Abstract
Study design: Retrospective descriptive study.
Objectives: (1) To obtain an overall appraisal of patients with epidural metastatic spinal cord compression (SCC) admitted to nine Dutch spinal cord units (SCUs) between 01-01-1990 and 01-01-2000. (2) To identify factors that predict survival >1 year after in-patient rehabilitation of patients with epidural metastatic SCC.
Setting: Dutch SCUs. Rehabilitation Center Amsterdam, Amsterdam. Rehabilitation Center Heliomare, Wijk aan Zee. Rehabilitation Center De Hoogstraat, Utrecht. Rehabilitation Center Rijndam, Rotterdam. Rehabilitation Center Sophia Stichting, Den Haag. Rehabilitation Center Beatrixoord, Haren. Rehabilitation Center Het Roessingh, Enschede. Rehabilitation Center Sint Maartenskliniek, Nijmegen. Rehabilitation Center SRL Hoensbroeck, Hoensbroek.
Methods: (1) Clinical records were reviewed and demographic, clinical and functional data were collected according to a protocol. The date of admittance to the SCU, rehabilitation goals, date of discharge and date of death were recorded. (2) The odds ratio (OR) was calculated for all determinants on admittance to the SCU in order to find indicators that predict survival >1 year after discharge from the SCU. An OR ⩾2 was considered to be clinically significant.
Results: In total, 131 patients with epidural metastatic SCC were admitted. In all, 117 clinical records were retrieved and 97 clinical records provided complete data. The average age on admittance was 58 years. Among the patients 53% were male. The average Barthel score on admittance was 7.2/20 points. The average length of stay in the SCU was 104 days (3–336). Overall, 66% of the patients were discharged. The average Barthel score on discharge was 12.0 points. The average survival after discharge was 808 (0–3669) days. During their stay on the SCU, seven patients died. At 1 year after discharge, 52% of the patients were still alive. These patients suffered less complications, had been admitted less often to a hospital during rehabilitation, had made better functional progress and had been discharged home more often. A survival >1 year after discharge is related to ASIA D (OR 4.3), MRC 4 and 5 (OR 5.4), tumour in remission (OR 3.8) and independence or partial independence on the Barthel items: dressing (OR 4.3) and making transfers (OR 5.0).
Conclusions: Patients with epidural metastatic SCC may benefit from in-patient rehabilitation.
Similar content being viewed by others
Introduction
Metastases to the spine are a common manifestation of systemic neoplasia. The incidence of metastases to the spine has been estimated at 5%,1 but continues to increase, probably as a result of increasing survival for patients with cancer.2,3 Up to 10% of cancer patients develop symptomatic epidural metastatic spinal lesions.4,5,6 Cancer from breast, lung, prostate, haemopoietic and renal origins account for the majority of epidural spinal metastases.7,8,9 The thoracic spine is most commonly affected, followed by the lumbar spine and the cervical spine.7,10 The mean survival of patients with neoplastic spinal cord compression (SCC) after developing neurological signs is between 3 and 9 months.11,12,13,14 Although it has been estimated that over 80% of patients with neoplastic SCC survive no longer than 1 year, a select group of patients may survive for extended periods, sometimes as long as 4–9 years.12,13,14,15
Epidural metastatic SCC has a major influence on the patient's physical condition. Patients are confronted with significant impairment in mobility, self-care, bowel and bladder management and sexuality. A minority of cancer patients with SCC (10–14%) is admitted to a spinal cord unit (SCU).16
Prior studies have shown that patients with epidural metastatic SCC can achieve functional improvements in mobility and self-care in an SCU.17,18,19 One study reported a sum-score of six factors to be prognostic for survival >1 year after in-patient rehabilitation. These factors were: tumour biology (lymphoma, myeloma, breast and kidney tumours); SCC as presenting symptom of malignancy; a slow (>1 week) progression rate of neurological symptoms; tumour treated with a combination of surgery and radiotherapy; (partial) bowel control on admission: and (partial) independence with transfer activities on admission.20 In The Netherlands, patients with SCC due to epidural metastases are admitted to a general hospital. Here they receive treatment in order to fight the cancer and stop the neurological loss due to the SCC. Medically stable patients may be admitted to an SCU in a rehabilitation centre for in-patient rehabilitation. In The Netherlands, where rehabilitation centres are freestanding institutions, there are 25 centres, nine of which have an SCU. The specialists working in the SCUs are rehabilitation physicians who are specialised in SCC. Patients with SCC due to epidural metastases may be admitted to an SCU for in-patient rehabilitation, on the following criteria:
-
1
The prognosis of survival is >1 year.
-
2
The patient is physically able to receive rehabilitation therapy.
-
3
The patient has the possibility to go home after discharge from the SCU.
The objectives of this study were twofold. (1) To obtain an overall appraisal of patients with epidural metastatic SCC admitted to the nine Dutch SCUs between 01-01-1990 and 01-01-2000. (2) To identify factors related to both SCC and cancer that predict survival >1 year after in-patient rehabilitation in patients with epidural metastatic SCC.
Materials and methods
Clinical records from all patients with epidural metatstatic SCC admitted to the nine Dutch SCUs between 01-01-1990 and 01-01-2000 were reviewed, and data were collected according to a protocol. Demographic data included date of birth, gender, residence and housing. Clinical data included date of diagnosis of the primary malignancy, date of diagnosis of SCC, tumour biology, therapy applied for SCC, comorbidity, pain, progression rate of neurological symptoms, number of tumour locations, neurological level of SCC, ASIA classification of SCC and medical complications during admittance to the SCU. The functional ability was scored on a 20-point Barthel index on admittance, and on discharge.
Date of admittance, date of discharge and date of death were recorded.
Data analysis
Data collection was completed on 01-03-2002, and data were subsequently analysed. The patients were divided into two groups, according to their survival after discharge. Survival >1 year after discharge was determined as a dividing line in this study.
In order to find indicators for survival >1 year after discharge we determined the odds ratio (OR) of all demographic, clinical and functional factors on admission. We only determined the OR when the group of patients to be compared consisted of at least 20 patients. The OR is a way of estimating the strength of association between indicators and survival >1 year after discharge. The higher the odds, the greater the strength. These ratios are well suited to study indicators of prognosis,21 and were used earlier in a study on this topic.20
Results
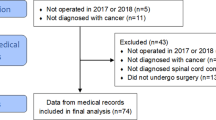
In all, 131 patients with epidural metastatic SCC were admitted to an SCU from 01-01-1990 to 01-01-2000. In total, 117 clinical records were retrieved. Two SCUs could not provide complete data. In 20 cases, the date of death could not be ascertained. These patients were similar to the other patients with regard to age, gender and Barthel score on admittance to the SCU. A total of 97 complete records were reviewed, and data were collected according to a protocol. Among them, 51 were men and 46 were women admitted with a mean age of 59 (24–79) years. The cervical spine was the site of the tumour in 7% of the cases, the thoracic spine in 82% and the lumbar spine in 11%. Comorbidity was present in 40%. In 50% the tumour was the first presenting sign of malignancy. In 40% pain preceded neurological signs, whereas 16% patients had no pain at all. In 50% one tumour site was found, in 49% more than one tumour site and the number of tumour sites was not known in 1%. There was remission of the tumour in 14%. The average Barthel score on admittance was 7.2/20 points. The distribution of other factors such as tumour biology, progression rate, therapy, MRC scale and ASIA classification are listed in Table 1.
The average stay in the SCU was 104 (3–336) days. During their stay in the SCU, 59% of the patients had a medical complication. Those 27% patients who had a medical complication during their stay at the SCU had to be admitted to a general hospital. The average Barthel score on discharge was 12.0 points. In all, 68% of the patients were discharged (Table 2).
The average survival after discharge until the completion of data collection on 01-03-2002 was 808 (0–3669) days. Survival after diagnosis of primary malignancy was 1902 (130–8771) days, whereas survival after SCC was 1109 (10–3669) days. During their stay in an SCU, seven patients died. Three patients were allowed to die in their home, close to their family. The other 87 patients were discharged in a physically ‘good’ condition.
Those patients who survived >1 year after in-patient rehabilitation were 52%. When compared with the patients who died during the first year after discharge from the SCU, there were no differences in age, gender or Barthel score on admittance (Table 1).
Of all the indicators analysed, nine showed a positive relationship with survival >1 year after discharge from the SCU (OR≥2) (Table 3). In addition to prolonged survival, this group also had less medical complications (48% versus 66%), were less often admitted to a hospital (18% versus 34%), had a higher Barthel score on discharge (15.5 versus 8.0) and were most often discharged (82% versus 49%) (Table 2).
Discussion
Patients with epidural metastatic SCC should not spend their remaining life-span in an SCU, but in their home environment, close to their friends and family. When possible, great effort must be made to discharge these patients directly to their home from the hospital. However, for some patients admittance to an SCU may be beneficial, especially when expected survival is relatively long.
The selection criteria applied in The Netherlands for admittance to an SCU for in-patient rehabilitation are listed in the introduction. The prognosis of survival >1 year after in-patient rehabilitation is considered to be the most important criterium.
An overall appraisal of patients with epidural metastatic SCC admitted to an SCU between 01-01-1990 and 01-01-2000 was obtained in this study. Two SCUs could not provide complete data, due to administrative reasons, and the date of death of 20 patients could not be determined. These 20 patients were compared with the remaining 97 patients in the study, and there were no differences in age, gender or Barthel score on admittance to the SCU. Therefore, there is no indication of selective loss to follow-up.
Patients with epidural metastatic SCC were admitted to an SCU for a shorter length of time than patients with an SCC due to other causes. Patients stayed in the SCU for an average of 104 (6–336) days. A study carried out by Schonherr et al22 reported that patients had an average stay of 154 days in an SCU in The Netherlands (traumatic and nontraumatic causes).
Comparison with studies preformed in other countries is difficult. In The Netherlands the organisation of SCUs differs from that in other countries. Patients with an SCC are admitted to an SCU after discharge from a general hospital. In comparison to most other countries, patients stay longer in an SCU in The Netherlands.
One of the most important findings is that some patients with epidural metastatic SCC survive for a long time. One year after discharge from the SCU, 52% of the patients were still alive. The average survival was 1472 (395–6600) days; this is an underestimation because at the time of data analysis (01-03-2002) 30 patients were still alive.
The group of patients who survived >1 year after discharge differed in some ways from the group who survived <1 year after discharge. Their functional progress was greater (7.4 points versus 2.5 points on the Barthel score). Earlier studies endorse this functional gain.10,16,17,18,19 The discharge destination differed between the two groups studied. Patients who survived >1 year after in-patient rehabilitation were discharged to their home more often than patients who survived <1 year (82% versus 64%). Therefore, it can be concluded that patients with a prolonged survival (>1 year after inpatient rehabilitation) not only live longer but also benefit more from in–patient rehabilitation.
Nine indicators were found to be prognostic for survival >1 year after discharge (Table 3). The highest OR was found for ASIA D and MRC 4 or 5, remission of the tumour and independence or partial independence on the Barthel items ‘transfer’ and ‘dressing’. In a study carried out by Hacking et al, tumour state and (partial) independence with transfers were also found to be prognostic for prolonged survival.20 Our study could not confirm these findings.
Our study shows that patients with epidural metastatic SCC can benefit from in-patient rehabilitation in an SCU. It should be noted that this study was performed in specialised SCUs where there is a selection of patients. Therefore, the results cannot be generalised to all patients with epidural metastatic SCC.
References
Barton KD, Hirano A, Akari S, Terry RD . Experience with metastatic neoplasm involving the spinal cord. Neurology 1959; 9: 91–105.
Bailar III JC, Gornik HL . Cancer undefeated. N Eng J Med 1997; 336: 1569–1574.
Haddart RA, Rajan B, Law M, Meyer L, Dearnaly DP . Spinal cord compression in prostate cancer: treatment outcome and prognostic factors. Radiother and Oncol 1997; 44: 229–236.
Sundaresan N, Steinberger AA, Moore F, Sachdey VP, Krol G, Hough L . Indications and results of combined anterior and posterior approaches for spine tumour surgery. J Neurosurg 1996; 85: 438–446.
Grant R, Papadopoulos SM, Greenberg HS . Metastatic epidural spinal cord compression. Neural Clin 1991; 9: 825–841.
Fornasier VL, Home JG . Metastases to the vertebral column. Cancer 1975; 36: 590–594.
Abdu WA, Provencher M . Primary bone and metastatic tumors of the cervical spine. Spine 1998; 23: 2767–2777.
Hammerberg KW . Surgical treatment of metastatic spine disease. Spine 1992; 17: 1148–1153.
Sioutos PJ, Arbit E, Meshulam CF, Galachich JH . Spinal metastases from solid tumors Analysis of factors affecting survival. Cancer 1995; 76: 1453–1459.
Murray PK . Functional outcome and survival in spinal cord injury secondary to neoplasia. Cancer 1985; 55: 197–201.
Ampill FL . Epidural compression from metastatic tumour with resultant paralysis. J Neuro-Oncol 1989; 7: 129–136.
Dunn RC, Kelly WA, Wohns RNW, Howe JF . Spinal epidural neoplasia: a 15 year review of the results of surgical therapy. J Neurosurg 1980; 52: 47–51.
Livingstone KE, Perrin RG . The neurosurgical management of spinal metastases causing cord and cauda equina compression. J Neurosurg 1978; 49: 839–843.
Soelberg SP, Borgesen SE, Ramusson B, Boge-Ramussen T, Holt Larsen B, Gjerris F . Metastatic epidural spinal cord compression. Cancer 1990; 65: 1502–1508.
Bach F Larsen B, Rohde K, Borgesen S, Gjerris F, Boge-Rasmustoms T et al. Metastatic spinal cord compression, occurrence, symptoms, clinical presentations and prognosis in 398 patients with spinal cord compression. Acta Neurochir 1990; 107: 37–43.
McKinley WO, Seel RT, Hardman JT . Nontraumatic spinal cord injury: incidence, epidemiology, and functional outcome. Arch Phys Med Rehab 1999; 80: 619–623.
McKinley WO, Conti-Wyneken AR, Vokac CW, Cifu DX . Rehabilitative functional outcome of patients with neoplastic spinal cord compression. Arch Phys Med Rehab 1996; 77: 892–895.
McKinley WO, Huang ME, Brunsvold KT . Neoplastic versus traumatic spinal cord injury: an outcome comparison after inpatient rehabilitation. Arch Phys Med Rehab 1999; 80: 1253–1257.
McKinley WO, Huang ME, Tweksbury MA . Neoplastic versus traumatic spinal cord injury: an inpatient rehabilitation comparison. Am J Phys Med Rehab 2000; 81: 359–363.
Hacking HGA, Van As HHJ, Lankhorst GJ . Factors related to the outcome of inpatient rehabilitation in patients with neoplastic spinal cord compression. Paraplegia 1993; 31: 367–374.
Sackett DL, Haynes RB, Tugwell P . A basic science for clinical medicine. In: Clinical Epidemiology. Little, Brown and Company: Lippincott Williams & Winkins, 1991.
Schonherr MC, Groothoff JW, Mulder GA, Eisma WH . Rehabilitation of patients with spinal cord lesions in The Netherlands: an epidemiologic study. Spinal Cord 1996; 34: 679–683.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eriks, I., Angenot, E. & Lankhorst, G. Epidural metastatic spinal cord compression: functional outcome and survival after inpatient rehabilitation. Spinal Cord 42, 235–239 (2004). https://doi.org/10.1038/sj.sc.3101555
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101555
Keywords
This article is cited by
-
Clinical outcome of surgical management for symptomatic metastatic spinal cord compression from prostate cancer
BMC Urology (2020)
-
Challenges in rehabilitation of patients with nontraumatic spinal cord dysfunction due to tumors
Wiener klinische Wochenschrift (2019)
-
Factors predictive of survival and estimated years of life lost in the decade following nontraumatic and traumatic spinal cord injury
Spinal Cord (2017)
-
Understanding the Role of Rehabilitation Medicine in the Care of Patients with Tumor Causing Spinal Cord Dysfunction
Current Physical Medicine and Rehabilitation Reports (2017)
-
Cancer rehabilitation: a barometer for survival?
Supportive Care in Cancer (2015)