Abstract
After myocardial ischemia, necrotic cell death occurs mainly during the first minutes of reperfusion through ATP-dependent hypercontracture leading to sarcolemmal rupture. Recent studies indicate that opening of a mitochondrial permeability transition pore (mPTP) is a critical event in reperfusion-induced necrosis.
Objective
We investigated the hypothesis that mPTP can induce hypercontracture.
Methods
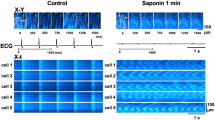
Both intact and digitonin-permeabilized rat cardiac myocytes were loaded with TMRE and submitted to oxidative damage (intermittent 568 nm laser illumination) to promote mPTP, detected as mitochondrial depolarization. The effect of cytosolic Ca2+ overload (5 mmol/L extracellular Ca2+) and ATP availability on mPTP-induced cell shortening were analyzed, and changes in cytosolic and mitochondrial Ca2+ were simultaneously monitored by confocal microscopy (Fluo-4 and Rhod-2).
Results
In the absence of Ca2+ overload, induction of mPTP was consistently followed by mitochondrial depolarization and rigor shortening that, in permeabilized cells, was prevented by ATP. Exposure of intact cardiac myocytes to 5mmol/L Ca2+ induced an increase in cytosolic and mitochondrial Ca2+ content. In Ca2+ overloaded myocytes, induction of mPTP resulted in a further increase in cytosolic Ca2+ and hypercontracture (> 50% reduction in length with distortion of cell geometry) that started before depolarization involved all mitochondria within the cell and could be prevented by the mPTP inhibitor cyclosporin A. In permeabilized myocytes, mPTP could promote hypercontracture when cytosolic Ca2+ overload was mimicked in the presence of ATP, and was prevented when ATP was removed from the intracellular-like medium.
Conclusions
mPTP opening may induce ATP-dependent hypercontracture in Ca2+ overloaded myocytes. This phenomenon could reconcile the apparently contradictory hypotheses of hypercontracture and mPTP opening as main determinants of necrosis during the first minutes of reperfusion.
Similar content being viewed by others
References
Altschuld RA,Wenger WC, Lamka KG, Kindig OR, Capen CC, Mizuhira V, Vander Heide RS (1985) Structural and functional properties of adult rat heart myocytes lysed with digitonin. J Biol Chem 260(26):14325–14334
Argaud L, Gateau-Roesch O, Muntean D, Chalabreysse L, Loufouat J, Robert D, Ovize M (2005) Specific inhibition of the mitochondrial permeability transition prevents lethal reperfusion injury. J Mol Cell Cardiol 38:367–374
Baines CP, Kaiser RA, Purcell NH, Blair NS, Osinska H, Hambleton MA, Brunskill EW, Sayen MR, Gottlieb RA, Dorn GW, Robbins J, Molkentin JD (2005) Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature 434:658–662
Barrabes JA, Garcia-Dorado D, Ruiz- Meana M, Piper HM, Solares J,Gonzalez MA, Oliveras J, Herrejon MP, Soler SJ (1996) Myocardial segment shrinkage during coronary reperfusion in situ. Relation to hypercontracture and myocardial necrosis. Pflugers Arch 431:519–526
Bonz A, Siegmund B, Ladilov Y, Vahl CF, Piper HM (1998) Metabolic recovery of isolated adult rat cardiomyocytes after energy depletion: existence of an ATP threshold? J Mol Cell Cardiol 30:2111–2119
Brennan JP, Southworth R, Medina RA, Davidson SM, Duchen MR, Shattock MJ (2006) Mitochondrial uncoupling, with low concentration FCCP, induces ROS-dependent cardioprotection independent of KATP channel activation. Cardiovasc Res 72:313–321
Clarke SJ, McStay GP, Halestrap AP (2002) Sanglifehrin A acts as a potent inhibitor of the mitochondrial permeability transition and reperfusion injury of the heart by binding to cyclophilin-D at a different site from cyclosporin A. J Biol Chem 277:34793–34799
Di Lisa F, Menabo R, Canton M, Barile M, Bernardi P (2001) Opening of the mitochondrial permeability transition pore causes depletion of mitochondrial and cytosolic NAD+ and is a causative event in the death of myocytes in postischemic reperfusion of the heart. J Biol Chem 276:2571–2575
Duchen MR, McGuinness O, Brown LA, Crompton M (1993) On the involvement of a cyclosporin A sensitive mitochondrial pore in myocardial reperfusion injury. Cardiovasc Res 27:1790–1794
Garcia-Dorado D, Theroux P, Duran JM, Solares J, Alonso J, Sanz E, Munoz R, Elizaga J, Botas J, Fernandez-Aviles F (1992) Selective inhibition of the contractile apparatus. A new approach to modification of infarct size, infarct composition, and infarct geometry during coronary artery occlusion and reperfusion. Circulation 85:1160–1174
Griffiths EJ, Halestrap AP (1995) Mitochondrial non-specific pores remain closed during cardiac ischaemia, but open upon reperfusion. Biochem J 307(Pt 1):93–98
Griffiths EJ,Ocampo CJ, Savage JS, Rutter GA, Hansford RG, Stern MD, Silverman HS (1998) Mitochondrial calcium transporting pathways during hypoxia and reoxygenation in single rat cardiomyocytes. Cardiovasc Res 39:423–433
Hausenloy D, Wynne A, Duchen M, Yellon D (2004) Transient mitochondrial permeability transition pore opening mediates preconditioning-induced protection. Circulation 109:1714–1717
Hausenloy DJ, Duchen MR, Yellon DM (2003) Inhibiting mitochondrial permeability transition pore opening at reperfusion protects against ischaemiareperfusion injury. Cardiovasc Res 60:617–625
Hausenloy DJ, Maddock HL, Baxter GF, Yellon DM (2002) Inhibiting mitochondrial permeability transition pore opening: a new paradigm for myocardial preconditioning? Cardiovasc Res 55:534–543
Haworth RA, Hunter DR, Berkoff HA (1981) Contracture in isolated adult rat heart cells. Role of Ca2+, ATP, and compartmentation. Circ Res 49:1119–1128
Holmuhamedov EL, Jovanovic S, Dzeja PP, Jovanovic A, Terzic A (1998) Mitochondrial ATP-sensitive K+ channels modulate cardiac mitochondrial function. Am J Physiol 275:H1567–H1576
Inserte J, Garcia-Dorado D, Hernando V, Soler-Soler J (2005) Calpain-mediated impairment of Na+/K+-ATPase activity during early reperfusion contributes to cell death after myocardial ischemia. Circ Res 97:465–473
Korge P, Goldhaber JI, Weiss JN (2001) Phenylarsine oxide induces mitochondrial permeability transition, hypercontracture, and cardiac cell death. Am J Physiol Heart Circ Physiol 280:H2203–H2213
Ladilov Y, Haffner S, Balser-Schafer C, Maxeiner H, Piper HM (1999) Cardioprotective effects of KB-R7943: a novel inhibitor of the reverse mode of Na+/Ca2+ exchanger. Am J Physiol 276:H1868–H1876
Miyata H, Lakatta EG, Stern MD, Silverman HS (1992) Relation of mitochondrial and cytosolic free calcium to cardiac myocyte recovery after exposure to anoxia. Circ Res 71:605–613
Miyazaki S, Fujiwara H, Onodera T, Kihara Y, Matsuda M, Wu D, Nakamura Y, Kumada T, Sasayama S, Kawai C, Hamashima Y (1987) Quantitative analysis of contraction band and coagulation necrosis after ischemia and reperfusion in the porcine heart. Circulation 75:1074–1082
Nakagawa T, Shimizu S, Watanabe T, Yamaguchi O, Otsu K, Yamagata H, Inohara H, Kubo T, Tsujimoto Y (2005) Cyclophilin D-dependent mitochondrial permeability transition regulates some necrotic but not apoptotic cell death.Nature 434(7033): 652–658
Penna C, Cappello S, Mancardi D, Raimondo S, Rastaldo R, Gattullo D, Losano G, Pagliaro P (2005) Post-conditioning reduces infarct size in the isolated rat heart: Role of coronary flow and pressure and the nitric oxide/ cGMP pathway. Basic Res Cardiol 101:168–179
Piper HM, Garcia-Dorado D, Ovize M (1998) A fresh look at reperfusion injury. Cardiovasc Res 38:291–300
Ruiz-Meana M, Garcia-Dorado D, Hofstaetter B, Piper HM, Soler-Soler J (1999) Propagation of cardiomyocyte hypercontracture by passage of Na(+) through gap junctions. Circ Res 85:280–287
Ruiz-Meana M, Garcia-Dorado D, Miro-Casas E, Abellan A, Soler-Soler J (2006) Mitochondrial Ca2+ uptake during simulated ischemia does not affect permeability transition pore opening upon simulated reperfusion. Cardiovasc Res 71:715–724
Schlack W, Uebing A, Schäfer M, Bier F, Schäfer S, Piper HM, Thämer V (1994) Regional contractile blockade at the onset of reperfusion reduces infarct size in the dog heart. Pflug Arch Eur J Phy 428:134–141
Sebbag L, Verbinski SG, Reimer KA, Jennings RB (2003) Protection of ischemic myocardium in dogs using intracoronary 2.3-butanedione monoxime (BDM). J Mol Cell Cardiol 35:165–176
Siegmund B, Koop A, Klietz T, Schwartz P, Piper HM (1990) Sarcolemmal integrity and metabolic competence of cardiomyocites under anoxia-reoxygenation. Am J Physiol 258:H285–H291
Sivaraman V, Mudalgiri NR, Di Salvo C, Kolvekar S, Hayward M, Yap J, Keogh B, Hausenloy DJ, Yellon DM (2007) Postconditioning protects human atrial muscle through the activation of the RISK pathway. Basic Res Cardiol 102:453–459
Sumida T, Otani H, Kyoi S, Okada T, Fujiwara H, Nakao Y, Kido M, Imamura H (2005) Temporary blockade of contractility during reperfusion elicits a cardioprotective effect of the p38 MAP kinase inhibitor SB-203580. Am J Physiol Heart Circ Physiol 288:H2726–H2734
Tani M, Hasegawa H, Suganuma Y, Shinmura K, Kayashi Y, Nakamura Y (1996) Protection of ischemic myocardium by inhibition of contracture in isolated rat heart. Am J Physiol 271:H2515–H2519
Vander Heide RS, Angelo JP, Altschuld RA, Ganote CE (1986) Energy dependence of contraction band formation in perfused hearts and isolated adult myocites. Am J Pathol 125:55–58
Vinten-Johansen J, Zhao ZQ, Zatta AJ, Kin H, Halkos ME, Kerendi F (2005) Postconditioning — A new link in nature’s armor against myocardial ischemia-reperfusion injury. Basic Res Cardiol 100:295–310
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruiz-Meana, M., Abellán, A., Miró-Casas, E. et al. Opening of mitochondrial permeability transition pore induces hypercontracture in Ca2+ overloaded cardiac myocytes. Basic Res Cardiol 102, 542–552 (2007). https://doi.org/10.1007/s00395-007-0675-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-007-0675-y