Abstract
Background
Perioperative nutrition remains a significant problem in patients undergoing neoadjuvant treatment for esophageal cancer. The aim of this study was to evaluate the effectiveness of esophageal stenting, feeding tube placement, or observation among esophageal cancer patients receiving neoadjuvant therapy.
Methods and Materials
A review of our prospectively maintained database of esophageal cancer patients identified 58 patients who underwent neoadjuvant chemoradiotherapy. Operative complications, tolerance of neoadjuvant therapy, and nutritional outcomes were evaluated according to the type of nutritional adjunct used.
Results
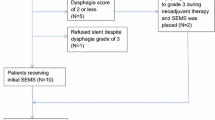
A total of 25 patients received esophageal stenting with self-expanding silicone stents. Of these, 19 patients had feeding tubes placed (without stenting), and 14 nonstented patients were maintained on oral diets alone. Stent patients showed a lower rate of interruption of chemoradiotherapy (8% vs. 29% vs. 47%, P = .011). The stent group also demonstrated greater mean improvement in albumin levels (0.14 g/dL vs. −0.39 g/dL vs. −0.45 g/dL, P < .001) and less percentage body weight loss (1.5% vs. 4.2% vs. 5.5%, P < .001). Nasogastric tubes were used for additional nutritional supplementation during the last week of therapy for two stent patients. The rate of stent migration was 24%. Overall, 31% of patients did not go on to resection because of progression to metastatic disease. The rate of major operative complication was 20% vs. 47% vs. 43% among stent, feeding tube, and oral nutrition patients respectively (P = .130).
Conclusions
Esophageal stenting in the neoadjuvant setting offers improved results compared with feeding tubes both in maintaining preoperative nutrition and in tolerance of neoadjuvant chemoradiotherapy. Future protocols of patients treated with multimodal therapy for cancer of the esophagus should investigate the potential therapeutic benefit of using removable silicone esophageal stents as an alternative to feeding tubes.
Similar content being viewed by others
References
Ries LAG, Melbert D, Krapcho M, Stinchcomb DG, Howlader N, Horner MJ, et al. editors. SEER cancer Statistics review, 1975–2005. Bethesda, MD.: National Cancer Institute. http://seer.cancer.gov/csr/1975_2005/, based on November 2007 SEER data submission, posted to the SEER website, 2008.
Reynolds JV, Muldoon C, Hollywood D, Ravi N, Rowley S, O’Byrne K, et al. Long-term outcomes following neoadjuvant chemoradiotherapy for esophageal cancer. Ann Surg. 2007;245:707–16.
Forastiere AA, Orringer MB, Perez-Tamayo C, Urba SG, Zahurak M. Preoperative chemoradiation followed by transhiatal esophagectomy for carcinoma of the esophagus: final report. J Clin Oncol. 1993;11:1118–23.
Larrea J, Vega S, Martinez T, Torrent JM, Vega V, Nunez V. The nutritional status and immunological situation of cancer patients. Nutr Hosp. 1992;7:178–84.
Han-Geurts IJ, Hop WC, Tran TC, Tilanus HW. Nutritional status as a risk factor in esophageal surgery. Dig Surg. 2006;23:159–63.
Cooper JS, Guo MD, Herskovic A, Macdonald JS, Martenson JA Jr, Al-Sarraf M, et al. Chemoradiotherapy of locally advanced esophageal cancer: long-term follow-up of a prospective randomized trial (RTOG 85-01). Radiation Therapy Oncology Group. JAMA. 1999;281:1623–7.
Jatoi A, Martenson JA, Foster NR, McLeod HL, Lair BS, Nichols F, et al. Paclitaxel, carboplatin, 5-fluorouracil, and radiation for locally advanced esophageal cancer: phase II results of preliminary pharmacologic and molecular efforts to mitigate toxicity and predict outcomes: North Central Cancer Treatment Group (N0044). Am J Clin Oncol. 2007;30:507–13.
Kassam Z, Wong RK, Ringash J, Ung Y, Kamra J, DeBoer G, et al. A phase I/II study to evaluate the toxicity and efficacy of accelerated fractionation radiotherapy for the palliation of dysphagia from carcinoma of the oesophagus. Clin Oncol (R Coll Radiol). 2008;20:53–60.
Daly JM, Weintraub FN, Shou J, Rosato EF, Lucia M. Enteral nutrition during multimodality therapy in upper gastrointestinal cancer patients. Ann Surg. 1995;221:327–38.
Windsor A, Braga M, Martindale R, Buenos R, Tepaske R, Kraehenbuehl L, et al. Fit for surgery: an expert panel review on optmising patients prior to surgery, with a particular focus on nutrition. Surgeon. 2004;2:315–9.
O’Gorman RB, Feliciano DV, Matthews KS, Matthews R, Bitondo CG, Mattox KL, et al. Correlation of immunologic and nutritional status with infectious complications after major abdominal trauma. Surgery. 1986;99:549–56.
Gibbs J, Cull W, Henderson W, Daley J, Hur K, Khuri SF. Preoperative serum albumin level as a predictor of operative mortality and morbidity: results from the National VA Surgical Risk Study. Arch Surg. 1999;134:36–42.
Reynolds JV, Ravi N, Hollywood D, Kennedy MJ, Rowley S, Ryan A, et al. Neoadjuvant chemoradiation may increase the risk of respiratory complications and sepsis after transthoracic esophagectomy. J Thorac Cardiovasc Surg. 2006;132:549–55.
Urschel JD, Vasan H. A meta-analysis of randomized controlled trials that compared neoadjuvant chemoradiation and surgery to surgery alone for resectable esophageal cancer. Am J Surg. 2003;185:538–43.
Braga M, Gianotti L, Gentilini O, Parisi V, Salis C, Di C, V. Early postoperative enteral nutrition improves gut oxygenation and reduces costs compared with total parenteral nutrition. Crit Care Med. 2001;29:242–8.
Heys SD, Walker LG, Smith I, Eremin O. Enteral nutritional supplementation with key nutrients in patients with critical illness and cancer: a meta-analysis of randomized controlled clinical trials. Ann Surg. 1999;229:467–77.
Kudsk KA, Minard G, Croce MA, Brown RO, Lowrey TS, Pritchard FE, et al. A randomized trial of isonitrogenous enteral diets after severe trauma. An immune-enhancing diet reduces septic complications. Ann Surg. 1996;224:531–40.
Moore FA, Feliciano DV, Andrassy RJ, McArdle AH, Booth FV, Morgenstein-Wagner TB, et al. Early enteral feeding, compared with parenteral, reduces postoperative septic complications. The results of a meta-analysis. Ann Surg. 1992;216:172–83.
Perioperative total parenteral nutrition in surgical patients. The Veterans Affairs Total Parenteral Nutrition Cooperative Study Group. N Engl J Med. 1991;325:525–32.
Bozzetti F, Braga M, Gianotti L, Gavazzi C, Mariani L. Postoperative enteral versus parenteral nutrition in malnourished patients with gastrointestinal cancer: a randomised multicentre trial. Lancet. 2001;358:1487–92.
Jenkinson AD, Lim J, Agrawal N, Menzies D. Laparoscopic feeding jejunostomy in esophagogastric cancer. Surg Endosc. 2007;21:299–302.
Margolis M, Alexander P, Trachiotis GD, Gharagozloo F, Lipman T. Percutaneous endoscopic gastrostomy before multimodality therapy in patients with esophageal cancer. Ann Thorac Surg. 2003;76:1694–7.
Ohnmacht GA, Allen MS, Cassivi SD, Deschamps C, Nichols FC, III, Pairolero PC. Percutaneous endoscopic gastrostomy risks rendering the gastric conduit unusable for esophagectomy. Dis Esophagus. 2006;19:311–2.
Martin R, Duvall R, Ellis S, Scoggins CR. The use of self-expanding silicone stents in esophageal cancer care: optimal pre-, peri-, and postoperative care. Surg Endosc. 2009;23:615–21.
Costamagna G, Shah SK, Tringali A, Mutignani M, Perri V, Riccioni ME. Prospective evaluation of a new self-expanding plastic stent for inoperable esophageal strictures. Surg Endosc. 2003;17:891–5.
Holm AN, de la Mora Levy JG, Gostout CJ, Topazian MD, Baron TH. Self-expanding plastic stents in treatment of benign esophageal conditions. Gastrointest Endosc. 2008;67:20–5.
Karbowski M, Schembre D, Kozarek R, Ayub K, Low D. Polyflex self-expanding, removable plastic stents: assessment of treatment efficacy and safety in a variety of benign and malignant conditions of the esophagus. Surg Endosc. 2008;22:1326–33.
Thompson AM, Rapson T, Gilbert FJ, Park KG. Endoscopic palliative treatment for esophageal and gastric cancer: techniques, complications, and survival in a population-based cohort of 948 patients. Surg Endosc. 2004;18:1257–62.
Ponsky JL, Gauderer MW. Percutaneous endoscopic gastrostomy: a nonoperative technique for feeding gastrostomy. Gastrointest Endosc. 1981;27:9–11.
Siddiqui AA, Loren D, Dudnick R, Kowalski T. Expandable polyester silicon-covered stent for malignant esophageal strictures before neoadjuvant chemoradiation: a pilot study. Dig Dis Sci. 2007;52:823–9.
Ott C, Ratiu N, Endlicher E, Rath HC, Gelbmann CM, Scholmerich J, et al. Self-expanding Polyflex plastic stents in esophageal disease: various indications, complications, and outcomes. Surg Endosc. 2007;21:889–96.
Conigliaro R, Battaglia G, Repici A, De Pretis G, Ghezzo L, Bittinger M, et al. Polyflex stents for malignant oesophageal and oesophagogastric stricture: a prospective, multicentric study. Eur J Gastroenterol Hepatol. 2007;19:195–203.
van ME, van der Gaast A, Looman CW, Tilanus HW, Muller K, Essink-Bot ML. Quality of life during neoadjuvant treatment and after surgery for resectable esophageal carcinoma. Int J Radiat Oncol Biol Phys. 2008;71:160–6.
Pennathur A, Luketich JD, Landreneau RJ, Ward J, Christie NA, Gibson MK, et al. Long-term results of a phase II trial of neoadjuvant chemotherapy followed by esophagectomy for locally advanced esophageal neoplasm. Ann Thorac Surg. 2008;85:1930–6.
Bozzetti F, Cozzaglio L, Gavazzi C, Bidoli P, Bonfanti G, Montalto F, et al. Nutritional support in patients with cancer of the esophagus: impact on nutritional status, patient compliance to therapy, and survival. Tumori. 1998;84:681–6.
Koea JB, Shaw JH. The effect of tumor bulk on the metabolic response to cancer. Ann Surg. 1992;215:282–8.
Saito T, Kuwahara A, Shigemitsu Y, Kinoshita T, Shimoda K, Miyahara M, et al. Factors related to malnutrition in patients with esophageal cancer. Nutrition. 1991;7:117–21.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bower, M., Jones, W., Vessels, B. et al. Nutritional Support with Endoluminal Stenting During Neoadjuvant Therapy for Esophageal Malignancy. Ann Surg Oncol 16, 3161–3168 (2009). https://doi.org/10.1245/s10434-009-0630-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-009-0630-2