Abstract
Background
Recent studies suggest that chronic inflammation-associated cancer is relevant to microbiome. Esophageal adenocarcinoma arises from an inflammatory condition called Barrett’s esophagus, which is caused by gastroesophageal reflux. We hypothesized that esophageal microbiome plays a role in carcinogenesis of esophageal adenocarcinoma.
Aim
We investigated whether alteration of microbiome using antibiotics affects the development of esophageal adenocarcinoma in a rat model.
Methods
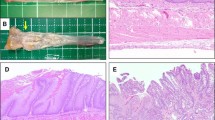
Seven-week-old male Wistar rats which had undergone esophagojejunostomy were divided into control (n = 21) and antibiotic groups (n = 22) at 21 weeks after surgery. Control animals were given drinking water, while the other group was given penicillin G and streptomycin in drinking water until rats were killed at 40 weeks after operation. Incidence rates of Barrett’s esophagus and adenocarcinoma in each group were evaluated by histological analysis. DNA was extracted from a portion of the distal esophagus, and the microbiome was investigated using terminal restriction fragment length polymorphism (T-RFLP) analysis.
Results
All rats in both groups developed Barrett’s esophagus. Incidence of esophageal adenocarcinoma was similar between both groups with a trend to reduced incidence in the antibiotics group (89 % in the control group, 71 % in the antibiotics group, P = 0.365). T-RFLP analysis showed that esophageal microbiome was different between two groups such as the proportion of Lactobacillales was lower in the antibiotics group and Clostridium cluster XIVa and XVIII was higher in the antibiotics group.
Conclusions
Alteration of microbiome does not affect the incidence of esophageal adenocarcinoma. Microbiome may not contribute to the development of esophageal adenocarcinoma.






Similar content being viewed by others
References
Rubenstein JH, Shaheen NJ. Epidemiology, diagnosis, and management of esophageal adenocarcinoma. Gastroenterology. 2015;149:302.e1–317.e1.
Spechler SJ. Barrett esophagus and risk of esophageal cancer: a clinical review. JAMA. 2013;310:627–636.
Paulson TG, Reid BJ. Focus on Barrett’s esophagus and esophageal adenocarcinoma. Cancer Cell. 2004;6:11–16.
Elinav E, Nowarski R, Thaiss CA, Hu B, Jin C, Flavell RA. Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms. Nat Rev Cancer. 2013;13:759–771.
Arthur JC, Perez-Chanona E, Muhlbauer M, et al. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science. 2012;338:120–123.
Honda K, Littman DR. The microbiome in infectious disease and inflammation. Annu Rev Immunol. 2012;30:759–795.
Guo W, Ding J, Huang Q, Jerrells T, Deitch EA. Alterations in intestinal bacterial flora modulate the systemic cytokine response to hemorrhagic shock. Am J Physiol. 1995;269:G827–G832.
Yoshimoto S, Loo TM, Atarashi K, et al. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature. 2013;499:97–101.
Miwa K, Sahara H, Segawa M, et al. Reflux of duodenal or gastro-duodenal contents induces esophageal carcinoma in rats. Int J Cancer. 1996;67:269–274.
Miyashita T, Miwa K, Fujimura T, et al. The severity of duodeno-esophageal reflux influences the development of different histological types of esophageal cancer in a rat model. Int J Cancer. 2013;132:1496–1504.
Shen XJ, Rawls JF, Randall T, et al. Molecular characterization of mucosal adherent bacteria and associations with colorectal adenomas. Gut Microbes. 2010;1:138–147.
Takahashi S, Tomita J, Nishioka K, Hisada T, Nishijima M. Development of a prokaryotic universal primer for simultaneous analysis of bacteria and archaea using next-generation sequencing. PLoS One. 2014;9:e105592.
Muyzer G, de Waal EC, Uitterlinden AG. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol. 1993;59:695–700.
Nagashima K, Hisada T, Sato M, Mochizuki J. Application of new primer-enzyme combinations to terminal restriction fragment length polymorphism profiling of bacterial populations in human feces. Appl Environ Microbiol. 2003;69:1251–1262.
Pei Z, Bini EJ, Yang L, Zhou M, Francois F, Blaser MJ. Bacterial biota in the human distal esophagus. Proc Natl Acad Sci USA. 2004;101:4250–4255.
Pei Z, Yang L, Peek RM Jr, Levine SM, Pride DT, Blaser MJ. Bacterial biota in reflux esophagitis and Barrett’s esophagus. World J Gastroenterol. 2005;11:7277–7283.
Yang L, Lu X, Nossa CW, Francois F, Peek RM, Pei Z. Inflammation and intestinal metaplasia of the distal esophagus are associated with alterations in the microbiome. Gastroenterology. 2009;137:588–597.
Liu N, Ando T, Ishiguro K, et al. Characterization of bacterial biota in the distal esophagus of Japanese patients with reflux esophagitis and Barrett’s esophagus. BMC Infect Dis. 2013;13:130.
Fan YP, Chakder S, Gao F, Rattan S. Inducible and neuronal nitric oxide synthase involvement in lipopolysaccharide-induced sphincteric dysfunction. Am J Physiol Gastrointest Liver Physiol. 2001;280:G32–G42.
Yang L, Francois F, Pei Z. Molecular pathways: pathogenesis and clinical implications of microbiome alteration in esophagitis and Barrett esophagus. Clin Cancer Res. 2012;18:2138–2144.
Colleypriest BJ, Ward SG, Tosh D. How does inflammation cause Barrett’s metaplasia? Curr Opin Pharmacol. 2009;9:721–726.
Lee JS, Oh TY, Ahn BO, et al. Involvement of oxidative stress in experimentally induced reflux esophagitis and Barrett’s esophagus: clue for the chemoprevention of esophageal carcinoma by antioxidants. Mutat Res. 2001;480–481:189–200.
Konturek PC, Nikiforuk A, Kania J, Raithel M, Hahn EG, Muhldorfer S. Activation of NFkappaB represents the central event in the neoplastic progression associated with Barrett’s esophagus: a possible link to the inflammation and overexpression of COX-2, PPARγ and growth factors. Dig Dis Sci. 2004;49:1075–1083.
O’Riordan JM, Abdel-latif MM, Ravi N, et al. Proinflammatory cytokine and nuclear factor kappa-B expression along the inflammation–metaplasia–dysplasia–adenocarcinoma sequence in the esophagus. Am J Gastroenterol. 2005;100:1257–1264.
Abdel-Latif MM, Kelleher D, Reynolds JV. Potential role of NF-kappaB in esophageal adenocarcinoma: as an emerging molecular target. J Surg Res. 2009;153:172–180.
Kohata Y, Nakahara K, Tanigawa T, et al. Rebamipide alters the esophageal microbiome and reduces the incidence of Barrett’s esophagus in a rat model. Dig Dis Sci. 2015;60:2654–2661.
Dapito DH, Mencin A, Gwak GY, et al. Promotion of hepatocellular carcinoma by the intestinal microbiota and TLR4. Cancer Cell. 2012;21:504–516.
Polk DB, Peek RM Jr. Helicobacter pylori: gastric cancer and beyond. Nat Rev Cancer. 2010;10:403–414.
Prorok-Hamon M, Friswell MK, Alswied A, et al. Colonic mucosa-associated diffusely adherent afaC+ Escherichia coli expressing lpfA and pks are increased in inflammatory bowel disease and colon cancer. Gut. 2014;63:761–770.
Kostic AD, Gevers D, Pedamallu CS, et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res. 2012;22:292–298.
Blackett KL, Siddhi SS, Cleary S, et al. Oesophageal bacterial biofilm changes in gastro-oesophageal reflux disease, Barrett’s and oesophageal carcinoma: association or causality? Aliment Pharmacol Ther. 2013;37:1084–1092.
Zaidi AH, Kelly LA, Kreft RE, et al. Associations of microbiota and toll-like receptor signaling pathway in esophageal adenocarcinoma. BMC Cancer. 2015;16:52.
Garcia JM, Splenser AE, Kramer J, et al. Circulating inflammatory cytokines and adipokines are associated with increased risk of Barrett’s esophagus: a case–control study. Clin Gastroenterol Hepatol. 2014;12:229.e3–238.e3.
Siahpush SH, Vaughan TL, Lampe JN, et al. Longitudinal study of insulin-like growth factor, insulin-like growth factor binding protein-3, and their polymorphisms: risk of neoplastic progression in Barrett’s esophagus. Cancer Epidemiol Biomark Prev. 2007;16:2387–2395.
Cho I, Yamanishi S, Cox L, et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature. 2012;488:621–626.
Funding
This study was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology in Japan (No. 15K08957).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Arakawa received lecture fees from Otsuka and Eisai and research grants from Otsuka, Eisai, Astellas, Abbott Japan, Takeda, Dainippon Sumitomo, and Daiichi Sankyo. Dr. Fujiwara received lecture fees from Takeda and research grants from Ono. The remaining authors have no conflicts to disclose.
Rights and permissions
About this article
Cite this article
Sawada, A., Fujiwara, Y., Nagami, Y. et al. Alteration of Esophageal Microbiome by Antibiotic Treatment Does Not Affect Incidence of Rat Esophageal Adenocarcinoma. Dig Dis Sci 61, 3161–3168 (2016). https://doi.org/10.1007/s10620-016-4263-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4263-6