Abstract
Purpose
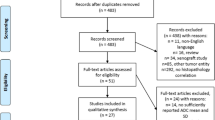
To evaluate the use of multiparametric MRI (mp MRI) parameters in order to predict prostate cancer aggressiveness as defined by pathological Gleason score or molecular markers in a cohort of patients defined with a Gleason score of 6 at biopsy.
Methods
Sixty-seven men treated by radical prostatectomy (RP) for a low grade (Gleason 6) on biopsy and mp MRI before biopsy were selected. The cycle cell proliferation (CCP) score assessed by the Prolaris test and Ki-67/PTEN expression assessed by immunohistochemistry were quantified on the RP specimens.
Results
49.25 % of the cancers were undergraded on biopsy compared to the RP specimens. Apparent diffusion coefficient (ADC) < 0.80 × 10−3 mm2/s (P value 0.003), Likert score >4 (P value 0.003) and PSA density >0.15 ng/ml/cc (P value 0.035) were significantly associated with a higher RP Gleason score. Regarding molecular markers of aggressiveness, ADC < 0.80 × 10−3 mm2/s and Likert score >4 were also significantly associated with a positive staining for Ki-67 (P value 0.039 and 0.01, respectively). No association was found between any analyzed MRI or clinical parameter and the CCP score.
Conclusion
Decreasing ADC value is a stronger indicator of aggressive prostate cancer as defined by molecular markers or postsurgical histology than biopsy characteristics.


Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Ervik M, Dikshit R et al (2012) GLOBOCAN v1.0, cancer incidence and mortality worldwide: IARC cancer base no. 11 [Internet]. Lyon, France: International Agency for Research on Cancer; 2013. http://globocan.iarc.fr. Accessed on 11/01/2016
Partin AW, Kattan MW, Subong EN et al (1997) Combination of prostate-specific antigen, clinical stage, and Gleason score to predict pathological stage of localized prostate cancer. A multi-institutional update. JAMA 277:1445–1451
Sved PD, Gomez P, Manoharan M, Kim SS, Soloway MS (2004) Limitations of biopsy Gleason grade: implications for counseling patients with biopsy Gleason score 6 prostate cancer. J Urol 172:98–102
Cuzick J, Berney DM, Fisher G et al (2012) Prognostic value of a cell cycle progression signature for prostate cancer death in a conservatively managed needle biopsy cohort. Br J Cancer 106:1095–1099. doi:10.1038/bjc.2012.39
Verma R, Gupta V, Singh J et al (2015) Significance of p53 and ki-67 expression in prostate cancer. Urol Ann 7:488–493. doi:10.4103/0974-7796.158507
Cuzick J, Yang ZH, Fisher G et al (2013) Prognostic value of PTEN loss in men with conservatively managed localised prostate cancer. Br J Cancer 108:2582–2589. doi:10.1038/bjc.2013.248
Hambrock T, Somford DM, Huisman HJ et al (2011) Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 259:453–461. doi:10.1148/radiol.11091409
Tamada T, Sone T, Jo Y et al (2008) Apparent diffusion coefficient values in peripheral and transition zones of the prostate: comparison between normal and malignant prostatic tissues and correlation with histologic grade. J Magn Reson Imaging 28:720–726. doi:10.1002/jmri.21503
Itou Y, Nakanishi K, Narumi Y, Nishizawa Y, Tsukuma H (2011) Clinical utility of apparent diffusion coefficient (ADC) values in patients with prostate cancer: Can ADC values contribute to assess the aggressiveness of prostate cancer? J Magn Reson Imaging 33:167–172. doi:10.1002/jmri.22317
Verma S, Rajesh A, Morales H et al (2011) Assessment of aggressiveness of prostate cancer: correlation of apparent diffusion coefficient with histologic grade after radical prostatectomy. AJR Am J Roentgenol 196:374–381. doi:10.2214/AJR.10.4441
Renard-Penna R, Cancel-Tassin G, Comperat E et al (2015) Multiparametric magnetic resonance imaging predicts postoperative pathology but misses aggressive prostate cancers as assessed by cell cycle progression score. J Urol 194:1617–1623. doi:10.1016/j.juro.2015.06.107
Puech P, Rouviere O, Renard-Penna R et al (2013) Prostate cancer diagnosis: multiparametric MR-targeted biopsy with cognitive and transrectal US–MR fusion guidance versus systematic biopsy-prospective multicenter study. Radiology 268:461–469. doi:10.1148/radiol.13121501
Renard-Penna R, Roupret M, Comperat E et al (2013) Accuracy of high resolution (1.5 tesla) pelvic phased array magnetic resonance imaging (MRI) in staging prostate cancer in candidates for radical prostatectomy: results from a prospective study. Urol Oncol 31:448–454. doi:10.1016/j.urolonc.2011.02.017
Barentsz JO, Richenberg J, Clements R et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757. doi:10.1007/s00330-011-2377-y
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69:16–40. doi:10.1016/j.eururo.2015.08.052
Schuetz A, Deleage C, Sereti I et al (2014) Initiation of ART during early acute HIV infection preserves mucosal Th17 function and reverses HIV-related immune activation. PLoS Pathog 10:e1004543. doi:10.1371/journal.ppat.1004543
Partin AW, Kattan MW, Subong EN et al (1997) Combination of prostate-specific antigen, clinical stage, and Gleason score to predict pathological stage of localized prostate cancer. A multi-institutional update. JAMA 277:1445–1451
Meiers I, Waters DJ, Bostwick DG (2007) Preoperative prediction of multifocal prostate cancer and application of focal therapy: review 2007. Urology 70:3–8. doi:10.1016/j.urology.2007.06.1129
Greene KL, Cowan JE, Cooperberg MR et al (2005) Who is the average patient presenting with prostate cancer? Urology 66:76–82
Louie-Johnsun M, Neill M, Treurnicht K, Jarmulowicz M, Eden C (2009) Final outcomes of patients with low-risk prostate cancer suitable for active surveillance but treated surgically. BJU Int 104:1501–1504. doi:10.1111/j.1464-410X.2009.08597.x
Itatani R, Namimoto T, Kajihara H et al (2014) Triage of low-risk prostate cancer patients with PSA levels 10 ng/ml or less: comparison of apparent diffusion coefficient value and transrectal ultrasound-guided target biopsy. AJR Am J Roentgenol 202:1051–1057. doi:10.2214/AJR.13.11602
De Cobelli F, Ravelli S, Esposito A et al (2015) Apparent diffusion coefficient value and ratio as noninvasive potential biomarkers to predict prostate cancer grading: comparison with prostate biopsy and radical prostatectomy specimen. AJR Am J Roentgenol 204:550–557. doi:10.2214/AJR.14.13146
Kobus T, Vos PC, Hambrock T et al (2012) Prostate cancer aggressiveness: in vivo assessment of MR spectroscopy and diffusion-weighted imaging at 3 T. Radiology 265:457–467. doi:10.1148/radiol.12111744
Bittencourt LK, Barentsz JO, de Miranda LC, Gasparetto EL (2012) Prostate MRI: diffusion-weighted imaging at 1.5T correlates better with prostatectomy Gleason Grades than TRUS-guided biopsies in peripheral zone tumours. Eur Radiol 22:468–475. doi:10.1007/s00330-011-2269-1
Vargas HA, Akin O, Franiel T et al (2011) Diffusion-weighted endorectal MR imaging at 3 T for prostate cancer: tumor detection and assessment of aggressiveness. Radiology 259:775–784. doi:10.1148/radiol.11102066
Renard-Penna R, Mozer P, Cornud F et al (2015) Prostate imaging reporting and data system and Likert scoring system: multiparametric MR imaging validation study to screen patients for initial biopsy. Radiology 275:458–468. doi:10.1148/radiol.14140184
Harada T, Abe T, Kato F et al (2015) Five-point Likert scaling on MRI predicts clinically significant prostate carcinoma. BMC Urol 15:9. doi:10.1186/s12894-015-0087-5
Benson MC, Whang IS, Pantuck A et al (1992) Prostate specific antigen density: a means of distinguishing benign prostatic hypertrophy and prostate cancer. J Urol 147:815–816
Revelos K, Petraki C, Gregorakis A et al (2005) p27(kip1) and Ki-67 (MIB1) immunohistochemical expression in radical prostatectomy specimens of patients with clinically localized prostate cancer. In Vivo 19:911–920
Zhang J, Jing H, Han X, Huang Z, Cao Z, Liu Q (2013) Diffusion-weighted imaging of prostate cancer on 3T MR: relationship between apparent diffusion coefficient values and Ki-67 expression. Acad Radiol 20:1535–1541. doi:10.1016/j.acra.2013.09.007
Cho E, Chung DJ, Yeo DM et al (2015) Optimal cut-off value of perfusion parameters for diagnosing prostate cancer and for assessing aggressiveness associated with Gleason score. Clin Imaging 39:834–840. doi:10.1016/j.clinimag.2015.04.020
Vignati A, Mazzetti S, Giannini V et al (2015) Texture features on T2-weighted magnetic resonance imaging: new potential biomarkers for prostate cancer aggressiveness. Phys Med Biol 60:2685–2701. doi:10.1088/0031-9155/60/7/2685
Acknowledgments
The authors thank Cecile Gaffory for helpful assistance in manuscript preparation. The authors thank Patrick Korman, Myriad Genetics SAS, France and Steve Stone, Myriad Genetics, Salt Lake City, UT, for blinded evaluation of gene expression signatures.
Author’s contribution
RRenard-Penna was involved in protocol/project development and data collection, analyzed the data and wrote the manuscript; G. Cancel-Tassin analyzed the data and wrote the manuscript; E. Comperat was involved in data collection, analyzed the data and wrote the manuscript; P. Mozer, P. Léon, J. Varinot and M. Roupret were involved in data collection; M. O. Bitker and O. Lucidarme were involved in data management; and O. Cussenot was involved in protocol/project development and data management, analyzed the data and wrote the manuscript.
Funding
Supported by Myriad Genetics which performed genetic tests. No author received any personal compensation for the study, financial or otherwise, nor does any investigator stand to benefit financially from the publication of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Olivier Cussenot has received a speaker honorarium and financial support for attending symposia from Myriad Genetics SAS, France. The other authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Renard Penna, R., Cancel-Tassin, G., Comperat, E. et al. Apparent diffusion coefficient value is a strong predictor of unsuspected aggressiveness of prostate cancer before radical prostatectomy. World J Urol 34, 1389–1395 (2016). https://doi.org/10.1007/s00345-016-1789-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-016-1789-3