Abstract
Objective
To test the hypothesis that alveolar plasminogen activator inhibitor-1 (PAI-1) can identify patients with witnessed aspiration at risk for progression to acute respiratory distress syndrome (ARDS).
Design
Prospective observational study.
Setting
Medical intensive care unit in a tertiary care center.
Patients
Fifty-one patients with witnessed aspiration who had a PaO2/FIO2<300 for a period no less than 4 h from admission.
Interventions
Alveolar fluid sampling was performed within 8 h of intubation via luminal suction of the distal airways using a 13-Fr catheter. Plasma levels were collected simultaneously by venipuncture.
Measurements and results
Alveolar PAI-1 antigen levels were more than five times higher in those who progressed to ARDS than in those with uncomplicated aspiration pneumonitis (2687±1498 ng/ml vs. 587±535 ng/ml, respectively; p<0.001), while plasma levels of PAI-1 antigen were not significantly different between the two groups. The measured activity of PAI-1 antigen paralleled the levels observed in both media. A cut-off level of alveolar PAI-1 >1518 ng/ml was found to be 82.4% (56.6%–96.0%) sensitive and 97.1% (84.6%–99.5%) specific in predicting progression to ARDS. There was also a significant inverse relationship between elevation of PAI-1 antigen levels and the degree of lung injury as assessed by the days of unassisted ventilation (r2=0.37; p<0.001).
Conclusions
Elevation of alveolar PAI-1 antigen levels postaspiration is the consequence of local rather than systemic activation of the fibrinolytic system. Measurement of alveolar PAI-1 antigen levels can be a useful clinical marker in predicting progression to ARDS after gastric aspiration.
Similar content being viewed by others
Introduction
Aspiration pneumonitis is reported to occur in 10–25% of patients hospitalized for overdose [1] and in about 1 in 3,000 patients postanesthesia [2]. The true incidence of the disease has been difficult to determine, considering that no diagnostic marker has been validated prospectively to assess its applicability in detecting the majority of events [3]. The syndrome is associated with a broad spectrum of clinical presentations and clinical outcomes, ranging from mild self-limited episodes to fulminant acute lung injury (ALI) characteristic of adult respiratory distress syndrome (ARDS). In a retrospective analysis of 50 patients who had been observed to aspirate gastric contents, the clinical findings at the onset tended to be similar in all individuals irrespective of their outcomes [4]. However, the subsequent clinical course was characterized by one of three patterns: The majority (62%) showed rapid clinical and radiographic improvement within 2–16 days; a minority (12%) demonstrated a rapid progression with worsening clinical and radiographic picture suggestive of ARDS; and the remainder (26%) exhibited rapid improvement from the initial insult followed by clinical deterioration associated with new or expanding radiographic infiltrates indicative of superimposed nosocomial infection.
Extensive research has demonstrated that activation of coagulation and inhibition of fibrinolysis are thought to underlie the events in ALI [5]. Mechanisms that contribute to the changes in the homeostatic balance include both tissue factor expression and increased plasminogen activator inhibitor (PAI-1) synthesis or release. Human bronchoalveolar lavage samples from ARDS patients have shown the levels of PAI-1 to be particularly elevated [6]. Similar experiments exposing PAI-1 knockout mice to bleomycin showed a decrease in fibrin deposition in the PAI-1 knockout mice, while controls showed PAI-1 to be increased by bleomycin [7].
Because aspiration treatment is largely supportive, early delineation of alveolar injury may allow more aggressive intervention with possible avoidance of detrimental sequelae. To date, a validated marker is lacking that would identify patients who go on to develop ARDS after aspiration. We hypothesized that PAI-1 levels in alveolar fluid become acutely elevated in those at risk for aspiration-induced lung injury and that these levels could predict progression to ARDS.
Methods
Patient population
The study protocol was approved by the Institutional Review Board of the University at Buffalo. The study was conducted in accordance with the Declaration of Helsinki. All intubated patients admitted with drug overdose who had a witnessed aspiration were considered for inclusion if their PaO2/FIO2 was <300 on 5 cm H2O of positive end expiratory pressure for at least 4 h from the time of admission. Exclusion criteria included patients with ARDS, acquired immunodeficiency syndrome, sepsis, or documented prior lung disease.
Alveolar fluid and plasma samplings
Alveolar fluid was obtained within 8 h of intubation via luminal suction of the distal airways using a 13-Fr catheter (Combicath, Saint-Leu-La-Foret Cedex, France) as previously described [8]. A soft 13-Fr suction catheter was advanced blindly into a wedged position in a distal bronchus via the endotracheal tube. Pulmonary edema fluid was collected in a suction trap by gentle suction. Bronchoalveolar lavage (BAL) was performed after removal of the inner catheter and analyzed according to standard criteria [9]. Four aliquots (20 ml each) of sterile saline were instilled and aspirated. The first 20 ml recovered was discarded. The rest of the samples were pooled together and transported to the microbiology laboratory for quantitative culture according to standard criteria. A threshold of ≥102 colony-forming unit (CFU)/ml was used to exclude patients with suspected pneumonia. The alveolar/plasma total protein ratio was measured according to standard methods [10]. Plasma samples were collected simultaneously with the alveolar fluid by venipuncture. Samples were centrifuged at 5,000 rpm for 10 min and stored at −70°C until all assays were performed concurrently.
Data collection
Demographic and clinical data were recorded on study enrollment and included age, gender, lung injury score (LIS) [11], and APACHE II score [12]. The radiologic score was obtained from a frontal chest radiograph. Each lung was divided into two quadrants, and each quadrant was scored either 0 or 1, depending on whether alveolar consolidation was absent or present. The development of ARDS was reported to be present only if it occurred with 72 h of admission. The definition of ARDS was based on the presence of bilateral infiltrates on chest radiograph, PaO2/FIO2 <200, and a pulmonary arterial wedge pressure of <18 mmHg, if measured, or the absence of cardiac dysfunction on a two-dimensional echocardiogram according to the clinical criteria outlined by the American European Consensus Conference [13]. Otherwise, the term “uncomplicated aspiration pneumonitis” (UAP) was used to refer to those who did not progress to ARDS. The severity of lung injury was assessed by the number of days alive with unassisted ventilation over the 28-day period postintubation and mechanical ventilation [14].
PAI-1 antigen and activity assays
Undiluted alveolar fluid and plasma samples were assayed for PAI-1. PAI-1 antigen levels were measured using specific commercially available ELISA according to the manufacturer’s instructions (American Diagnostica, Greenwich, CT, USA). This assay detects both active and latent forms of PAI-1 but not PAI-2. PAI-1 activity was assayed spectrophotometrically. Samples were incubated with exogenous tissue-type plasminogen activator (TPA) at room temperature for 10 min in conditions under which PAI-1 but not other low-affinity inhibitors could bind to the TPA. Subsequently, the samples were acidified and snap-frozen to eliminate residual α2-antiplasmin activity. Residual TPA activity was assayed spectrophotometrically by incubation with plasminogen (KabiVitrum, Stockholm, Sweden) and a chromogenic substrate, S-2251 (KabiVitrum, Stockholm, Sweden) [15]. Standard curves were obtained with serial dilutions of pooled normal plasma. Results were expressed as arbitrary units (AU), where 1 AU is defined as the amount of PAI-1 that inhibits 1 IU of TPA within 10 min.
Statistical analysis
Data are expressed as mean ± SD when appropriate. Continuous variables were compared using unpaired Student’s t-test or the Mann–Whitney U-test if the variables were not normally distributed. Categorical variables were compared using chi-square test with Yates correction or Fisher’s exact test when necessary. All assays were performed in duplicate. To determine relevant outcome associations, regression analysis between PAI-1 antigen levels and days of unassisted ventilation was performed. Cook’s distance was used to assess for potential outliers [16]. A receiver operator characteristic (ROC) curve was generated for the PAI-1 antigen levels, and the area under the curve (AUC) was used to determine the accuracy of the PAI-1 antigen levels in predicting progression to ARDS. Statistical significance was set at p≤0.05. All statistical analyses were performed using SPSS software (version 11.0; SPSS, Chicago, IL, USA).
Results
Study population
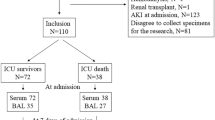
Between February 2003 and April 2005, a total of 63 patients met the inclusion criteria. One patient could not be enrolled because of lack of informed consent, seven were excluded because of bacterial growth ≥100 CFUs/ml in BAL samples, and four had a measured alveolar fluid/plasma total protein ratio <0.65. Of the remaining 51 participants, 29 (57%) were admitted for overdose with tricyclics, eight (15%) with selective serotonin reuptake inhibitors, six (11%) with acetaminophen, three (6%) with ethylene glycol, two (4%) with a combination of muscle relaxants and opioids, two (4%) with benzodiazepines, and one (2%) with salicylate. A total of 39 patients received empiric antimicrobial therapy, seven of whom were initiated prior to BAL. The decision to institute antimicrobial therapy and the choice of antibiotics prescribed were determined by the primary care physicians. Baseline characteristics on admission are shown in Table 1. Seventeen patients developed ARDS during the course of their hospitalization. The groups were comparable in age, gender, severity of illness, lung injury score, and smoking status. Although the radiologic scores were similar between the two groups during the first 8 h of admission, those patients who went to develop ARDS had significantly higher values after 48 h post-intensive care unit (ICU) admission compared with those with UAP (3.1±0.7 vs. 1.4±0.6, respectively; p<0.001). Similarly, the compliance rates and the LIS worsened significantly in those who developed ARDS at 48 h postenrollment, but they improved in those with UAP (Table 2).
PAI-1 antigen levels
To assess whether PAI-1 levels can be useful to discriminate between those with UAP and those who develop ARDS, we measured the PAI-1 antigen level in plasma and alveolar fluid by ELISA in both patient groups. Alveolar PAI-1 antigen was more than five times higher in those who progressed to ARDS than in those with UAP (means ± SD: 2,687±1,498 ng/ml vs. 587±535 ng/ml, respectively; p<0.001) (Fig. 1). In contrast, the difference in plasma PAI-1 antigen level was not significantly different between those with UAP and those who developed ARDS (120±103 ng/ml vs. 138±54 ng/ml, respectively; p=0.4).
To determine whether PAI-1 was predominantly of pulmonary origin, we calculated the ratio of PAI-1 antigen to that of simultaneously drawn plasma. PAI-1 antigen levels were approximately 20 times higher in alveolar fluid compared with plasma in those who progressed to ARDS (Fig. 2), whereas the ratio of the total protein concentration in alveolar fluid to plasma was equal to 1.1±0.3. Furthermore, the ratio of alveolar to plasma PAI-1 antigen level was significantly lower in those with UAP compared with those who developed ARDS (5.7±4.8 vs. 19.5±9.2, respectively; p<0.001).
PAI-1 levels did not correlate with age, and there was no difference between men and women. However, there was a strong inverse correlation between PAI-1 levels and the days of unassisted ventilation (r2=0.37, p<0.001; Fig. 3). The Cook’s distance values ranged between 0 and 0.45, with a mean of 0.025 and a standard deviation of 0.065. The ROC curves for alveolar and plasma PAI-1 antigen levels are shown in Fig. 4. A cut-off level of alveolar PAI-1>1,518 ng/ml was found to be 82.4% (56.6–96.0%) sensitive and 97.1% (84.6%–99.5%) specific in predicting progression to ARDS. The AUCs for alveolar and plasma PAI-1 levels were 0.93±0.043 and 0.65±0.085, respectively (p<0.001), indicating a superior predictive accuracy of alveolar PAI-1 levels in predicting progression to ARDS following aspiration.
PAI-1 activity
The PAI-1 activity levels paralleled the PAI-1 antigen levels in both plasma and alveolar fluids. While the activity levels in plasma of PAI-1 levels were not different between those with UAP and those who progressed to ARDS (2.7±1.7 vs. 3.2±1.2, respectively; p=0.3), the activity of PAI-1 in alveolar fluid was significantly higher in those who developed ARDS compared with those with UAP (61.6±34.4 vs. 13.5±12.3, respectively; p<0.001).
Outcome
Four of the 51 subjects required tracheostomy. Nosocomial pneumonia complicated the course of six (35%) out of the 17 who developed ARDS, compared with three (9%) of 34 patients with UAP (p=0.05). Two patients with ARDS and one with UAP died during their hospitalization. Mortality was attributed to multiorgan failure in two of the ARDS patients and to pulmonary embolus in the patient with UAP.
Discussion
Intraalveolar and intravascular fibrin deposition is frequently found in the setting of ALI [17, 18]. The disturbance in the alveolar homeostasis has been attributed both to enhanced procoagulant and decreased alveolar fibrinolytic activities [19]. Similar changes were also reported in patients with severe pneumonia requiring mechanical ventilation [17, 20, 21], and these changes coincided with profoundly suppressed fibrinolytic activity in the alveolar space. However, none of these studies was able to identify an early marker for progression to ARDS.
Several lines of evidence have indicated that expression of alveolar PAI-1 is increased in tissues of endotoxemic animals [22]. Yet in some, PAI-1 levels were either undetectable or inconsistent with the underlying pathology [6]. These variations have been attributed either to differences in the timing of sample collection or to the dilution effect of BAL, which may render undetectable some factors that are present in very low concentrations. To overcome these limitations, we used a standard bronchoalveolar suction catheter wedged into the distal airway to sample the alveolar fluid. Our results pointed to a fivefold elevation of alveolar PAI-1 antigen levels in patients who progressed to ARDS following aspiration compared with those with UAP. Using similar techniques, Prabhakaran and colleagues [23] reported an elevenfold increase in mean alveolar PAI-1 antigen levels in 26 patients with ALI compared with 25 others with hydrostatic pulmonary edema. We speculate that the aspiration of gastric contents leads to inflammatory changes in the lung with local release of inflammatory cytokines and chemokines. A sharp rise in alveolar TNF-α following the intratracheal installation of gastric juice in swine but not after normal saline [24] corroborates this hypothesis. Because TNF-α has important regulatory function in the depression of fibrinolytic activity through induction of PAI-1 expression in the lung [25], our findings underline the pathophysiologic importance of PAI-1 in tissue injury postaspiration.
Although it is plausible that the changes in the PAI-1 levels observed in the pulmonary compartment were the result of leakage across the endothelial-alveolar barrier, the alveolar to plasma protein ratio remained constant despite the twentyfold increase in alveolar PAI-1 levels compared with plasma. This observation emphasizes the compartmentalized coagulation in the pathophysiology of ALI. The current study differs with the conclusions of an earlier report in which systemic activation of coagulation and impaired fibrinolysis were not found to play a major role in the outcomes of patients with ALI [26]. It should be noted, however, that the prior study in question did not examine intraalveolar coagulation, had different outcome measures, and did not emphasize the examination of samples collected soon after respiratory failure.
The strength of our study lies in the homogeneity of the study population. Previous investigations have included groups of patients with a myriad of disorders ranging from sepsis and pneumonia to trauma [23, 26]. In contrast, our study population was limited to overdose patients who had aspiration episodes that were witnessed. While PAI-1 elevations in the prior investigations may be considered an epiphenomenon, we have attempted to minimize this effect by excluding all patients with sepsis or suspected infectious pneumonitis, relying on a threshold of ≥100 CFU/ml on BAL quantitative culture.
Yet the differential rise of alveolar PAI-1 antigen levels among the few participants raises a series of questions that require further investigations: Is the expression of alveolar PAI-1 directly related to the acidity, volume, or composition of the aspirate? Is there any genetic predisposition for overexpressing PAI-1 following acute insult to the lung? Because it is unlikely that the answer to the first question would come from human studies, it is the recent finding of the genetic polymorphism of the PAI-1 gene that could hold promising clinical implications. In a contemporary investigation, Westendorp and coworkers [27] looked at whether genetic differences in the fibrinolytic system influenced the development of meningococcal septic shock. Those patients whose relatives were carriers of the 4G/4G genotype of the PAI-1 promoter gene had a sixfold higher risk of developing septic shock than meningococcal meningitis. Whether these observations could be extrapolated to ARDS victims of gastric aspiration is yet to be determined.
It should be noted that our study was not designed to independently study the effects of ventilator-induced lung injury on PAI-1 levels; we did not address whether ventilator-mediated effects on the inflammatory process may have an independent effect on PAI-1 levels or activity. Because we obtained no biopsy material, we were unable to determine the cellular source of PAI-1.
In summary, this study demonstrated that PAI-1 levels can be used as a valid biomarker of progression to ARDS in those patients with documented aspiration. The potential benefit of early detection of significant aspiration injury can be useful to target patients with aggressive therapy when such intervention is available.
References
Roy TM, Ossorio MA, Cipolla LM, Fields CL. Snider HL, Anderson WH (1989) Pulmonary complications after tricyclic antidepressant overdose. Chest 96:852–856
Olsson GL, Hallen B, Hambraeus-Jonzon K (1986) Aspiration during anaesthesia: a computer-aided study of 185,358 anaesthetics. Acta Anaesthesiol Scand 30:84–92
Marik PE (2001) Aspiration pneumonitis and aspiration pneumonia. N Engl J Med 344:665–671
Bynum L, Pierce A (1976) Pulmonary aspiration of gastric contents. Am Rev Respir Dis 114:1129–1136
Olman M, Mackman N, Gladson C, Moser K, Loskutoff DJ (1995) Changes in procoagulant and fibrinolytic gene expression during bleomycin induced lung injury in the mouse. J Clin Invest 96:1621–1630
Idell S, James KK, Levin EG, Schwartz BS, Manchanda N, Maunder RJ, Martin TR, McLarty J, Fair DS (1989) Local abnormalities in coagulation and fibrinolytic pathways predispose to alveolar fibrin deposition in the adult respiratory distress syndrome. J Clin Invest 84:695–705
Eitzman DT, McCoy RD, Zheng X, Fay WP, Shen T, Ginsburg D, Simon RH (1996) Bleomycin-induced pulmonary fibrosis in transgenic mice that either lack or overexpress the murine plasminogen activator-1 gene. J Clin Invest 97:232–237
Verghese GM, Ware LB, Matthay BA, and Matthay MA (1999) Alveolar epithelial fluid transport and the resolution of clinically severe hydrostatic pulmonary edema. J Appl Physiol 87:1301–1312
El-Solh AA, Pietrantoni C, Bhat A, Aquilina AT, Okada M, Grover V, Gifford N (2003) Microbiology of severe aspiration pneumonia in institutionalized elders. Am J Respir Crit Care Med 167:1650–1654
Smith PK, Krohn RI, Hermason GT, Mallia AK, Gartner FK, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchonimic acid. Anal Biochem 150:76–85
Murray JF, Matthay MA, Luce JM, Flick MR (1988) An expanded definition of the adult respiratory distress syndrome. Am Rev Respir Dis 138:720–723
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Bernard GR, Artigas A, Brigham K, Carlet J, Falke K, Hudson L, Lamy M, Le Gall JR, Morris A, Spragg R (1994) The American-European consensus conference on ARDS. Am J Respir Crit Care Med 149:818–824
Schoenfeld DA, Bernard GR (2002) Evaluation of ventilator-free days as an efficacy measure in clinical trials of treatments for acute respiratory distress syndrome. Crit Care Med 30:1772–1777
Chmielewska J, Wiman B (1986) Determination of tissue plasminogen activator and its “fast” inhibitor in plasma. Clin Chem 32:482–485
Kleinbaum DG, Kupper LL, Muller K (1998) Applied regression analysis and other multivariable methods, 3rd edn. Duxbury Press, Pacific Grove, CA, pp 228–237
Gunther A, Mosavi P, Heinemann S, Ruppert C, Muth H, Markart P, Grimminger F, Walmrath D, Temmesfeld-Wollbruck B, Seeger W (2000) Alveolar fibrin formation caused by enhanced procoagulant and depressed fibrinolytic capacities in severe pneumonia: comparison with the acute respiratory distress syndrome. Am J Respir Crit Care Med 161:454–462
Bertozzi P, Astedt B, Zenzius L, Lynch K, LeMaire F, Zapol W, Chapman HA Jr (1990) Depressed bronchoalveolar urokinase activity in patients with adult respiratory distress syndrome. N Engl J Med 322:890–897
Idell S, Gonzalez K, Bradford H, Macarthur CK, Fein AM, Maunder RJ, Garcia JG, Griffith DE, Weiland J, Martin TR (1987) Procoagulant activity in bronchoalveolar lavage in the adult respiratory distress syndrome. Contribution of tissue factor associated with Factor VII. Am Rev Respir Dis 136:1466-1474
El Solh AA, Okada M, Pietrantoni C, Aquilina A, Berbary E (2004) Procoagulant and fibrinolytic activity in ventilator-associated pneumonia: impact of inadequate antimicrobial therapy. Intensive Care Med 30:1914–1920
Schultz MJ, Millo J, Levi M, Hack CE, Weverling GJ, Garrard CS, van der Poll T (2004) Local activation of coagulation and inhibition of fibrinolysis in the lung during ventilator associated pneumonia. Thorax 59:130–135
Quax PH, van den Hoogen CM, Verheijen JH, Padro T, Zeheb R, Gelehrter TD, van Berkel TJ, Kuiper J, Emeis JJ (1990) Endotoxin induction of plasminogen activator and plasminogen activator inhibitor type 1 mRNA in rat tissues in vivo. J Biol Chem 265:15560–15563
Prabhakaran P, Ware LB, White KE, Cross MT, Matthay MA, Olman MA (2003) Elevated levels of plasminogen activator inhibitor-1 in pulmonary edema are associated with mortality in acute lung injury. Am J Physiol Lung Cell Mol Physiol 285:L20–L28
Chandrasekhar A, Timberlake G, Prabhakar G, Barringer L (2001) Elevation of alveolar cytokine response to aspiration of gastric contents. J Appl Res Clin Exp Therap 1(1):http://jrnlappliedresearch.com/articles/Vol1Iss1/Chendrasekhar.htm
Fan J, Kapus A, Li H, Rizoli S, Marshall J, Rotstein O (2000) Priming for enhanced alveolar fibrin deposition following hemorrhagic shock: role of tumor necrosis factor. Am J Respir Cell Mol Biol 22:412–421
Groeneveld AN, Kindt I, Raijmakers PG, Hack CE, Thijs LG (1997) Systemic coagulation and fibrinolysis in patients with or at risk for the adult respiratory distress syndrome. Thromb Haemost 78:1444–1449
Westendorp RG, Hottenga JJ, Slagboom PE (1999) Variation in plasminogen-activator-inhibitor-1 gene and risk of meningococcal septic shock. Lancet 354:561–563
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
El Solh, A.A., Bhora, M., Pineda, L. et al. Alveolar plasminogen activator inhibitor-1 predicts ARDS in aspiration pneumonitis. Intensive Care Med 32, 110–115 (2006). https://doi.org/10.1007/s00134-005-2847-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-005-2847-2