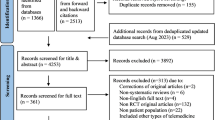
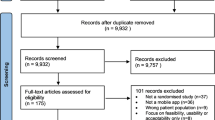
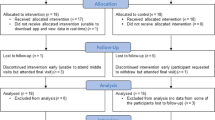
Abstract
The past decade in rheumatology has seen tremendous innovation in digital health technologies, including the electronic health record, virtual visits, mobile health, wearable technology, digital therapeutics, artificial intelligence and machine learning. The increased availability of these technologies offers opportunities for improving important aspects of rheumatology, including access, outcomes, adherence and research. However, despite its growth in some areas, particularly with non-health-care consumers, digital health technology has not substantially changed the delivery of rheumatology care. This Review discusses key barriers and opportunities to improve application of digital health technologies in rheumatology. Key topics include smart design, voice enablement and the integration of electronic patient-reported outcomes. Smart design involves active engagement with the end users of the technologies, including patients and clinicians through focus groups, user testing sessions and prototype review. Voice enablement using voice assistants could be critical for enabling patients with hand arthritis to effectively use smartphone apps and might facilitate patient engagement with many technologies. Tracking many rheumatic diseases requires frequent monitoring of patient-reported outcomes. Current practice only collects this information sporadically, and rarely between visits. Digital health technology could enable patient-reported outcomes to inform appropriate timing of face-to-face visits and enable improved application of treat-to-target strategies. However, best practice standards for digital health technologies do not yet exist. To achieve the potential of digital health technology in rheumatology, rheumatology professionals will need to be more engaged upstream in the technology design process and provide leadership to effectively incorporate the new tools into clinical care.
Key points
-
Digital health technology (DHT) offers enormous potential to improve rheumatology care but this potential has so far been largely unrealized.
-
The electronic health record, virtual visits, mobile health, wearable technology, digital therapeutics, artificial intelligence and machine learning could all have a role individually and in combination to reshape rheumatology practice.
-
Increased use of user-centred design in the development of digital rheumatology tools is needed and will facilitate electronic patient-reported outcomes becoming a cornerstone of rheumatology.
-
As rheumatology patients often have difficulties using their hands, voice-enabled technology might be particularly critical in this field.
-
A more concerted effort on the part of rheumatology professionals to participate in shaping the development and implementation of DHTs could make the benefits realized sooner and more effectively.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Friedberg, M. W. et al. Factors affecting physician professional satisfaction and their implications for patient care, health systems, and health policy. Rand Health Q. 3, 1 (2014).
Arndt, B. G. et al. Tethered to the EHR: primary care physician workload assessment using EHR event log data and time-motion observations. Ann. Fam. Med. 15, 419–426 (2017).
Keyhani, S. et al. Electronic health record components and the quality of care. Med. Care 46, 1267–1272 (2008).
Gross, P. A. & Bates, D. W. A pragmatic approach to implementing best practices for clinical decision support systems in computerized provider order entry systems. J. Am. Med. Inform. Assoc. 14, 25–28 (2007).
Ledwich, L. J., Harrington, T. M., Ayoub, W. T., Sartorius, J. A. & Newman, E. D. Improved influenza and pneumococcal vaccination in rheumatology patients taking immunosuppressants using an electronic health record best practice alert. Arthritis Rheum. 61, 1505–1510 (2009).
Schmajuk, G. & Yazdany, J. Leveraging the electronic health record to improve quality and safety in rheumatology. Rheumatol. Int. 37, 1603–1610 (2017).
Ancker, J. S. et al. Effects of workload, work complexity, and repeated alerts on alert fatigue in a clinical decision support system. BMC Med. Inform. Decis. Mak. 17, 36 (2017).
Orgun, B. & Vu, J. HL7 ontology and mobile agents for interoperability in heterogeneous medical information systems. Comput. Biol. Med. 36, 817–836 (2006).
Warner, J. L. et al. Temporal phenome analysis of a large electronic health record cohort enables identification of hospital-acquired complications. J. Am. Med. Inform. Assoc. 20, e281–e287 (2013).
Gostin, L. O. & Nass, S. Reforming the HIPAA privacy rule: safeguarding privacy and promoting research. JAMA 301, 1373–1375 (2009).
Bari, L. & O’Neill, D. P. Rethinking patient data privacy in the era of digital health. Health Affairs https://www.healthaffairs.org/do/10.1377/hblog20191210.216658/full/ (2019).
Bowman, S. Impact of electronic health record systems on information integrity: quality and safety implications. Perspect. Health Inf. Manage. 10, 1c (2013).
Tai-Seale, M. et al. Technology-enabled consumer engagement: promising practices at four health care delivery organizations. Health Aff. 38, 383–390 (2019).
Delbanco, T. et al. Inviting patients to read their doctors’ notes: a quasi-experimental study and a look ahead. Ann. Intern. Med. 157, 461–470 (2012).
Wu, A. W., Kharrazi, H., Boulware, L. E. & Snyder, C. F. Measure once, cut twice–adding patient-reported outcome measures to the electronic health record for comparative effectiveness research. J. Clin. Epidemiol. 66, S12–S20 (2013).
Yazdany, J. et al. Rheumatology informatics system for effectiveness: a national informatics-enabled registry for quality improvement. Arthritis Care Res. 68, 1866–1873 (2016).
Tonner, C., Schmajuk, G. & Yazdany, J. A new era of quality measurement in rheumatology: electronic clinical quality measures and national registries. Curr. Opin. Rheumatol. 29, 131–137 (2017).
Austin, L. et al. Providing ‘the bigger picture’: benefits and feasibility of integrating remote monitoring from smartphones into the electronic health record. Rheumatology 59, 367–378 (2020).
Jain, T., Lu, R. J. & Mehrotra, A. Prescriptions on demand: the growth of direct-to-consumer telemedicine companies. JAMA 322, 925–926 (2019).
Tan, L. F., Mason, N. & Gonzaga, W. J. Virtual visits for upper respiratory tract infections in adults associated with positive outcome in a Cox model. Telemed. J. E. Health 23, 200–204 (2017).
Ashwood, J. S., Mehrotra, A., Cowling, D. & Uscher-Pines, L. Direct-to-consumer telehealth may increase access to care but does not decrease spending. Health Aff. 36, 485–491 (2017).
Dixon, R. F. & Stahl, J. E. A randomized trial of virtual visits in a general medicine practice. J. Telemed. Telecare 15, 115–117 (2009).
Mehrotra, A., Paone, S., Martich, G. D., Albert, S. M. & Shevchik, G. J. A comparison of care at e-visits and physician office visits for sinusitis and urinary tract infection. JAMA Int. Med. 173, 72–74 (2013).
American College of Rheumatology Committee on Rheumatology Training and Workforce Issues. et al. Regional distribution of adult rheumatologists. Arthritis Rheum. 65, 3017–3025 (2013).
McDougall, J. A., Ferucci, E. D., Glover, J. & Fraenkel, L. Telerheumatology: a systematic review. Arthritis Care Res. 69, 1546–1557 (2017).
Silver, L. Smartphone ownership is growing rapidly around the world, but not always equally. Pew Research Center https://www.pewresearch.org/global/2019/02/05/smartphone-ownership-is-growing-rapidly-around-the-world-but-not-always-equally/ (2019).
Rhoades, H., Wenzel, S., Rice, E., Winetrobe, H. & Henwood, B. No digital divide? Technology use among homeless adults. J. Soc. Distress Homeless 26, 73–77 (2017).
Singh, K. et al. Many mobile health apps target high-need, high-cost populations, but gaps remain. Health Aff. 35, 2310–2318 (2016).
Najm, A. et al. Mobile health apps for self-management of rheumatic and musculoskeletal diseases: systematic literature review. JMIR Mhealth Uhealth 7, e14730 (2019).
Veinot, T. C., Mitchell, H. & Ancker, J. S. Good intentions are not enough: how informatics interventions can worsen inequality. J. Am. Med. Inf. Assoc. 25, 1080–1088 (2018).
Vangeepuram, N. et al. Smartphone ownership and perspectives on health apps among a vulnerable population in East Harlem, New York. Mhealth 4, 31 (2018).
Xcertia. mHealth App Guidelines. https://www.himss.org/sites/hde/files/media/file/2020/04/17/xcertia-guidelines-2019-final.pdf (2019).
Luo, D. et al. Mobile apps for individuals with rheumatoid arthritis: a systematic review. J. Clin. Rheumatol. 25, 133–141 (2019).
Grainger, R., Townsley, H., White, B., Langlotz, T. & Taylor, W. J. Apps for people with rheumatoid arthritis to monitor their disease activity: a review of apps for best practice and quality. JMIR Mhealth Uhealth 5, e7 (2017).
Del Mar, B. The history of clinical Holter monitoring. Ann. Noninvasive Electrocardiol. 10, 226–230 (2005).
Dinh-Le, C., Chuang, R., Chokshi, S. & Mann, D. Wearable health technology and electronic health record integration: scoping review and future directions. JMIR Mhealth Uhealth 7, e12861 (2019).
Patel, M. S., Asch, D. A. & Volpp, K. G. Wearable devices as facilitators, not drivers, of health behavior change. JAMA 313, 459–460 (2015).
Jakicic, J. M. et al. Effect of wearable technology combined with a lifestyle intervention on long-term weight loss: the IDEA randomized clinical trial. JAMA 316, 1161–1171 (2016).
Shuger, S. L. et al. Electronic feedback in a diet- and physical activity-based lifestyle intervention for weight loss: a randomized controlled trial. Int. J. Behav. Nutr. Phys. Act. 8, 41 (2011).
Donnelly, J. E. et al. Aerobic exercise alone results in clinically significant weight loss for men and women: Midwest exercise trial 2. Obesity 21, E219–E228 (2013).
Ridgers, N. D. et al. A cluster-randomised controlled trial to promote physical activity in adolescents: the Raising Awareness of Physical Activity (RAW-PA) study. BMC Public. Health 17, 6 (2017).
Gossec, L. et al. Detection of flares by decrease in physical activity, collected using wearable activity trackers in rheumatoid arthritis or axial spondyloarthritis: an application of machine learning analyses in rheumatology. Arthritis Care Res. 71, 1336–1343 (2019).
US Food and Drug Association. Proposed Regulatory Framework for Modifications to Artificial Intelligence/Machine Learning (AI/ML)-Based Software as a Medical Device (SaMD) - Discussion Paper and Request for Feedback. https://www.fda.gov/files/medical%20devices/published/US-FDA-Artificial-Intelligence-and-Machine-Learning-Discussion-Paper.pdf (2019).
Waltz, E. Pear approval signals FDA readiness for digital treatments. Nat. Biotechnol. 36, 481–482 (2018).
Christensen, D. R. et al. Adding an internet-delivered treatment to an efficacious treatment package for opioid dependence. J. Consult. Clin. Psychol. 82, 964–972 (2014).
Berman, M. A. et al. Change in glycemic control with use of a digital therapeutic in adults with type 2 diabetes: cohort study. JMIR Diabetes 3, e4 (2018).
Blakey, J. D. et al. Digital technologies and adherence in respiratory diseases: the road ahead. Eur. Respir. J. 52, 1801147 (2018).
Rogozinski, B., Greenleaf, W. Sackman, J. & Cahana, A. in Handbook of Pain and Palliative Care (ed. Moore. R.) 601–624 (Springer, 2018).
Abramoff, M. D., Lavin, P. T., Birch, M., Shah, N. & Folk, J. C. Pivotal trial of an autonomous AI-based diagnostic system for detection of diabetic retinopathy in primary care offices. NPJ Digit. Med. 1, 39 (2018).
He, J. et al. The practical implementation of artificial intelligence technologies in medicine. Nat. Med. 25, 30–36 (2019).
Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 25, 44–56 (2019).
Rajkomar, A., Dean, J. & Kohane, I. Machine learning in medicine. N. Engl. J. Med. 380, 1347–1358 (2019).
European Medicines Agency. Big data. https://www.ema.europa.eu/en/about-us/how-we-work/big-data (2019)
Kim, K. J. & Tagkopoulos, I. Application of machine learning in rheumatic disease research. Korean J. Intern. Med. 34, 708–722 (2019).
Orange, D. E. et al. Identification of three rheumatoid arthritis disease subtypes by machine learning integration of synovial histologic features and RNA sequencing data. Arthritis Rheumatol. 70, 690–701 (2018).
Lezcano-Valverde, J. M. et al. Development and validation of a multivariate predictive model for rheumatoid arthritis mortality using a machine learning approach. Sci. Rep. 7, 10189 (2017).
Norgeot, B. et al. Assessment of a deep learning model based on electronic health record data to forecast clinical outcomes in patients with rheumatoid arthritis. JAMA Netw. Open 2, e190606 (2019).
Kedra, J. et al. Current status of use of big data and artificial intelligence in RMDs: a systematic literature review informing EULAR recommendations. RMD Open 5, e001004 (2019).
Gossec, L. et al. EULAR points to consider for the use of big data in rheumatic and musculoskeletal diseases. Ann. Rheum. Dis. 79, 69–76 (2020).
Weiskopf, N. G. & Weng, C. Methods and dimensions of electronic health record data quality assessment: enabling reuse for clinical research. J. Am. Med. Inform. Assoc. 20, 144–151 (2013).
Herschman, J. et al. Development of a smartphone app for adolescents with lupus: a collaborative meeting-based methodology inclusive of a wide range of stakeholders. Rev. Panam. Salud Publica. 35, 471–476 (2014).
Dantas, L. O. et al. Mobile health technologies for the management of systemic lupus erythematosus: a systematic review. Lupus 29, 144–156 (2020).
Howren, A. et al. eHealth-supported decentralized multi-disciplinary care for gout involving rheumatology, pharmacy, and dietetics: proof-of-concept study. Clin. Rheumatol. 39, 1241–1249 (2019).
Jones, D., Skrepnik, N., Toselli, R. M. & Leroy, B. Incorporating novel mobile health technologies into management of knee osteoarthritis in patients treated with intra-articular hyaluronic acid: rationale and protocol of a randomized controlled trial. JMIR Res. Protoc. 5, e164 (2016).
Damberg, C. L. et al. Measuring success in health care value-based purchasing programs: findings from an environmental scan, literature review, and expert panel discussions. Rand Health Q. 4, 9 (2014).
Hernández, M. A., Sharit, J., Pirolli, P. & Czaja, S. J. Adapting information search tools for use by health consumers: challenges and lessons for software designers. Int. J. Human–Computer Interact. 34, 445–456 (2018).
Shneiderman, B. Designing the user interface: strategies for effective human–computer interaction (Addison-Wesley Longman, 1997).
Schnall, R. et al. A user-centered model for designing consumer mobile health (mHealth) applications (apps). J. Biomed. Inform. 60, 243–251 (2016).
McCurdie, T. et al. mHealth consumer apps: the case for user-centered design. Biomed. Instrum. Technol. 46 (Suppl. 2), 49–56 (2012).
Rudin, R. S., Bates, D. W. & MacRae, C. Accelerating innovation in health IT. N. Engl. J. Med. 375, 815–817 (2016).
Ratwani, R. M., Benda, N. C., Hettinger, A. Z. & Fairbanks, R. J. Electronic health record vendor adherence to usability certification requirements and testing standards. JAMA 314, 1070–1071 (2015).
Sarkar, U. et al. Usability of commercially available mobile applications for diverse patients. J. Gen. Intern. Med. 31, 1417–1426 (2016).
Najm, A. et al. EULAR points to consider for the development, evaluation and implementation of mobile health applications aiding self-management in people living with rheumatic and musculoskeletal diseases. RMD Open 5, e001014 (2019).
Mathur, P. et al. High level architecture and evaluation of patient linkages for READY – an electronic measurement tool for rheumatoid arthritis. Presented at the American Medical Informatics Association Annual Symposium (2015).
Pincus, T. et al. RAPID3 (Routine Assessment of Patient Index Data 3) severity categories and response criteria: similar results to DAS28 (Disease Activity Score) and CDAI (Clinical Disease Activity Index) in the RAPID 1 (Rheumatoid Arthritis Prevention of Structural Damage) clinical trial of certolizumab pegol. Arthritis Care Res. 63, 1142–1149 (2011).
Aletaha, D. & Smolen, J. S. The simplified disease activity index (SDAI) and clinical disease activity index (CDAI) to monitor patients in standard clinical care. Best. Prac. Res. Clin. Rheumatol. 21, 663–675 (2007).
Pincus, T., Summey, J. A., Soraci, S. A. Jr, Wallston, K. A. & Hummon, N. P. Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum. 26, 1346–1353 (1983).
Navarro-Millan, I. et al. Perspectives of rheumatoid arthritis patients on electronic communication and patient-reported outcome data collection: a qualitative study. Arthritis Care Res. 71, 80–87 (2019).
Solomon, D. H. et al. Patient adherence with a smartphone app for disease monitoring in rheumatoid arthritis [abstract THU0155]. Ann. Rheum. Dis. 78 (Suppl. 2), 350–351 (2019).
Rudin, R. S. et al. A clinically integrated mhealth app and practice model for collecting patient-reported outcomes between visits for asthma patients: implementation and feasibility. Appl. Clin. Inf. 10, 783–793 (2019).
Singh, K., Meyer, S. R. & Westfall, J. M. Consumer-facing data, information, and tools: self-management of health in the digital age. Health Aff. 38, 352–358 (2019).
US Department of Health and Human Services. HealthIT.gov https://www.healthit.gov/topic/laws-regulation-and-policy/notice-proposed-rulemaking-improve-interoperability-health (2020).
Yuan, B. & Li, J. The policy effect of the General Data Protection Regulation (GDPR) on the digital public health sector in the European Union: an empirical investigation. Int. J. Environ. Res. Public. Health 16, 1070 (2019).
Burmester, G. R. Rheumatology 4.0: big data, wearables and diagnosis by computer. Ann. Rheum. Dis. 77, 963–965 (2018).
Jadczyk, T. et al. Feasibility of a voice-enabled automated platform for medical data collection: CardioCube. Int. J. Med. Inform. 129, 388–393 (2019).
Chung, A. E., Griffin, A. C., Selezneva, D. & Gotz, D. Health and fitness apps for hands-free voice-activated assistants: content analysis. JMIR Mhealth Uhealth 6, e174 (2018).
Konig, A. et al. Automatic speech analysis for the assessment of patients with predementia and Alzheimer’s disease. Alzheimers Dement. 1, 112–124 (2015).
Erdogdu Sakar, B., Serbes, G. & Sakar, C. O. Analyzing the effectiveness of vocal features in early telediagnosis of Parkinson’s disease. PLoS ONE 12, e0182428 (2017).
He, L. & Cao, C. Automated depression analysis using convolutional neural networks from speech. J. Biomed. Inform. 83, 103–111 (2018).
Matcham, F., Rayner, L., Steer, S. & Hotopf, M. The prevalence of depression in rheumatoid arthritis: a systematic review and meta-analysis: reply. Rheumatology 53, 578–579 (2014).
Greenberg, A. Hackers can use lasers to ‘speak’ to your Amazon Echo or Google Home. Wired https://www.wired.com/story/lasers-hack-amazon-echo-google-home (2019).
Wakabayashi, D. & Wingfield, N. Alexa, we’re still trying to figure out what to do with you. New York Times https://www.nytimes.com/2018/01/15/technology/virtual-assistants-alexa.html (2018).
Acknowledgements
D.H.S. received support from NIH P30-AR072577 (VERITY) to support this review. He has also received research support for digital health technology development from Pfizer and Janssen. R.S.R. received support from AHRQ 1R18HS026432-01.
Author information
Authors and Affiliations
Contributions
D.H.S. researched data for article. Both authors substantially contributed to the discussion of content, wrote the article and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Rheumatology thanks A. Najm and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Center for Devices and Radiological Health: https://www.fda.gov/about-fda/office-medical-products-and-tobacco/center-devices-and-radiological-health
HelloRache: https://hellorache.com/
Mayo Clinic First Aid app: https://www.mayoclinic.org/voice/apps
Suki: https://www.suki.ai/
Glossary
- Artificial intelligence and machine learning
-
(AI/ML). Artificial intelligence is the broader concept of machines (computers or other) being able to carry out tasks in a way that we would consider ‘smart’. Machine learning is a current application of artificial intelligence based around the idea that machines with access to data can learn for themselves.
- Clinical decision support
-
A support system used in health information technology designed to provide clinicians with decision support around clinical issues. It usually involves accessing information in an electronic health record (for example, laboratory tests, diagnoses, medications, allergies and vaccination record) and uses logic based on medical guidelines.
- Best practice alerts
-
A type of clinical decision support used in many electronic health records, where the results of the clinical decision support are displayed as alerts to clinicians.
- Health Level Seven
-
(HL7). HL7 refers to standards for the transfer of data between software applications used by various health-care providers. The standards are most applicable to the software application level, which is described as ‘layer 7’.
- Fast Healthcare Interoperability Resources
-
(FHIR). A standard developed by Health Level 7 for describing data formats and elements and an application programming interface that facilitates exchange of electronic health record data.
- Health Insurance Portability and Accountability Act
-
An act signed into law by the US government in 1996 that was created to specify the appropriate flow of health-care information. It specifically stipulates how personally identifiable information should be maintained and protected from fraud and theft.
- Software as a Medical Device
-
(SaMD). Software intended to be used for medical purposes that performs its functions without being part of a hardware medical device.
- 510(k)
-
A premarket submission made to the FDA to demonstrate that a medical device (or digital health technology) to be marketed is at least as safe and substantially equivalent to a legally marketed device that is not subject to premarket approval.
Rights and permissions
About this article
Cite this article
Solomon, D.H., Rudin, R.S. Digital health technologies: opportunities and challenges in rheumatology. Nat Rev Rheumatol 16, 525–535 (2020). https://doi.org/10.1038/s41584-020-0461-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-020-0461-x
This article is cited by
-
Delivering Digital Health Solutions that Patients Need: A Call to Action
Therapeutic Innovation & Regulatory Science (2024)
-
Protocol for a systematic literature review of smartphone apps to support the self-management of rheumatic and musculoskeletal diseases: development strategies, theoretical underpinnings and barriers to engagement
Systematic Reviews (2023)
-
Willingness, perceived facilitators and barriers to use remote care among healthcare professionals – a cross-sectional study
BMC Health Services Research (2023)
-
Predictors for response to electronic patient-reported outcomes in routine care in patients with rheumatoid arthritis: a retrospective cohort study
Rheumatology International (2023)
-
Significance of Digital Health Technologies (DHTs) to manage communicable and non-communicable diseases in Low and Middle-Income Countries (LMICs)
Health and Technology (2023)