Abstract
Purpose
To assess contrast-enhanced ultrasound (CEUS) targeted biopsy (TB) for clinically significant prostate cancer (PCa) detection compared with systematic biopsy (SB).
Methods
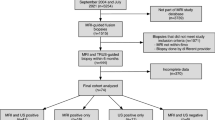
A total of 1024 consecutive patients scheduled for prostate biopsy were enrolled in this prospective study. CEUS was performed by an experienced radiologist blinded to all clinical data. Suspicious lesions on postcontrast images were sampled in addition to standard 12-core SB. The clinically significant PCa detection rate by CEUS-TB was evaluated in comparison with SB in the total cohort and in different subgroups.
Results
In 378 of 1024 patients (36.9%), the diagnosis of PCa was histologically confirmed. PCa was detected by CEUS-TB in 306 patients (29.9%, 306/1024) and SB in 317 patients (31.0%, 317/1024, P = 0.340). Among 378 PCa patients, 326 (86.2%, 326/378) were diagnosed with significant PCa using Epstein criteria. The significant PCa detection rate of CEUS-TB was 28.7% (294/1024), which was higher than that of SB (25.3%, 259/1024, P = 0.000). CEUS-TB resulted in 67 additional cases of clinically significant PCa, including 51 patients missed by SB and 16 patients under-graded by SB. Conversely, SB detected 32 additional significant PCa missed by TB. In the subgroup analysis, CEUS-TB yielded a higher significant cancer detection rate than SB in patients with a PSA level ≤ 10.0 ng/ml or prostate volume from 30 to 60 ml.
Conclusion
The clinically significant PCa detection rate could be improved by the extra sampling of abnormalities on postcontrast images, especially in patients with a PSA level ≤ 10.0 ng/ml or prostate volume from 30 to 60 ml.


Similar content being viewed by others
References
Mai Z, Xiao Y, Yan W et al (2018) Comparison of lesions detected and undetected by template-guided transperineal saturation prostate biopsy. BJU Int 121:415–420
Ploussard G, Salomon L, Xylinas E et al (2010) Pathological findings and prostate specific antigen outcomes after radical prostatectomy in men eligible for active surveillance—does the risk of misclassification vary according to biopsy criteria? J Urol 183:539–544
Prorok PC, Wright P, Riley TR et al (2018) Overall and multiphasic findings of the prostate, lung, colorectal and ovarian (PLCO) randomized cancer screening trial. Rev Recent Clin Trials. https://doi.org/10.2174/1574887113666180409153059
Loeb S, Bjurlin MA, Nicholson J et al (2014) Overdiagnosis and overtreatment of prostate cancer. Eur Urol 65:1046–1055
Miyata Y, Sakai H (2015) Reconsideration of the clinical and histopathological significance of angiogenesis in prostate cancer: usefulness and limitations of microvessel density measurement. Int J Urol 22:806–815
Erbersdobler A, Isbarn H, Dix K et al (2010) Prognostic value of microvessel density in prostate cancer: a tissue microarray study. World J Urol 28:687–692
Eisenberg ML, Cowan JE, Carroll PR et al (2010) The adjunctive use of power Doppler imaging in the preoperative assessment of prostate cancer. BJU Int 105:1237–1241
Halpern EJ (2006) Contrast-enhanced ultrasound imaging of prostate cancer. Rev Urol 8(Suppl 1):S29–S37
Postema AW, Frinking PJ, Smeenge M et al (2016) Dynamic contrast-enhanced ultrasound parametric imaging for the detection of prostate cancer. BJU Int 117:598–603
Halpern EJ, Ramey JR, Strup SE et al (2016) Detection of prostate carcinoma with contrast-enhanced sonography using intermittent harmonic imaging. Cancer 104:2373–2383
Huang H, Zhu ZQ, Zhou ZG et al (2016) Contrast-enhanced transrectal ultrasound for prediction of prostate cancer aggressiveness: the role of normal peripheral zone time-intensity curves. Sci Rep 6:38643
Halpern EJ, Gomella LG, Forsberg F et al (2012) Contrast enhanced transrectal ultrasound for the detection of prostate cancer: a randomized, double-blind trial of dutasteride pretreatment. J Urol 188:1739–1745
Sano F, Terao H, Kawahara T et al (2011) Contrast-enhanced ultrasonography of the prostate: various imaging findings that indicate prostate cancer. BJU Int 107:1404–1410
Xie SW, Li HL, Du J et al (2012) Contrast-enhanced ultrasonography with contrast-tuned imaging technology for the detection of prostate cancer: comparison with conventional ultrasonography. BJU Int 109:1620–1626
Epstein JI, Allsbrook WC Jr, Amin MB et al (2005) The 2005 international society of urological pathology (ISUP) consensus conference on gleason grading of prostatic carcinoma. Am J Surg Pathol 29:1228–1242
Karram S, Trock BJ, Netto GJ et al (2011) Should intervening benign tissue be included in the measurement of discontinuous foci of cancer on prostate needle biopsy? Correlation with radical prostatectomy findings. Am J Surg Pathol 35:1351–1355
Ploussard G, Epstein JI, Montironi R et al (2011) The contemporary concept of significant versus insignificant prostate cancer. Eur Urol 60:291–303
Fütterer JJ, Briganti A, De Visschere P et al (2015) Can clinically significant prostate cancer be detected with multiparametric magnetic resonance imaging? A systematic review of the literature. Eur Urol 68:1045–1053
Sonn GA, Chang E, Natarajan S et al (2014) Value of targeted prostate biopsy using magnetic resonance-ultrasound fusion in men with prior negative biopsy and elevated prostate-specific antigen. Eur Urol 65:809–815
Russo F, Regge D, Armando E et al (2016) Detection of prostate cancer index lesions with multiparametric magnetic resonance imaging (mp-MRI) using whole-mount histological sections as the reference standard. BJU Int 118:84–94
Siddiqui MM, Rais-Bahrami S, Truong H et al (2013) Magnetic resonance imaging/ultrasound-fusion biopsy significantly upgrades prostate cancer versus systematic 12-core transrectal ultrasound biopsy. Eur Urol 64:713–719
Kaufmann S, Russo GI, Bamberg F et al (2018) Prostate cancer detection in patients with prior negative biopsy undergoing cognitive-, robotic- or in-bore MRI target biopsy. World J Urol 36:761–768
Mitterberger MJ, Aigner F, Horninger W et al (2010) Comparative efficiency of contrast-enhanced colour Doppler ultrasound targeted versus systematic biopsy for prostate cancer detection. Eur Radiol 20:2791–2796
Punglia RS, D’Amico AV, Catalona WJ et al (2003) Effect of verification bias on screening for prostate cancer by measurement of prostate-specific antigen. N Engl J Med 349:335–342
El Hajj A, Ploussard G, de la Taille A et al (2013) Analysis of outcomes after radical prostatectomy in patients eligible for active surveillance (PRIAS). BJU Int 111:53–59
Funding
This work was supported by a grant from the National Natural Science Foundation of China (no. 81671708), the Science and Technology Commission of Shanghai Municipality (no. 15411961000), the Shanghai Shen Kang Hospital Development Center Research Project (no. 16CR3092B) and the Fund of Shanghai Jiaotong University (no. YG2014ZD04).
Author information
Authors and Affiliations
Contributions
ZY: data collection and management, data analysis, manuscript writing. CY: project development, manuscript editing. JJ: data collection, data analysis, manuscript editing. QT: data collection, data analysis. LW: data collection. QY: data collection. GW: data collection. WL: data collection. QJ: project development, data collection.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Yunkai, Z., Yaqing, C., Jun, J. et al. Comparison of contrast-enhanced ultrasound targeted biopsy versus standard systematic biopsy for clinically significant prostate cancer detection: results of a prospective cohort study with 1024 patients. World J Urol 37, 805–811 (2019). https://doi.org/10.1007/s00345-018-2441-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2441-1