Summary
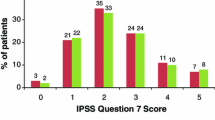
A double-blind placebo-controlled multicentre study was conducted to assess the efficacy and tolerability of the 5α-reductase inhibitor Permixon® in patients with symptomatic benign prostatic hyperplasia (BPH) who had previously been shown to be unresponsive to the placebo effect. Following a 30-day single-blind placebo run-in period, 176 nonresponders to placebo (those patients showing <30% improvement in peak urinary flow rate) were randomised to double-blind oral treatment with Permixon® 160mg twice daily or matching placebo for 30 days. Improvement in dysuria severity was seen in a significantly greater proportion ofPermixon® recipients (31.3%) than placebo recipients (16.1%). Daytime urinary frequency fell significantly in Permixon®-treated patients (11.3% reduction), but was unchanged in placebo recipients. Nocturnal urinary frequency fell to a significantly greater extent with Permixon® (32.5% reduction) than with placebo (17.7% reduction). Permixon® produced a significantly greater increase in mean peak urinary flow rate than did placebo (28.9 vs 8.5%). The global efficacy of Permixon® was judged by the patients and physicians to be satisfactory or better in 71.3 and 56.6% of cases, respectively; corresponding values for placebo were 67.5 and 47.2%, respectively. The overall tolerability of Permixon® was comparable to that of placebo. In conclusion, Permixon® appears to be significantly more effective than placebo and well tolerated in the short term treatment of mild to moderate symptomatic BPH.
Similar content being viewed by others
References
Berry SJ, Coffey DS, Walsh PC, et al. The development of human benign prostatic hyperplasia with age. J Urology 1984; 132: 474–79
Caine M. The present role of alpha-adrenergic blockers in the treatment of benign prostatic hypertrophy. J Urology 1986; 136: 1–4
Gingell JC. Review of current and future approaches to the management of benign prostatic hyperplasia. Postgrad Med J 1992; 68: 702–6
Lepor H. Nonoperative management of benign prostatic hyperplasia. J Urology 1989; 141: 1283–9
McConnell JD. Androgen ablation and blockade in the treatment of benign prostatic hyperplasia. Urol Clin North Am 1990; 17: 661–70
Isaacs JT, Brendler CB, Walsh PC. Changes in the metabolism of dihydrotestosterone in the hyperplastic human prostate. J Clin Endocrinol Metab 1983; 56: 139–46
Romijn JC. Steroid hormones, receptors and benign prostatic hyperplasia. New developments in biosciences. Vol. 5. Berlin: W de Gruyter. 1989: 63–72
Gormley GJ, Stoner E, Bruskewitz RC, et al. The effect of finasteride in men with benign prostatic hyperplasia. N Engl J Med 1992; 327: 1185–91
Stoner E, The Finasteride Study Group. The clinical effects of a 5α-reductase inhibitor, finasteride, on benign prostatic hyperplasia. J Urology 1992; 147: 1298–302
Délos S, Carsol JL, Lehlé C, et al. Metabolism of testosterone in primary culture of prostate epithelial cells [abstract no. 254]. Endocrine Society Congress 1994
Hirsch KS, Jones CD, Audia JE, et al. LY191704: a selective, nonsteroidal inhibitor of human steroid 5α-reductase type 1. Proc Natl Acad Sci USA 1993; 90: 5277–81
Sultan C, Terraza A, Devillier C, et al. Inhibition of androgen metabolism and binding by a liposterolic extract of Serenoa repens in human foreskin fibroblasts. J Steroid Biochem Mol Biol 1984; 20: 515–9
Ravenna L, Di Silverio F, Gulino A, et al. Antiandrogenic activity of Serenoa repens on prostatic cell growth [abstract]. XVI Meeting of the International Study Group for Steroid Hormones, Nov 28-Dec 1, 1993
Paubert-Braquet M, Bazan N, Servent N, et al. Lipidosterolic extract of Serenoa repens inhibits estradiol/testosterone induced prostate enlargement in the rat. Congress of the European Association of Urology, Berlin, Jul 1994
Di Silverio F, D’Eramo G, Lubrano C, et al. Evidence that Serenoa repens extract displays an antiestrogenic activity in prostatic tissue of benign prostatic hypertrophy. Eur Urol 1992; 21: 309–14
Tarayre JP, Delhon A, Lauressergue S, et al. Action anti-oedémateuse d’un extrait hexanique de drupes de Serenoa repens. Batr Ann Pharm Franç 1983; 4: 559–70
Martorana G, Giberti C, Pizzorno R, et al. Long term study with Serenoa repens extract in patients with prostate adenoma. Urologia 1986; 53: 366–9
Stausch G, Perles P, Vergult G, et al. Comparison of finasteride (Proscar®) and Serenoa repens (Permixon®) in the inhibition of 5α-reductase in healthy male volunteers. Eur Urol 1994; 26: 247–52
Champault G, Patel JC, Bannard AM. A double-blind trial of an extract of the plant Serenoa repens in benign prostatic hyperplasia. Br J Clin Pharmacol 1984; 18: 461–2
Smith R, Memon A, Smart CJ, et al. The value of Permixon in benign prostatic hypertrophy. Brit J Urol 1986; 58: 36–40
Birkhoff JD, Wiederhorn A, Hamilton ML, et al. Natural history of benign prostatic hypertrophy and acute urinary retention. Urology 1976; 7: 48–52
Castro JE. Pills for the benign prostate: trial designs. Proc R Soc Med 1972; 65: 126–7
Acknowledgements
The authors would like to thank all the participating investigators of this multicentre study. They are: A Archimbaud, J Aurouze, G Chekly, B Crepin, E David, JL Descotes, J Dufour, F Faisant, the late G Faure, JP Gauthier, J Hermabessiere, A Jaupitre, B Mathes, JC Miermont, JJ Rambeaud, M Des Roseaux, E Schramek, G Sotton, C Vilain, L Vincens.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Descotes, J., Rambeaud, J., Deschaseaux, P. et al. Placebo-Controlled Evaluation of the Efficacy and Tolerability of Permixon® in Benign Prostatic Hyperplasia after Exclusion of Placebo Responders. Clinical Drug Investigation 9, 291–297 (1995). https://doi.org/10.2165/00044011-199509050-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199509050-00007