Abstract
Background
Understanding the natural progression of untreated gastric cancer is critical for determining the disease prognosis as well as treatment options and timing. The aim of this study is to analyze the natural history of gastric cancer.
Patients and Methods
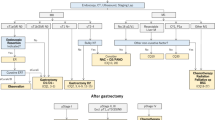
We included patients with gastric cancer who had not received any treatment and were staged using endoscopy/endoscopic ultrasonography and computed tomography on at least two follow-up visits during intervals of nontreatment. Tumor volumes were also measured in addition to the staging. Survival of each stage at diagnosis was also analyzed.
Results
A total of 101 patients were included. The mean follow-up period was 35.1 ± 34.4 months. The gastric cancer doubling time was 11.8 months for T1 and 6.2 months for T4. The progression time from early gastric cancer to advanced gastric cancer was 34 months. It decreased as the stages advanced: from 34 months between tumor-nodes-metastasis stage I and II to 1.8 months between stage III and IV. No variable was identified as a risk factor for cancer progression. The 5-year survival rates of untreated patients were 46.2% in stage I and 0% in stage II, stage III, and stage IV.
Conclusions
The progression and doubling times of gastric cancer shorten as the stages advance. Objective data reported in this study can be a critical factor in determining treatment timing and screening interval.



Similar content being viewed by others
References
Ito Y, Miyauchi A, Inoue H, et al. An observational trial for papillary thyroid microcarcinoma in Japanese patients. World J Surg. 2010;34(1):28–35.
Albertsen PC. Observational studies and the natural history of screen-detected prostate cancer. Curr Opin Urol. 2015;25(3):232–237.
Everett SM, Axon AT. Early gastric cancer: disease or pseudo-disease? Lancet. 1998;351(9112):1350–1352.
Stael von Holstein C, Eriksson S, Huldt B, Hammar E. Endoscopic screening during 17 years for gastric stump carcinoma A prospective clinical trial. Scand J Gastroenterol. 1991;26(10):1020–1026.
Yang JY, Lee HJ, Kim TH, et al. Short- and long-term outcomes after gastrectomy in elderly gastric cancer patients. Ann Surg Oncol. 2017;24(2):469–477.
Ryu EB, Chang JM, Seo M, Kim SA, Lim JH, Moon WK. Tumour volume doubling time of molecular breast cancer subtypes assessed by serial breast ultrasound. Eur Radiol 2014;24(9):2227–2235.
Rucker G, Messerer D. Remission duration: an example of interval-censored observations. Stat Med. 1988;7(11):1139–1145.
Wellner JA, Zhan YH. A hybrid algorithm for computation of the nonparametric maximum likelihood estimator from censored data. J Am Stat Assoc. 1997;92(439):945–959.
Friedman M. Piecewise exponential models for survival-data with covariates. Ann Stat. 1982;10(1):101–113.
Ahn HS, Lee HJ, Hahn S, et al. Evaluation of the seventh American Joint Committee on Cancer/International Union Against Cancer Classification of gastric adenocarcinoma in comparison with the sixth classification. Cancer. 2010;116(24):5592–5598.
Meiselas LE. Observations on the natural history of gastric cancer. Am J Med Sci. 1953;226(4):383–386.
Tsukuma H, Oshima A, Narahara H, Morii T. Natural history of early gastric cancer: a non-concurrent, long term, follow up study. Gut. 2000;47(5):618–621.
Fujisaki J, Nakajima T, Hirasawa T, et al. Natural history of gastric cancer-a case followed up for eight years: early to advanced gastric cancer. Clin J Gastroenterol. 2012;5(5):351–354.
Sato Y, Fujino T, Kasagawa A, et al. Twelve-year natural history of a gastric adenocarcinoma of fundic gland type. Clin J Gastroenterol. 2016;9(6):345–351.
Han DS, Suh YS, Kong SH, et al. Nomogram predicting long-term survival after d2 gastrectomy for gastric cancer. J Clin Oncol. 2012;30(31):3834–3840.
Takizawa K, Ono H, Kakushima N, et al. Risk of lymph node metastases from intramucosal gastric cancer in relation to histological types: how to manage the mixed histological type for endoscopic submucosal dissection. Gastric Cancer. 2013;16(4):531–536.
Oh SY, Lee KG, Suh YS, et al. Lymph node metastasis in mucosal gastric cancer: reappraisal of expanded indication of endoscopic submucosal dissection. Ann Surg 2017;265(1):137–142.
Acknowledgement
Statistical analysis was performed by the Medical Research Collaborating Center of SNUH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oh, SY., Lee, JH., Lee, HJ. et al. Natural History of Gastric Cancer: Observational Study of Gastric Cancer Patients Not Treated During Follow-Up. Ann Surg Oncol 26, 2905–2911 (2019). https://doi.org/10.1245/s10434-019-07455-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-07455-z