Abstract
Background
The effect of lymph node metastasis on local tumor control and distant failure in patients with anorectal melanoma has not been fully studied. Understanding the significance of lymphatic dissemination might assist in stratifying patients for either organ preservation or radical surgery.
Methods
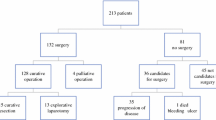
A retrospective review of all patients with anorectal melanoma who underwent surgery at our institution between 1985 and 2010. Abdominoperineal resection (APR) was performed in 25 patients (39 %), and wide local excision (WLE) in 40 (61%). Extent of primary surgery and locoregional lymphadenectomy (mesorectal vs. inguinal vs. none) and pattern of treatment failure were analyzed. Recurrence-free survival (RFS) and disease-specific survival (DSS) were calculated.
Results
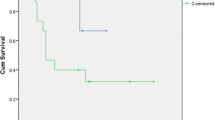
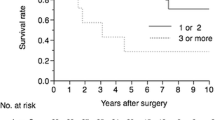
In patients undergoing APR, DSS was not associated with presence (29 %) or absence (71 %) of metastatic melanoma in mesorectal lymph nodes. There was a trend toward improved DSS in patients with clinically negative inguinal lymph nodes (n = 17) compared with patients with proven inguinal metastasis (n = 6; P = 0.12). Type of surgery (WLE vs. APR) was not associated with subsequent development of distant disease. Twelve patients (18 %) had synchronous local and distant recurrence. Synchronous recurrence was not associated with surgical strategy used to treat primary tumor (P = 0.28). Perineural invasion (PNI) was significantly correlated with RFS (P = 0.002).
Conclusions
Outcome following resection of anorectal melanoma is independent of locoregional lymph node metastasis; lymphadenectomy should be reserved for gross symptomatic disease. PNI is a powerful prognostic marker warranting further exploration in clinical trials.


Similar content being viewed by others
References
Robert C, Thomas L, Bondarenko I, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364:2517–26.
Chang AE, Karnell LH, Menck HR. The National Cancer Data Base report on cutaneous and noncutaneous melanoma: a summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer. 1998;83:1664–78.
Row D, Weiser MR. Anorectal melanoma. Clin Colon Rectal Surg. 2009;22(2):120–6.
Carvajal RD, Antonescu CR, Wolchok JD, et al. KIT as a therapeutic target in metastatic melanoma. JAMA. 2011;305:2327–34.
Antonescu CR, Busam KJ, Francone TD, et al. L576P KIT mutation in anal melanomas correlates with KIT protein expression and is sensitive to specific kinase inhibition. Int J Cancer. 2007;121:257–64.
Guo J, Si L, Kong Y, et al. Phase II, open-label, single-arm trial of imatinib mesylate in patients with metastatic melanoma harboring c-Kit mutation or amplification. J Clin Oncol. 2011;29(21):2904–9.
Iddings DM, Fleisig AJ, Chen SL, Faries MB, Morton DL. Practice patterns and outcomes for anorectal melanoma in the USA, reviewing three decades of treatment: is more extensive surgical resection beneficial in all patients? Ann Surg Oncol. 2010;17:40–4.
Nilsson PJ, Ragnarsson-Olding BK. Importance of clear resection margins in anorectal malignant melanoma. Br J Surg. 2010;97:98–103.
Yeh JJ, Shia J, Hwu WJ, et al. The role of abdominoperineal resection as surgical therapy for anorectal melanoma. Ann Surg. 2006;244:1012–7.
Brady MS, Kavolius JP, Quan SH. Anorectal melanoma: a 64-year experience at Memorial Sloan-Kettering Cancer Center. Dis Colon Rectum. 1995;38(2):146–51.
Nilsson PJ, Ragnarsson-Olding BK. Importance of clear resection margins in anorectal malignant melanoma. Br J Surg. 2010;97:98–103.
Droesch JT, Flum DR, Mann GN. Wide local excision or abdominoperineal resection as the initial treatment for anorectal melanoma? Am J Surg. 2005;189:446–9.
Gershenwald JE, Thompson W, Mansfield PF, et al. Multi-institutional melanoma lymphatic mapping experience: the prognostic value of sentinel lymph node status in 612 stage I or II melanoma patients. J Clin Oncol.1999;17:976–83.
Slingluff CL Jr, Vollmer RT, Seigler HF. Anorectal melanoma: clinical characteristics and results of surgical management in twenty-four patients. Surgery.1990;107:1–9.
Pessaux P, Pocard M, Elias D, et al. Surgical management of primary anorectal melanoma. Br J Surg. 2004;91:1183–7.
Yeh JJ, Weiser MR, Shia J, Hwu WJ. Response of stage IV anal mucosal melanoma to chemotherapy. Lancet Oncol. 2005;6:438–9.
Ueno H, Hase K, Mochizuki H. Criteria for extramural perineural invasion as a prognostic factor in rectal cancer. Br J Surg. 2001;88:994–1000.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perez, D.R., Trakarnsanga, A., Shia, J. et al. Locoregional Lymphadenectomy in the Surgical Management of Anorectal Melanoma. Ann Surg Oncol 20, 2339–2344 (2013). https://doi.org/10.1245/s10434-012-2812-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2812-6