Abstract
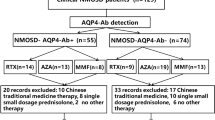
Neuromyelitis optica spectrum disorder (NMOSD) often follows a relapsing course. As disability in NMOSD is attack-related, effective treatments are needed. We aimed to compare the efficacy of azathioprine (AZA) and rituximab (RIT) as maintenance therapy in NMOSD patients. An open, randomized clinical trial was conducted during September 2015 to December 2016, in Isfahan, Iran. Initially, 100 NMOSD patients were approached, 86 entered the study, and 68 cases completed the trial. All patients had a relapsing–remitting course with expanded disability extended scale (EDSS) ≤7 (median 2.75, range = 0–7). Patients were randomized into two groups, which did not differ according to age, gender distribution, and disease duration. In the AZA group, 35 patients [20 aquaporin-4 (AQP4)-IgG positive] were started on 50 mg/day oral AZA and increased to 2–3 mg/kg/day (with oral prednisolone as adjunctive therapy). In the RIT group, 33 patients (13 aquaporin-4-IgG positive) received 1 g intravenous rituximab and repeated 2 weeks later and then every 6 months. Annualized relapse rate (ARR) was measured as the primary outcome and EDSS as the secondary outcome after 12 months of intervention. The mean ARR [standard deviation (SD)] in the AZA group decreased from 1 (0.38) to 0.51 (0.55) (P value <0.001) and in the RIT group decreased from 1.30 (0.68) to 0.21 (0.42) (P value <0.001). ARR after intervention minus ARR before intervention [mean (SD)] was 1.09 (0.72) in RIT group and 0.49 (0.59) in AZA group (P value <0.001). EDSS after intervention minus EDSS before intervention [mean (SD)] was 0.98 (1.14) in RIT group and 0.44 (0.54) in AZA group (P value <0.001). Nineteen patients (54.3%) in AZA group and 26 patients (78.8%) in RIT group became relapse-free after intervention (P value = 0.033). AZA and RIT can both effectively decrease ARR and EDSS in NMOSD patients. RIT was significantly more effective than AZA treatment.
Trial Registration Name of registry: clinicaltrials.gov; ID: NCT03002038; URL: https://clinicaltrials.gov/ct2/show/NCT03002038.

Similar content being viewed by others
References
Bedi GS, Brown AD, Delgado SR, Usmani N, Lam BL, Sheremata WA (2011) Impact of rituximab on relapse rate and disability in neuromyelitis optica. Mult Scler J 17:1225–1230
Bichuetti DB, de Oliveira EML, Oliveira DM, de Souza NA, Gabbai AA (2010) Neuromyelitis optica treatment: analysis of 36 patients. Arch Neurol 67:1131–1136
Carreón-Guarnizo E, Hernández-Clares R, Palazón-Cabanes B, Carrasco-Torres R, Salgado G, Jimenez-Veiga J, León-Hernández A, Martín-Fernández JJ, Meca-Lallana J (2015) Safety of rituximab as first-line therapy in neuromyelitis optica (NMO) and NMO-spectrum disorders: five years of experience (P5. 264). Neurology 84(P5):264
Costanzi C, Matiello M, Lucchinetti CF, Weinshenker BG, Pittock SJ, Mandrekar J, Thapa P, McKeon A (2011) Azathioprine tolerability, efficacy, and predictors of benefit in neuromyelitis optica. Neurology 77:659–666
Cree B, Lamb S, Morgan K, Chen A, Waubant E, Genain C (2005) An open label study of the effects of rituximab in neuromyelitis optica. Neurology 64:1270–1272
Elsone L, Kitley J, Luppe S, Lythgoe D, Mutch K, Jacob S, Brown R, Moss K, McNeillis B, Goh YY (2014) Long-term efficacy, tolerability and retention rate of azathioprine in 103 aquaporin-4 antibody-positive neuromyelitis optica spectrum disorder patients: a multicentre retrospective observational study from the UK. Mult Scler J 20:1533–1540
Hinson S, Pittock SJ, Lucchinetti CF, Roemer S, Fryer J, Kryzer T, Lennon VA (2007) Pathogenic potential of IgG binding to water channel extracellular domain in neuromyelitis optica. Neurology 69:2221–2231
Jeong IH, Park B, Kim S-H, Hyun J-W, Joo J, Kim HJ (2015) Comparative analysis of treatment outcomes in patients with neuromyelitis optica spectrum disorder using multifaceted endpoints. Mult Scler J 22:329–339
Kanesalingam R, Chia Y, Eow G, Easaw P, Rafia M (2013) The spectrum of neuromyelitis optica. J Neurol Sci 333:e363
Kim S-H, Huh S-Y, Lee SJ, Joung A, Kim HJ (2013) A 5-year follow-up of rituximab treatment in patients with neuromyelitis optica spectrum disorder. JAMA Neurol 70:1110–1117
Kim S-H, Kim W, Li XF, Jung I-J, Kim HJ (2011) Repeated treatment with rituximab based on the assessment of peripheral circulating memory B cells in patients with relapsing neuromyelitis optica over 2 years. Arch Neurol 68:1412–1420
Lucchinetti CF, Mandler RN, McGavern D, Bruck W, Gleich G, Ransohoff RM, Trebst C, Weinshenker B, Wingerchuk D, Parisi JE (2002) A role for humoral mechanisms in the pathogenesis of Devic’s neuromyelitis optica. Brain 125:1450–1461
Mandler RN, Ahmed W, Dencoff JE (1998) Devic’s neuromyelitis optica: a prospective study of seven patients treated with prednisone and azathioprine. Neurology 51:1219–1220
Mealy MA, Wingerchuk DM, Palace J, Greenberg BM, Levy M (2014) Comparison of relapse and treatment failure rates among patients with neuromyelitis optica: multicenter study of treatment efficacy. JAMA Neurol 71:324–330
Pittock SJ, Weinshenker BG, Lucchinetti CF, Wingerchuk DM, Corboy JR, Lennon VA (2006) Neuromyelitis optica brain lesions localized at sites of high aquaporin 4 expression. Arch Neurol 63:964–968
Qiu W, Kermode AG, Li R, Dai Y, Wang Y, Wang J, Zhong X, Li C, Lu Z, Hu X (2015) Azathioprine plus corticosteroid treatment in Chinese patients with neuromyelitis optica. J Clin Neurosci 22:1178–1182
Radaelli M, Moiola L, Sangalli F, Esposito F, Barcella V, Ferrè L, Rodegher M, Colombo B, Fazio R, Martinelli V (2015) Neuromyelitis optica spectrum disorders: long-term safety and efficacy of rituximab in Caucasian patients. Mult Scler J 22:511–519
Sahraian M, Moinfar Z, Khorramnia S, Mohammad Ebrahim M (2010) Relapsing neuromyelitis optica: demographic and clinical features in Iranian patients. Eur J Neurol 17:794–799
Sand IK (2016) Neuromyelitis optica spectrum disorders. CONTINUUM: lifelong learning. Neurology 22:864–896
Sato DK, Lana-Peixoto MA, Fujihara K, Seze J (2013) Clinical spectrum and treatment of neuromyelitis optica spectrum disorders: evolution and current status. Brain Pathol 23:647–660
Trebst C, Jarius S, Berthele A, Paul F, Schippling S, Wildemann B, Borisow N, Kleiter I, Aktas O, Kümpfel T (2014) Update on the diagnosis and treatment of neuromyelitis optica: recommendations of the Neuromyelitis Optica Study Group (NEMOS). J Neurol 261:1–16
Waters P, Jarius S, Littleton E, Leite MI, Jacob S, Gray B, Geraldes R, Vale T, Jacob A, Palace J (2008) Aquaporin-4 antibodies in neuromyelitis optica and longitudinally extensive transverse myelitis. Arch Neurol 65:913–919
Wingerchuk DM, Banwell B, Bennett JL, Cabre P, Carroll W, Chitnis T, De Seze J, Fujihara K, Greenberg B, Jacob A (2015) International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85:177–189
Zéphir H, Bernard-Valnet R, Lebrun C, Outteryck O, Audoin B, Bourre B, Pittion S, Wiertlewski S, Ouallet JC, Neau J-P (2015) Rituximab as first-line therapy in neuromyelitis optica: efficiency and tolerability. J Neurol 262:2329–2335
Acknowledgements
We want to thank vice-chancellor for research and technology of Isfahan University of Medical Sciences for funding and supporting this project (Grant number: 395275).
Author information
Authors and Affiliations
Contributions
Dr. ZN contributed in designing the study, conducting the study, data collection, data interpretation, critically reviewing the manuscript, and approving the final manuscript as submitted. Dr. SB contributed in designing the study, data collection, data analysis, data interpretation, preparing the first draft of the manuscript, and approving the final manuscript as submitted. Prof. VS contributed in the first idea of the study, designing the study, supervising the study, conducting the study, data interpretation, critically reviewing the manuscript, and approving the final manuscript as submitted. Dr. NA contributed in the first idea of the study, designing the study, conducting the study, data interpretation, critically reviewing the manuscript, and approving the final manuscript as submitted. Dr. FA contributed in the first idea of the study, designing the study, conducting the study, data interpretation, critically reviewing the manuscript, and approving the final manuscript as submitted.
Corresponding author
Ethics declarations
Conflicts of interests
Nothing to report.
Ethical approval
This study was approved by the regional bioethics committee of Isfahan University of Medical Sciences (approval code:195105) and therefore have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Rights and permissions
About this article
Cite this article
Nikoo, Z., Badihian, S., Shaygannejad, V. et al. Comparison of the efficacy of azathioprine and rituximab in neuromyelitis optica spectrum disorder: a randomized clinical trial. J Neurol 264, 2003–2009 (2017). https://doi.org/10.1007/s00415-017-8590-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-017-8590-0