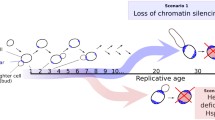
Abstract
DNA damage is an important factor in aging in all eukaryotes. Although connections between DNA damage and aging have been extensively investigated in complex organisms, only a relatively few studies have investigated DNA damage as an aging factor in the model organism S. cerevisiae. Several of these studies point to DNA replication stress as a cause of age-dependent DNA damage in the replicative model of aging, which measures how many times budding yeast cells divide before they senesce and die. Even fewer studies have investigated how DNA damage contributes to aging in the chronological aging model, which measures how long cells in stationary phase cultures retain reproductive capacity. DNA replication stress also has been implicated as a factor in chronological aging . Since cells in stationary phase are generally considered to be “post-mitotic” and to reside in a quiescent G0/G1 state, the notion that defects in DNA replication might contribute to chronological aging appears to be somewhat paradoxical. However, the results of recent studies suggest that a significant fraction of cells in stationary phase cultures are not quiescent, especially in experiments that employ defined medium, which is frequently employed to assess chronological lifespan. Most cells that fail to achieve quiescence remain in a viable, but non-dividing state until they eventually die, similar to the senescent state in mammalian cells. In this chapter we discuss the role of DNA damage and DNA replication stress in both replicative and chronological aging in S. cerevisiae. We also discuss the relevance of these findings to the emerging view that DNA damage and DNA replication stress are important components of the senescent state that occurs at early stages of cancer.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
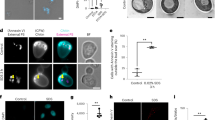
Similar content being viewed by others
References
Aguilaniu H, Gustafsson L, Rigoulet M, Nystrom T (2003) Asymmetric inheritance of oxidatively damaged proteins during cytokinesis. Science 299:1751–1753
Allen C, Buttner S, Aragon AD, Thomas JA, Meirelles O, Jaetao JE, Benn D, Ruby SW, Veenhuis M, Madeo F et al (2006) Isolation of quiescent and nonquiescent cells from yeast stationary-phase cultures. J Cell Biol 174:89–100
Aragon AD, Rodriguez AL, Meirelles O, Roy S, Davidson GS, Tapia PH, Allen C, Joe R, Benn D, Werner-Washburne M (2008) Characterization of differentiated quiescent and nonquiescent cells in yeast stationary-phase cultures. Mol Biol Cell 19:1271–1280
Barros MH, Bandy B, Tahara EB, Kowaltowski AJ (2004) Higher respiratory activity decreases mitochondrial reactive oxygen release and increases life span in Saccharomyces cerevisiae. J Biol Chem 279:49883–49888
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K, Guldberg P, Sehested M, Nesland JM, Lukas C et al (2005) DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434:864–870
Bester AC, Roniger M, Oren YS, Im MM, Sarni D, Chaoat M, Bensimon A, Zamir G, Shewach DS, Kerem B (2011) Nucleotide deficiency promotes genomic instability in early stages of cancer development. Cell 145:435–446
Bhattacharyya S, Rolfsmeier ML, Dixon MJ, Wagoner K, Lahue RS (2002) Identification of RTG2 as a modifier gene for CTG*CAG repeat instability in Saccharomyces cerevisiae. Genetics 162:579–589
Blagosklonny MV (2011) Cell cycle arrest is not senescence. Aging (Albany NY) 3:94–101
Borghouts C, Benguria A, Wawryn J, Jazwinski SM (2004) Rtg2 protein links metabolism and genome stability in yeast longevity. Genetics 166:765–777
Burhans WC, Weinberger M (2007) DNA replication stress, genome instability and aging. Nucleic Acids Res 35:7545–7556
Burhans WC, Weinberger M (2010) Histone genes, DNA replication, apoptosis and aging: what are the connections? Cell Cycle 9:4047–4048
Burtner CR, Murakami CJ, Kennedy BK, Kaeberlein M (2009) A molecular mechanism of chronological aging in yeast. Cell Cycle 8:1256–1270
Casper AM, Mieczkowski PA, Gawel M, Petes TD (2008) Low levels of DNA polymerase alpha induce mitotic and meiotic instability in the ribosomal DNA gene cluster of Saccharomyces cerevisiae. PLoS Genet 4:e1000105
Chaudhuri L, Sarsour EH, Goswami PC (2010) 2-(4-Chlorophenyl)benzo-1,4-quinone induced ROS-signaling inhibits proliferation in human non-malignant prostate epithelial cells. Environ Int 36:924–930
Chen JH, Hales CN, Ozanne SE (2007) DNA damage, cellular senescence and organismal ageing: causal or correlative? Nucleic Acids Res 35:7417–7428
Dang W, Steffen KK, Perry R, Dorsey JA, Johnson FB, Shilatifard A, Kaeberlein M, Kennedy BK, Berger SL (2009) Histone H4 lysine 16 acetylation regulates cellular lifespan. Nature 459:802–807
Desany BA, Alcasabas AA, Bachant JB, Elledge SJ (1998) Recovery from DNA replicational stress is the essential function of the S-phase checkpoint pathway. Genes Dev 12:2956–2970
Di Micco R, Fumagalli M, Cicalese A, Piccinin S, Gasparini P, Luise C, Schurra C, Garre M, Nuciforo PG, Bensimon A et al (2006) Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 444:638–642
Fabrizio P, Battistella L, Vardavas R, Gattazzo C, Liou LL, Diaspro A, Dossen JW, Gralla EB, Longo VD (2004) Superoxide is a mediator of an altruistic aging program in Saccharomyces cerevisiae. J Cell Biol 166:1055–1067
Fabrizio P, Longo VD (2008) Chronological aging-induced apoptosis in yeast. Biochim Biophys Acta 1783:1280–1285
Fasullo M, Tsaponina O, Sun M, Chabes A (2010) Elevated dNTP levels suppress hyper-recombination in Saccharomyces cerevisiae S-phase checkpoint mutants. Nucleic Acids Res 38:1195–1203
Freitas AA, de Magalhaes JP (2011) A review and appraisal of the DNA damage theory of ageing. Mutat Res 728:12–22
Ganley AR, Ide S, Saka K, Kobayashi T (2009) The effect of replication initiation on gene amplification in the rDNA and its relationship to aging. Mol Cell 35:683–693
Gasch AP, Spellman PT, Kao CM, Carmel-Harel O, Eisen MB, Storz G, Botstein D, Brown PO (2000) Genomic expression programs in the response of yeast cells to environmental changes. Mol Biol Cell 11:4241–4257
Granot D, Levine A, Dor-Hefetz E (2003) Sugar-induced apoptosis in yeast cells. FEMS Yeast Res 4:7–13
Guarente L (2000) Sir2 links chromatin silencing, metabolism, and aging. Genes Dev 14:1021–1026
Hakansson P, Hofer A, Thelander L (2006) Regulation of mammalian ribonucleotide reduction and dNTP pools after DNA damage and in resting cells. J Biol Chem 281:7834–7841
Hartman J Lt. (2007) Buffering of deoxyribonucleotide pool homeostasis by threonine metabolism. Proc Natl Acad Sci USA 104:11700–11705
Heeren G, Rinnerthaler M, Laun P, von Seyerl P, Kossler S, Klinger H, Hager M, Bogengruber E, Jarolim S, Simon-Nobbe B et al (2009) The mitochondrial ribosomal protein of the large subunit, Afo1p, determines cellular longevity through mitochondrial back-signaling via TOR1. Aging (Albany NY) 1:622–636
Heo SJ, Tatebayashi K, Ohsugi I, Shimamoto A, Furuichi Y, Ikeda H (1999) Bloom’s syndrome gene suppresses premature ageing caused by Sgs1 deficiency in yeast. Genes Cells 4:619–625
Herker E, Jungwirth H, Lehmann KA, Maldener C, Frohlich KU, Wissing S, Buttner S, Fehr M, Sigrist S, Madeo F (2004) Chronological aging leads to apoptosis in yeast. J Cell Biol 164:501–507
Herrero AB, Moreno S (2011) Lsm1 promotes genomic stability by controlling histone mRNA decay. EMBO J 30:2008–2018
Hoopes LL, Budd M, Choe W, Weitao T, Campbell JL (2002) Mutations in DNA replication genes reduce yeast life span. Mol Cell Biol 22:4136–4146
Jarolim S, Millen J, Heeren G, Laun P, Goldfarb DS, Breitenbach M (2004) A novel assay for replicative lifespan in Saccharomyces cerevisiae. FEMS Yeast Res 5:169–177
Jazwinski SM (2005) Rtg2 protein: at the nexus of yeast longevity and aging. FEMS Yeast Res 5:1253–1259
Kaeberlein M, Powers RW 3rd, Steffen KK, Westman EA, Hu D, Dang N, Kerr EO, Kirkland KT, Fields S, Kennedy BK (2005) Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science 310:1193–1196
Laschober GT, Ruli D, Hofer E, Muck C, Carmona-Gutierrez D, Ring J, Hutter E, Ruckenstuhl C, Micutkova L, Brunauer R et al (2010) Identification of evolutionarily conserved genetic regulators of cellular aging. Aging Cell 9:1084–1097
Laun P, Ramachandran L, Jarolim S, Herker E, Liang P, Wang J, Weinberger M, Burhans DT, Suter B, Madeo F et al (2005) A comparison of the aging and apoptotic transcriptome of Saccharomyces cerevisiae. FEMS Yeast Res 5:1261–1272
Lengronne A, Schwob E (2002) The yeast CDK inhibitor Sic1 prevents genomic instability by promoting replication origin licensing in late G(1). Mol Cell 9:1067–1078
Lesur I, Campbell JL (2004) The transcriptome of prematurely aging yeast cells is similar to that of telomerase-deficient cells. Mol Biol Cell 15:1297–1312
Lindstrom DL, Gottschling DE (2009) The mother enrichment program: a genetic system for facile replicative life span analysis in Saccharomyces cerevisiae. Genetics 183:413–422, 411SI–413SI
Lindstrom DL, Leverich CK, Henderson KA, Gottschling DE (2011) Replicative age induces mitotic recombination in the ribosomal RNA gene cluster of Saccharomyces cerevisiae. PLoS Genet 7:e1002015
Lopez Castel A, Cleary JD, Pearson CE (2010) Repeat instability as the basis for human diseases and as a potential target for therapy. Nat Rev Mol Cell Biol 11:165–170
Luo J, Solimini NL, Elledge SJ (2009) Principles of cancer therapy: oncogene and non-oncogene addiction. Cell 136:823–837
Madia F, Gattazzo C, Wei M, Fabrizio P, Burhans WC, Weinberger M, Galbani A, Smith JR, Nguyen C, Huey S et al (2008) Longevity mutation in SCH9 prevents recombination errors and premature genomic instability in a Werner/Bloom model system. J Cell Biol 180:67–81
Madia F, Wei M, Yuan V, Hu J, Gattazzo C, Pham P, Goodman MF, Longo VD (2009) Oncogene homologue Sch9 promotes age-dependent mutations by a superoxide and Rev1/Polzeta-dependent mechanism. J Cell Biol 186:509–523
McMurray MA, Gottschling DE (2003) An age-induced switch to a hyper-recombinational state. Science 301:1908–1911
McVey M, Kaeberlein M, Tissenbaum HA, Guarente L (2001) The short life span of Saccharomyces cerevisiae sgs1 and srs2 mutants is a composite of normal aging processes and mitotic arrest due to defective recombination. Genetics 157:1531–1542
Merker RJ, Klein HL (2002) hpr1Delta affects ribosomal DNA recombination and cell life span in Saccharomyces cerevisiae. Mol Cell Biol 22:421–429
Mesquita A, Weinberger M, Silva A, Sampaio-Marques B, Almeida B, Leao C, Costa V, Rodrigues F, Burhans WC, Ludovico P (2010) Caloric restriction or catalase inactivation extends yeast chronological lifespan by inducing H2O2 and superoxide dismutase activity. Proc Natl Acad Sci USA 107:15123–15128
Mortimer RK, Johnston JR (1959) Life span of individual yeast cells. Nature 183:1751–1752
Negrini S, Gorgoulis VG, Halazonetis TD (2010) Genomic instability–an evolving hallmark of cancer. Nat Rev Mol Cell Biol 11:220–228
Palermo V, Cundari E, Mangiapelo E, Falcone C, Mazzoni C (2010) Yeast lsm pro-apoptotic mutants show defects in S-phase entry and progression. Cell Cycle 9:3991–3996
Pasero P, Bensimon A, Schwob E (2002) Single-molecule analysis reveals clustering and epigenetic regulation of replication origins at the yeast rDNA locus. Genes Dev 16:2479–2484
Pichova A, Vondrakova D, Breitenbach M (1997) Mutants in the Saccharomyces cerevisiae RAS2 gene influence life span, cytoskeleton, and regulation of mitosis. Can J Microbiol 43:774–781
Powers RW 3rd, Kaeberlein M, Caldwell SD, Kennedy BK, Fields S (2006) Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes Dev 20:174–184
Pruitt SC, Bailey KJ, Freeland A (2007) Reduced Mcm2 expression results in severe stem/progenitor cell deficiency and cancer. Stem Cells 25:3121–3132
Qin H, Lu M, Goldfarb DS (2008) Genomic instability is associated with natural life span variation in Saccharomyces cerevisiae. PLoS One 3:e2670
Ramachandran L, Burhans DT, Laun P, Wang J, Liang P, Weinberger M, Wissing S, Jarolim S, Suter B, Madeo F et al (2006) Evidence for ORC-dependent repression of budding yeast genes induced by starvation and other stresses. FEMS Yeast Res 6:763–776
Riesen M, Morgan A (2009) Calorie restriction reduces rDNA recombination independently of rDNA silencing. Aging Cell 8:624–632
Ringvoll J, Uldal L, Roed MA, Reite K, Baynton K, Klungland A, Eide L (2007) Mutations in the RAD27 and SGS1 genes differentially affect the chronological and replicative lifespan of yeast cells growing on glucose and glycerol. FEMS Yeast Res 7:848–859
Rodier F, Campisi J (2011) Four faces of cellular senescence. J Cell Biol 192:547–556
Rowe LA, Degtyareva N, Doetsch PW (2008) DNA damage-induced reactive oxygen species (ROS) stress response in Saccharomyces cerevisiae. Free Radic Biol Med 45:1167–1177
Salmon TB, Evert BA, Song B, Doetsch PW (2004) Biological consequences of oxidative stress-induced DNA damage in Saccharomyces cerevisiae. Nucleic Acids Res 32:3712–3723
Sarsour EH, Venkataraman S, Kalen AL, Oberley LW, Goswami PC (2008) Manganese superoxide dismutase activity regulates transitions between quiescent and proliferative growth. Aging Cell 7:405–417
Sinclair DA, Guarente L (1997) Extrachromosomal rDNA circles – a cause of aging in yeast. Cell 91:1033–1042
Singh DK, Ahn B, Bohr VA (2009) Roles of RECQ helicases in recombination based DNA repair, genomic stability and aging. Biogerontology 10:235–252
Smith DL Jr, Li C, Matecic M, Maqani N, Bryk M, Smith JS (2009) Calorie restriction effects on silencing and recombination at the yeast rDNA. Aging Cell 8:633–642
Veatch JR, McMurray MA, Nelson ZW, Gottschling DE (2009) Mitochondrial dysfunction leads to nuclear genome instability via an iron-sulfur cluster defect. Cell 137:1247–1258
Wang Y, Pierce M, Schneper L, Guldal CG, Zhang X, Tavazoie S, Broach JR (2004) Ras and Gpa2 mediate one branch of a redundant glucose signaling pathway in yeast. PLoS Biol 2:E128
Weinberger M, Feng L, Paul A, Smith DL, Hontz RD, Smith JS, Vujcic M, Singh KK, Huberman J, Burhans WC (2007) DNA replication stress is a determinant of chronological lifespan in budding yeast. PLOS One 2:e748
Weinberger M, Mesquita A, Caroll T, Marks L, Yang H, Zhang Z, Ludovico P, Burhans WC (2010) Growth signaling promotes chronological aging in budding yeast by inducing superoxide anions that inhibit quiescence. Aging (Albany NY) 2:709–726
Weitao T, Budd M, Campbell JL (2003a) Evidence that yeast SGS1, DNA2, SRS2, and FOB1 interact to maintain rDNA stability. Mutat Res 532:157–172
Weitao T, Budd M, Hoopes LL, Campbell JL (2003b) Dna2 helicase/nuclease causes replicative fork stalling and double-strand breaks in the ribosomal DNA of Saccharomyces cerevisiae. J Biol Chem 278:22513–22522
Zanetti M, Zwacka R, Engelhardt J, Katusic Z, O’Brien T (2001) Superoxide anions and endothelial cell proliferation in normoglycemia and hyperglycemia. Arterioscler Thromb Vasc Biol 21:195–200
Zinzalla V, Graziola M, Mastriani A, Vanoni M, Alberghina L (2007) Rapamycin-mediated G1 arrest involves regulation of the Cdk inhibitor Sic1 in Saccharomyces cerevisiae. Mol Microbiol 63:1482–1494
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Burhans, W.C., Weinberger, M. (2011). DNA Damage and DNA Replication Stress in Yeast Models of Aging. In: Breitenbach, M., Jazwinski, S., Laun, P. (eds) Aging Research in Yeast. Subcellular Biochemistry, vol 57. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-2561-4_9
Download citation
DOI: https://doi.org/10.1007/978-94-007-2561-4_9
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-2560-7
Online ISBN: 978-94-007-2561-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)