Purpose
Abstract
Muscle weakness in long-stay ICU patients contributes to 1-year mortality. Whether electrophysiological screening is an alternative diagnostic tool in unconscious/uncooperative patients remains unknown. We aimed to determine the diagnostic properties of abnormal compound muscle action potential (CMAP), sensory nerve action potential (SNAP), and spontaneous electrical activity (SEA) for Medical Research Council (MRC)-defined weakness and their predictive value for 1-year mortality.
Methods
Data were prospectively collected during the EPaNIC trial (ClinicalTrials.gov: NCT00512122). First, sensitivity, specificity, positive (PPV) and negative predictive values (NPV) of abnormal CMAP, SNAP, and SEA for weakness were determined. Subsequently, association between 1-year mortality and abnormal findings on electrophysiological screening was assessed by univariate and multivariate analyses correcting for weakness and other risk factors and the prediction model involved only a development phase.
Results
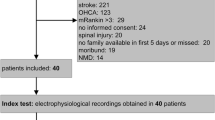
A total of 730 patients were electrophysiologically screened of whom 432 were tested for weakness. On day 8, normal CMAP excluded weakness with a high NPV (80.5 %). By day 15, abnormal SNAP and the presence of SEA had a high PPV (91.7 and 80.0 %, respectively). Only a reduced CMAP on day 8 was associated with higher 1-year mortality [35.6 vs 15.2 % (p < 0.001)]. This association remained significant after correction for weakness and other risk factors [OR 2.463 (95 % CI 1.113–5.452), p = 0.026]. Also among conscious/cooperative patients without weakness, reduced CMAP was independently associated with a higher likelihood of death occurring during 1 year [HR 2.818 (95 % CI 1.074–7.391), p = 0.035].
Conclusions
The diagnostic properties of electrophysiological screening vary over time. Abnormal CMAP documented early during critical illness carries information about longer-term outcome, which should be further investigated mechanistically.



Similar content being viewed by others
References
Kress JP, Hall JB (2014) ICU-acquired weakness and recovery from critical illness. N Engl J Med 370:1626–1635
Fan E, Cheek F, Chlan L, Gosselink R, Hart N, Herridge MS, Hopkins RO, Hough CL, Kress JP, Latronico N, Moss M, Needham DM, Rich MM, Stevens RD, Wilson KC, Winkelman C, Zochodne DW, Ali NA, ATS Committee on ICU-acquired Weakness in Adults, American Thoracic Society (2014) An official American Thoracic Society clinical practice guideline: the diagnosis of intensive care unit-acquired weakness in adults. Am J Respir Crit Care Med 190:1437–1446
De Jonghe B, Sharshar T, Lefaucheur JP, Authier FJ, Durand-Zaleski I, Boussarsar M, Cerf C, Renaud E, Mesrati F, Carlet J, Raphael JC, Outin H, Bastuji-Garin S (2002) Paresis acquired in the intensive care unit: a prospective multicenter study. JAMA 288:2859–2867
Stevens RD, Dowdy DW, Michaels RK, Mendez-Tellez PA, Pronovost PJ, Needham DM (2007) Neuromuscular dysfunction acquired in critical illness: a systematic review. Intensive Care Med 33:1876–1891
De Jonghe B, Bastuji-Garin S, Durand MC, Malissin I, Rodrigues P, Cerf C, Outin H, Sharshar T (2007) Respiratory weakness is associated with limb weakness and delayed weaning in critical illness. Crit Care Med 35:2007–2015
Sharshar T, Bastuji-Garin S, Stevens RD, Durand MC, Malissin I, Rodriguez P, Cerf C, Outin H, De Jonghe B (2009) Presence and severity of intensive care unit-acquired paresis at time of awakening are associated with increased intensive care unit and hospital mortality. Crit Care Med 37:3047–3053
Ali NA, O’Brien JM Jr, Hoffmann SP, Phillips G, Garland A, Finley JC, Almoosa K, Hejal R, Wolf KM, Lemeshow S, Connors AF Jr, Marsh CB (2008) Acquired weakness, handgrip strength, and mortality in critically ill patients. Am J Respir Crit Care Med 178:261–268
Hermans G, Van Mechelen H, Clerckx B, Vanhullebusch T, Mesotten D, Wilmer A, Casaer MP, Meersseman P, Debaveye Y, Van Cromphaut S, Wouters PJ, Gosselink R, Van den Berghe G (2014) Acute outcomes and 1-year mortality of intensive care unit-acquired weakness. A cohort study and propensity-matched analysis. Am J Respir Crit Care Med 190:410–420
Herridge MS, Tansey CM, Matte A, Tomlinson G, az-Granados N, Cooper A, Guest CB, Mazer CD, Mehta S, Stewart TE, Kudlow P, Cook D, Slutsky AS, Cheung AM (2011) Functional disability 5 years after acute respiratory distress syndrome. N Engl J Med 364:1293–1304
Fan E, Dowdy DW, Colantuoni E, Mendez-Tellez PA, Sevransky JE, Shanholtz C, Himmelfarb CR, Desai SV, Ciesla N, Herridge MS, Pronovost PJ, Needham DM (2014) Physical complications in acute lung injury survivors: a two-year longitudinal prospective study. Crit Care Med 42:849–859
Hermans G, Casaer MP, Clerckx B, Guiza F, Vanhullebusch T, Derde S, Meersseman P, Derese I, Mesotten D, Wouters P, Van Cromphaut S, Debaveye Y, Gosselink R, Gunst J, Wilmer A, Van den Berghe G, Vanhorebeek I (2013) Effect of tolerating macronutrient deficit on the development of intensive-care unit acquired weakness: a subanalysis of the EPaNIC trial. Lancet Respir Med 1:621–629
Latronico N, Bolton CF (2011) Critical illness polyneuropathy and myopathy: a major cause of muscle weakness and paralysis. Lancet Neurol 10:931–941
Lacomis D (2013) Electrophysiology of neuromuscular disorders in critical illness. Muscle Nerve 47:452–463
Latronico N, Smith M (2014) Introducing simplified electrophysiological test of peripheral nerves and muscles in the ICU: choosing wisely. Intensive Care Med 40:746–748
Moss M, Yang M, Macht M, Sottile P, Gray L, McNulty M, Quan D (2014) Screening for critical illness polyneuromyopathy with single nerve conduction studies. Intensive Care Med 40:683–690
Moons KG, Altman DG, Reitsma JB, Ioannidis JP, Macaskill P, Steyerberg EW, Vickers AJ, Ransohoff DF, Collins GS (2015) Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): explanation and elaboration. Ann Intern Med 162:W1–73
Casaer MP, Hermans G, Wilmer A, Van den Berghe G (2011) Impact of early parenteral nutrition completing enteral nutrition in adult critically ill patients (EPaNIC trial): a study protocol and statistical analysis plan for a randomized controlled trial. Trials 12:21
Casaer MP, Mesotten D, Hermans G, Wouters PJ, Schetz M, Meyfroidt G, Van Cromphaut S, Ingels C, Meersseman P, Muller J, Vlasselaers D, Debaveye Y, Desmet L, Dubois J, Van Assche A, Vanderheyden S, Wilmer A, Van den Berghe G (2011) Early versus late parenteral nutrition in critically ill adults. N Engl J Med 365:506–517
Latronico N, Bertolini G, Guarneri B, Botteri M, Peli E, Andreoletti S, Bera P, Luciani D, Nardella A, Vittorielli E, Simini B, Candiani A (2007) Simplified electrophysiological evaluation of peripheral nerves in critically ill patients: the Italian multi-centre CRIMYNE study. Crit Care 11:R11
Hermans G, Van Mechelen H, Clerckx B, Vanhullebusch T, Mesotten D, Wilmer A, Casaer MP, Meersseman P, Debaveye Y, Van Cromphaut S, Wouters PJ, Gosselink R, Van den Berghe G (2014) Acute outcomes and 1-year mortality of ICU-acquired weakness: a cohort study and propensity matched analysis. Am J Respir Crit Care Med 190:410–420
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, Moher D, Rennie D, de Vet HC, Lijmer JG, Standards for Reporting of Diagnostic Accuracy Group (2003) The STARD statement for reporting studies of diagnostic accuracy: explanation and elaboration. Ann Intern Med 138:W1–12
Midi H, Sarkar SK, Rana S (2010) Collinearity diagnostics of binary logistic regression model. J Interdiscip Math 13:253–267
SAS Institute (1999) StatView: Using StatView. SAS Institute, ISBN 9781580251624
Latronico N, Nattino G, Guarneri B, Fagoni N, Amantini A, Bertolini G, Gi VSI (2014) Validation of the peroneal nerve test to diagnose critical illness polyneuropathy and myopathy in the intensive care unit: the multicentre Italian CRIMYNE-2 diagnostic accuracy study. F1000Res 3:127
Weber-Carstens S, Koch S, Spuler S, Spies CD, Bubser F, Wernecke KD, Deja M (2009) Nonexcitable muscle membrane predicts intensive care unit-acquired paresis in mechanically ventilated, sedated patients. Crit Care Med 37:2632–2637
Wieske L, Verhamme C, Witteveen E, Bouwes A, Dettling-Ihnenfeldt DS, van der Schaaf M, Schultz MJ, van Schaik IN, Horn J (2015) Feasibility and diagnostic accuracy of early electrophysiological recordings for ICU-acquired weakness: an observational cohort study. Neurocrit Care 22:385–394
Bednarik J, Lukas Z, Vondracek P (2003) Critical illness polyneuromyopathy: the electrophysiological components of a complex entity. Intensive Care Med 29:1505–1514
Witt NJ, Zochodne DW, Bolton CF, Grand’Maison F, Wells G, Young GB, Sibbald WJ (1991) Peripheral nerve function in sepsis and multiple organ failure. Chest 99:176–184
Lefaucheur JP, Nordine T, Rodriguez P, Brochard L (2006) Origin of ICU acquired paresis determined by direct muscle stimulation. J Neurol Neurosurg Psychiatry 77:500–506
Zifko UA, Zipko HT, Bolton CF (1998) Clinical and electrophysiological findings in critical illness polyneuropathy. J Neurol Sci 159:186–193
Tennila A, Salmi T, Pettila V, Roine RO, Varpula T, Takkunen O (2000) Early signs of critical illness polyneuropathy in ICU patients with systemic inflammatory response syndrome or sepsis. Intensive Care Med 26:1360–1363
Cankayali I, Dogan YH, Solak I, Demirag K, Eris O, Demirgoren S, Moral AR (2007) Neuromuscular deterioration in the early stage of sepsis in rats. Crit Care 11:R1
Van den Berghe G, Schoonheydt K, Becx P, Bruyninckx F, Wouters PJ (2005) Insulin therapy protects the central and peripheral nervous system of intensive care patients. Neurology 64:1348–1353
Hermans G, Wilmer A, Meersseman W, Milants I, Wouters PJ, Bobbaers H, Bruyninckx F, Van den Berghe G (2007) Impact of intensive insulin therapy on neuromuscular complications and ventilator-dependency in MICU. Am J Respir Crit Care Med 175:480–489
Garnacho-Montero J, Madrazo-Osuna J, Garcia-Garmendia JL, Ortiz-Leyba C, Jimenez-Jimenez FJ, Barrero-Almodovar A, Garnacho-Montero MC, Moyano-Del-Estad MR (2001) Critical illness polyneuropathy: risk factors and clinical consequences. A cohort study in septic patients. Intensive Care Med 27:1288–1296
Garnacho-Montero J, Amaya-Villar R, Garcia-Garmendia JL, Madrazo-Osuna J, Ortiz-Leyba C (2005) Effect of critical illness polyneuropathy on the withdrawal from mechanical ventilation and the length of stay in septic patients. Crit Care Med 33:349–354
Khan J, Harrison TB, Rich MM, Moss M (2006) Early development of critical illness myopathy and neuropathy in patients with severe sepsis. Neurology 67:1421–1425
Leijten FS, Harinck-de Weerd JE, Poortvliet DC, de Weerd AW (1995) The role of polyneuropathy in motor convalescence after prolonged mechanical ventilation. JAMA 274:1221–1225
Druschky A, Herkert M, Radespiel-Troger M, Druschky K, Hund E, Becker CM, Hilz MJ, Erbguth F, Neundorfer B (2001) Critical illness polyneuropathy: clinical findings and cell culture assay of neurotoxicity assessed by a prospective study. Intensive Care Med 27:686–693
Weber-Carstens S, Schneider J, Wollersheim T, Assmann A, Bierbrauer J, Marg A, Al Hasani H, Chadt A, Wenzel K, Koch S, Fielitz J, Kleber C, Faust K, Mai K, Spies CD, Luft FC, Boschmann M, Spranger J, Spuler S (2013) Critical illness myopathy and GLUT4: significance of insulin and muscle contraction. Am J Respir Crit Care Med 187:387–396
Latronico N, Fenzi F, Recupero D, Guarneri B, Tomelleri G, Tonin P, De Maria G, Antonini L, Rizzuto N, Candiani A (1996) Critical illness myopathy and neuropathy. Lancet 347:1579–1582
Rich MM, Bird SJ, Raps EC, McCluskey LF, Teener JW (1997) Direct muscle stimulation in acute quadriplegic myopathy. Muscle Nerve 20:665–673
Trojaborg W, Weimer LH, Hays AP (2001) Electrophysiologic studies in critical illness associated weakness: myopathy or neuropathy—a reappraisal. Clin Neurophysiol 112:1586–1593
Acknowledgments
We are indebted to all patients and their families for their participation in this study. The clinical trial assistants, in particular Alexandra Hendrickx, Sylvia Van Hulle, and Jenny Gielens, are acknowledged for administrative support. We are grateful to the ICU nursing and medical staff for excellent patient care. This work was supported by the Research Foundation-Flanders (FWO), Belgium (G.0399.12, G.0592.12). GH and MPC hold a Postdoctoral Fellowship from the Clinical Research Fund (KOF) of the University Hospitals Leuven, Belgium. GVdB, via the University of Leuven, receives structural research financing via the Methusalem program, funded by the Flemish Government (METH08/07) and holds an European Research Council (ERC) Advanced grant (AdvG-2012-321670) from the Ideas Program of the EU FP7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they do not have any conflict of interest.
Additional information
G. Hermans and H. Van Mechelen contributed equally.
Take-home message: The diagnostic properties of electrophysiological screening vary over time. Abnormal CMAP documented early during critical illness carries information about longer-term outcome.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hermans, G., Van Mechelen, H., Bruyninckx, F. et al. Predictive value for weakness and 1-year mortality of screening electrophysiology tests in the ICU. Intensive Care Med 41, 2138–2148 (2015). https://doi.org/10.1007/s00134-015-3979-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-015-3979-7