R, Feng L, Huang Y, Yuan Y, Wang Z (2011) Mutations in mitochondrially encoded complex I enzyme as the second common cause in a cohort of Chinese patients with mitochondrial myopathy, encephalopathy, lactic acidosis and stroke-like episodes. J Hum Genet 56(11):759–764. https://doi.org/10.1038/jhg.2011.96
Sone J, Hishikawa N, Koike H, Hattori N, Hirayama M, Nagamatsu M, Yamamoto M, Tanaka F, Yoshida M, Hashizume Y, Imamura H, Yamada E, Sobue G (2005) Neuronal intranuclear hyaline inclusion disease showing motor-sensory and autonomic neuropathy. Neurology 65:1538–1543. https://doi.org/10.1212/01.wnl.0000184490.22527.90
Yokoi S, Yasui K, Hasegawa Y, Niwa K, Noguchi Y, Tsuzuki T, Mimuro M, Sone J, Watanabe H, Katsuno M, Yoshida M, Sobue G (2016) Pathological background of subcortical hyperintensities on diffusion-weighted images in a case of neuronal intranuclear inclusion disease. Clin Neuropathol 35:375–380. https://doi.org/10.5414/NP300961
Acknowledgements
The authors would appreciate the patients and their families for their enthusiasm and participation in this study.
Funding
The work was supported by the National Natural Science Foundation of China (No. 81460199), and Double thousand talents program of Jiangxi province.
Author information
Authors and Affiliations
Contributions
LH and WB draft manuscript and analysis of data. WL, WH, and LX and contributed to the acquisition and analysis of data. ZM, DJ, and WZ and performed the genetic analysis and pathological study. CY and YY contributed to the critical revision of the manuscript. FP and HD contributed to the study design and drafting the manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors have declared no conflicts of interest.
Ethical approval
The research was approved by ethics committee of the first affiliated hospital of Nanchang University.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liang, H., Wang, B., Li, Q. et al. Clinical and pathological features in adult-onset NIID patients with cortical enhancement. J Neurol 267, 3187–3198 (2020). https://doi.org/10.1007/s00415-020-09945-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09945-7