Abstract
Purpose
To assess the impact of 6 % tetrastarch [hydroxyethyl starch (HES) 130/0.4 and 130/0.42] in severe sepsis patients. The primary outcome measure was 90-day mortality.
Methods
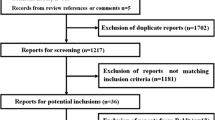
A structured literature search was undertaken to identify prospective randomised controlled trials (RCTs) in adult patients with severe sepsis receiving 6 % tetrastarch (of potato or waxy maize origin) as part of fluid resuscitation in comparison with other non-HES fluids after randomisation in the critical care setting. A systematic review and meta-analysis were performed.
Results
Six RCTs were included (n = 3,033): three from 2012 (n = 2,913) had low risk of bias. Median tetrastarch exposure was 37.4 ml/kg (range 30–43 ml/kg). Ninety-day mortality was associated with tetrastarch exposure [relative risk (RR) 1.13; 95 % confidence interval (CI) 1.02–1.25; p = 0.02] compared with crystalloid. The number needed to harm (NNH) was 28.8 (95 % CI 14.6–942.5). Publication bias and statistical heterogeneity (I 2 = 0 %) were not present. Tetrastarch exposure was also associated with renal replacement therapy (p = 0.01; NNH 15.7) and allogeneic transfusion support (p = 0.001; NNH 9.9). No difference between groups was observed for 28-day mortality, for comparison with colloid as control, or for waxy maize-derived tetrastarch, but power was lacking. Overall mortality was associated with tetrastarch exposure (RR 1.13; 95 % CI 1.02–1.25; p = 0.02).
Conclusions
In our analysis, 6 % tetrastarch as part of initial fluid resuscitation for severe sepsis was associated with harm and, as alternatives exist, in our view should be avoided.






Similar content being viewed by others
References
Finfer S, Liu B, Taylor C, Bellomo R, Billot L, Cook D, Du B, McArthur C, Myburgh J (2010) Resuscitation fluid use in critically ill adults: an international cross-sectional study in 391 intensive care units. Crit Care 14:R185
Investigators FS, Trials SCC (2008) Preferences for colloid use in Scandinavian intensive care units. Acta Anaesthesiol Scand 52:750–758
Myburgh JA, Finfer S, Bellomo R, Billot L, Cass A, Gattas D, Glass P, Lipman J, Liu B, McArthur C, McGuinness S, Rajbhandari D, Taylor CB, Webb SA (2012) Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med 367:1901–1911
Hartog CS, Kohl M, Reinhart K (2011) A systematic review of third-generation hydroxyethyl starch (HES 130/0.4) in resuscitation: safety not adequately addressed. Anesth Analg 112:635–645
Wiedermann CJ, Joannidis M (2012) Mortality after hydroxyethyl starch 130/0.4 infusion: an updated meta-analysis of randomized trials. Swiss Med Wkly 142:w13656
Hartog CS, Skupin H, Natanson C, Sun J, Reinhart K (2012) Systematic analysis of hydroxyethyl starch (HES) reviews: proliferation of low-quality reviews overwhelms the results of well-performed meta-analyses. Intensive Care Med 38:1258–1271
Reinhart K, Perner A, Sprung CL, Jaeschke R, Schortgen F, Johan Groeneveld AB, Beale R, Hartog CS, European Society of Intensive Care M (2012) Consensus statement of the ESICM task force on colloid volume therapy in critically ill patients. Intensive Care Med 38:368–383
Brunkhorst FM, Engel C, Bloos F, Meier-Hellmann A, Ragaller M, Weiler N, Moerer O, Gruendling M, Oppert M, Grond S, Olthoff D, Jaschinski U, John S, Rossaint R, Welte T, Schaefer M, Kern P, Kuhnt E, Kiehntopf M, Hartog C, Natanson C, Loeffler M, Reinhart K (2008) Intensive insulin therapy and pentastarch resuscitation in severe sepsis. N Engl J Med 358:125–139
Schortgen F, Lacherade JC, Bruneel F, Cattaneo I, Hemery F, Lemaire F, Brochard L (2001) Effects of hydroxyethylstarch and gelatin on renal function in severe sepsis: a multicentre randomised study. Lancet 357:911–916
Shafer SL (2011) Shadow of doubt. Anesth Analg 112:498–500
Reinhart K, Takala J (2011) Hydroxyethyl starches: what do we still know? Anesth Analg 112:507–511
Wiedermann CJ (2008) Systematic review of randomized clinical trials on the use of hydroxyethyl starch for fluid management in sepsis. BMC Emerg Med 8:1
Gattas DJ, Dan A, Myburgh J, Billot L, Lo S, Finfer S (2012) Fluid resuscitation with 6% hydroxyethyl starch (130/0.4) in acutely ill patients: an updated systematic review and meta-analysis. Anesth Analg 114:159–169
Bunn F, Trivedi D (2012) Colloid solutions for fluid resuscitation. Cochrane Database Syst Rev 6:CD001319
Perel P, Roberts I (2012) Colloids versus crystalloids for fluid resuscitation in critically ill patients. Cochrane Database Syst Rev 6:CD000567
Freiman JA, Chalmers TC, Smith H Jr, Kuebler RR (1978) The importance of beta, the type II error and sample size in the design and interpretation of the randomized control trial. Survey of 71 “negative” trials. N Engl J Med 299:690–694
Kasper SM, Meinert P, Kampe S, Gorg C, Geisen C, Mehlhorn U, Diefenbach C (2003) Large-dose hydroxyethyl starch 130/0.4 does not increase blood loss and transfusion requirements in coronary artery bypass surgery compared with hydroxyethyl starch 200/0.5 at recommended doses. Anesthesiology 99:42–47
Kozek-Langenecker SA, Jungheinrich C, Sauermann W, Van der Linden P (2008) The effects of hydroxyethyl starch 130/0.4 (6%) on blood loss and use of blood products in major surgery: a pooled analysis of randomized clinical trials. Anesth Analg 107:382–390
Neff TA, Doelberg M, Jungheinrich C, Sauerland A, Spahn DR, Stocker R (2003) Repetitive large-dose infusion of the novel hydroxyethyl starch 130/0.4 in patients with severe head injury. Anesth Analg 96:1453–1459 table of contents
Niemi T, Schramko A, Kuitunen A, Kukkonen S, Suojaranta-Ylinen R (2008) Haemodynamics and acid-base equilibrium after cardiac surgery: comparison of rapidly degradable hydroxyethyl starch solutions and albumin. Scand J Surg 97:259–265
Schramko AA, Suojaranta-Ylinen RT, Kuitunen AH, Kukkonen SI, Niemi TT (2009) Rapidly degradable hydroxyethyl starch solutions impair blood coagulation after cardiac surgery: a prospective randomized trial. Anesth Analg 108:30–36
Ranieri VM, Thompson BT, Barie PS, Dhainaut JF, Douglas IS, Finfer S, Gardlund B, Marshall JC, Rhodes A, Artigas A, Payen D, Tenhunen J, Al-Khalidi HR, Thompson V, Janes J, Macias WL, Vangerow B, Williams MD (2012) Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med 366:2055–2064
Russell JA, Walley KR, Singer J, Gordon AC, Hebert PC, Cooper DJ, Holmes CL, Mehta S, Granton JT, Storms MM, Cook DJ, Presneill JJ, Ayers D (2008) Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med 358:877–887
Sprung CL, Annane D, Keh D, Moreno R, Singer M, Freivogel K, Weiss YG, Benbenishty J, Kalenka A, Forst H, Laterre PF, Reinhart K, Cuthbertson BH, Payen D, Briegel J (2008) Hydrocortisone therapy for patients with septic shock. N Engl J Med 358:111–124
Perner A, Haase N, Guttormsen AB, Tenhunen J, Klemenzson G, Aneman A, Madsen KR, Moller MH, Elkjaer JM, Poulsen LM, Bendtsen A, Winding R, Steensen M, Berezowicz P, Soe-Jensen P, Bestle M, Strand K, Wiis J, White JO, Thornberg KJ, Quist L, Nielsen J, Andersen LH, Holst LB, Thormar K, Kjaeldgaard AL, Fabritius ML, Mondrup F, Pott FC, Moller TP, Winkel P, Wetterslev J (2012) Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med 367:124–134
Guidet B, Martinet O, Boulain T, Philippart F, Poussel JF, Maizel J, Forceville X, Feissel M, Hasselmann M, Heininger A, Van Aken H (2012) Assessment of hemodynamic efficacy and safety of 6% hydroxyethylstarch 130/0.4 vs. 0.9% NaCl fluid replacement in patients with severe sepsis: the CRYSTMAS study. Crit Care 16:R94
Cohen J, Guyatt G, Bernard GR, Calandra T, Cook D, Elbourne D, Marshall J, Nunn A, Opal S (2001) New strategies for clinical trials in patients with sepsis and septic shock. Crit Care Med 29:880–886
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Workgroup A (2004) Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 8:R204–R212
Cook DJ, Guyatt GH, Ryan G, Clifton J, Buckingham L, Willan A, McIlroy W, Oxman AD (1993) Should unpublished data be included in meta-analyses? Current convictions and controversies. JAMA 269:2749–2753
Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, Jaeschke R, Reinhart K, Angus DC, Brun-Buisson C, Beale R, Calandra T, Dhainaut JF, Gerlach H, Harvey M, Marini JJ, Marshall J, Ranieri M, Ramsay G, Sevransky J, Thompson BT, Townsend S, Vender JS, Zimmerman JL, Vincent JL (2008) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2008. Intensive Care Med 34:17–60
Sawyer AG, Ball AD (1981) Statistical power and effect size in marketing-research. J Marketing Res 18:275–290
Sahai H, Khurshid A (1996) Formulae and tables for the determination of sample sizes and power in clinical trials for testing differences in proportions for the two-sample design: a review. Stat Med 15:1–21
Onwuegbuzie AJ, Leech NL (2004) Post hoc power: a concept whose time has come. Underst Stat 3:201–230
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European society of intensive care medicine. Intensive Care Med 22:707–710
Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Dolecek M, Svoboda P, Kantorova I, Scheer P, Sas I, Bibrova J, Radvanova J, Radvan M (2009) Therapeutic influence of 20% albumin versus 6% hydroxyethylstarch on extravascular lung water in septic patients: a randomized controlled trial. Hepatogastroenterology 56:1622–1628
Palumbo D, Servillo G, D’Amato L, Volpe ML, Capogrosso G, De Robertis E, Piazza O, Tufano R (2006) The effects of hydroxyethyl starch solution in critically ill patients. Minerva Anestesiol 72:655–664
Dubin A, Pozo MO, Casabella CA, Murias G, Palizas F Jr, Moseinco MC, Kanoore Edul VS, Palizas F, Estenssoro E, Ince C (2010) Comparison of 6% hydroxyethyl starch 130/0.4 and saline solution for resuscitation of the microcirculation during the early goal-directed therapy of septic patients. J Crit Care 25:659 e1–659 e8
Lv J, Zhao HY, Liu F, An YZ (2012) [The influence of lactate Ringer solution versus hydroxyethyl starch on coagulation and fibrinolytic system in patients with septic shock]. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 24:38–41
Zhu GC, Quan ZY, Shao YS, Zhao JG, Zhang YT (2011) [The study of hypertonic saline and hydroxyethyl starch treating severe sepsis]. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 23:150–153
Li F, Sun H, Han XD (2008) [The effect of different fluids on early fluid resuscitation in septic shock]. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 20:472–475
Protsenko DN, Leiderman IN, Grigor’ev EV, Kokarev EA, Levit AL, Gel’fand BR (2009) [Evaluation of the effectiveness and safety of synthetic colloid solutions in the treatment of severe abdominal sepsis: a randomized comparative study]. Anesteziologiia i reanimatologiia: 9–13
Jacob M, Chappell D, Conzen P, Wilkes MM, Becker BF, Rehm M (2008) Small-volume resuscitation with hyperoncotic albumin: a systematic review of randomized clinical trials. Crit Care 12:R34
Committee TCvHSTCM (2011) The crystalloid versus hydroxyethyl starch trial: protocol for a multi-centre randomised controlled trial of fluid resuscitation with 6% hydroxyethyl starch (130/0.4) compared to 0.9% sodium chloride (saline) in intensive care patients on mortality. Intensive Care Med 37:816–823
Hartog CS, Reinhart K (2012) CRYSTMAS study adds to concerns about renal safety and increased mortality in sepsis patients. Crit Care 16:454
Ertmer C, Rehberg S, Van Aken H, Westphal M (2009) Relevance of non-albumin colloids in intensive care medicine. Best Pract Res Clin Anaesthesiol 23:193–212
Sahin SH, Memis D, Sut N (2009) High C-Reactive protein and amylase levels as prognostic markers in non-pancreatic severe sepsis patients. Trak Univ Tip Fak De 26:9–17
Bellmann R, Feistritzer C, Wiedermann CJ (2012) Effect of molecular weight and substitution on tissue uptake of hydroxyethyl starch. Clin Pharmacokinet 51:225–236
Hartog CS, Brunkhorst FM, Engel C, Meier-Hellmann A, Ragaller M, Welte T, Kuhnt E, Reinhart K (2011) Are renal adverse effects of hydroxyethyl starches merely a consequence of their incorrect use? Wien Klin Wochenschr 123:145–155
Dart AB, Mutter TC, Ruth CA, Taback SP (2010) Hydroxyethyl starch (HES) versus other fluid therapies: effects on kidney function. Cochrane Db Syst Rev
Vincent JL, Sakr Y, Sprung C, Harboe S, Damas P (2008) Are blood transfusions associated with greater mortality rates? Results of the sepsis occurrence in acutely ill patients study. Anesthesiology 108:31–39
Matsota P, Politou M, Kalimeris K, Apostolaki S, Merkouri E, Gialeraki A, Travlou A, Kostopanagiotou G (2010) Do different substitution patterns or plant origin in hydroxyethyl starches affect blood coagulation in vitro? Blood Coagul Fibrin 21:448–451
von Roten I, Madjdpour C, Frascarolo P, Burmeister MA, Fisch A, Schramm S, Bombeli T, Spahn DR (2006) Molar substitution and C2/C6 ratio of hydroxyethyl starch: influence on blood coagulation. Br J Anaesth 96:455–463
Nielsen VG (2005) Colloids decrease clot propagation and strength: role of factor XIII-fibrin polymer and thrombin-fibrinogen interactions. Acta Anaesthesiol Scand 49:1163–1171
Sossdorf M, Marx S, Schaarschmidt B, Otto GP, Claus RA, Reinhart K, Hartog CS, Losche W (2009) HES 130/0.4 impairs haemostasis and stimulates pro-inflammatory blood platelet function. Crit Care 13:R208
Godier A, Durand M, Smadja D, Jeandel T, Emmerich J, Samama CM (2010) Maize- or potato-derived hydroxyethyl starches: is there any thromboelastometric difference? Acta Anaesthesiol Scand 54:1241–1247
Sirtl C, Laubenthal H, Zumtobel V, Kraft D, Jurecka W (1999) Tissue deposits of hydroxyethyl starch (HES): dose-dependent and time-related. Br J Anaesth 82:510–515
Stander S, Szepfalusi Z, Bohle B, Stander H, Kraft D, Luger TA, Metze D (2001) Differential storage of hydroxyethyl starch (HES) in the skin: an immunoelectron-microscopical long-term study. Cell Tissue Res 304:261–269
Schmidt-Hieber M, Loddenkemper C, Schwartz S, Arntz G, Thiel E, Notter M (2006) Hydrops lysosomalis generalisatus–an underestimated side effect of hydroxyethyl starch therapy? Eur J Haematol 77:83–85
Acknowledgments
We acknowledge the Medical Research Council (MRC) Chain–Florey Fellowship scheme based at the Clinical Sciences Centre, Imperial College London. The research was supported by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Patel, A., Waheed, U. & Brett, S.J. Randomised trials of 6 % tetrastarch (hydroxyethyl starch 130/0.4 or 0.42) for severe sepsis reporting mortality: systematic review and meta-analysis. Intensive Care Med 39, 811–822 (2013). https://doi.org/10.1007/s00134-013-2863-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-013-2863-6