Abstract
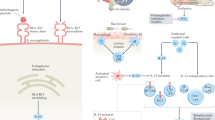
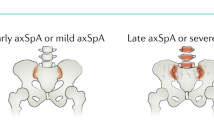
First-line therapy for spondyloarthritis (SpA) has not yet altered in the wake of new classification criteria; NSAIDs and physical therapy are recommended. Anti-TNF agents can be used when NSAIDs fail, but their efficacy has potentially been limited in previous trials by inclusion criteria requiring the presence of established, active disease. Now, not only patients with axial SpA (axSpA) with radiographic signs of sacroiliitis (that is, with ankylosing spondylitis), but also patients in whom structural damage is not—yet—visible radiographically (non-radiographic axSpA) can be included in trials of therapy for axSpA. TNF blockers, it seems already, are at least similarly effective in patients with non-radiographic axSpA as in those with established AS. Short symptom duration and a positive C-reactive protein test at baseline are currently the best predictors for a good response to TNF-blocking agents. Biologic agents besides anti-TNF therapies have so far failed in the treatment of axSpA. New bone formation seems currently to be best prevented by NSAIDs, not by TNF blockers. Whether earlier effective treatment of bony inflammation with anti-TNF therapy will be able to prevent ossification at a later stage has yet to be determined. New classification criteria for peripheral SpA will also allow treatment trials to be conducted more systematically than has previously been possible in this subgroup of patients.
Key Points
-
The term axial spondyloarthritis (axSpA) now includes patients with ankylosing spondylitis (radiographically visible sacroiliitis with or without syndesmophytes) as well as non-radiographic axSpA
-
Responses to TNF-blockers are similar between patients with non-radiographic and radiographic axSpA
-
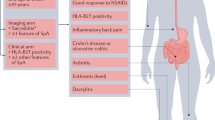
Short symptom duration and elevated C-reactive protein levels are the best predictors for a good response to anti-TNF therapy
-
Antibiotic treatment might be an interesting treatment option in a subgroup of patients with peripheral spondyloarthritis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
van Tubergen, A. & Weber, U. Diagnosis and classification in spondyloarthritis: identifying a chameleon. Nat. Rev. Rheumatol. http://dx.doi.org/10.1038/nrrheum.2012.33.
Rudwaleit, M. & Sieper, J. Referral strategies for an early diagnosis of axial spondyloarthritis. Nat. Rev. Rheumatol. http://dx.doi.org/10.1038/nrrheum.2012.39.
Tse, S. M. L. & Laxer, R. M. New advances in juvenile spondyloarthritis. Nat. Rev. Rheumatol. http://dx.doi.org/10.1038/nrrheum.2012.37.
Rudwaleit, M. et al. The development of Assessment of SpondyloArthritis international Society classification criteria for axial spondyloarthritis (part II): validation and final selection. Ann. Rheum. Dis. 68, 777–783 (2009).
Rudwaleit, M. et al. The Assessment of SpondyloArthritis International Society classification criteria for peripheral spondyloarthritis and for spondyloarthritis in general. Ann. Rheum. Dis. 70, 25–31 (2011).
Taylor, W. et al. Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum. 54, 2665–2673 (2006).
Braun, J. et al. 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann. Rheum. Dis. 70, 896–904 (2011).
Zochling, J. et al. ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann. Rheum. Dis. 65, 442–452 (2006).
Braun, J. et al. International ASAS consensus statement for the use of anti-tumour necrosis factor agents in patients with ankylosing spondylitis. Ann. Rheum. Dis. 62, 817–824 (2003).
Braun, J. et al. First update of the international ASAS consensus statement for the use of anti-TNF agents in patients with ankylosing spondylitis. Ann. Rheum. Dis. 65, 316–320 (2006).
van der Heijde, D. et al. 2010 Update of the international ASAS recommendations for the use of anti-TNF agents in patients with axial spondyloarthritis. Ann. Rheum. Dis. 70, 905–908 (2011).
van der Linden, S., Valkenburg, H. A. & Cats, A. Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum. 27, 361–368 (1984).
Sieper, J. & Rudwaleit, M. How early should ankylosing spondylitis be treated with tumour necrosis factor blockers? Ann. Rheum. Dis. 64 (Suppl. 4), iv61–iv64 (2005).
van der Heijde, D. et al. Efficacy and safety of adalimumab in patients with ankylosing spondylitis: results of a multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 54, 2136–2146 (2006).
Haibel, H. et al. Efficacy of adalimumab in the treatment of axial spondylarthritis without radiographically defined sacroiliitis: results of a twelve-week randomized, double-blind, placebo-controlled trial followed by an open-label extension up to week fifty-two. Arthritis Rheum. 58, 1981–1991 (2008).
Barkham, N. et al. Clinical and imaging efficacy of infliximab in HLA-B27-Positive patients with magnetic resonance imaging-determined early sacroiliitis. Arthritis Rheum. 60, 946–954 (2009).
Song, I. H. et al. Effects of etanercept versus sulfasalazine in early axial spondyloarthritis on active inflammatory lesions as detected by whole-body MRI (ESTHER): a 48-week randomised controlled trial. Ann. Rheum. Dis. 70, 590–596 (2011).
Certolizumab pegol in subjects with active axial spondyloarthritis. ClinicalTrials.gov [online], (2012).
Study comparing etanercept (ETN) against a placebo for etanercept on a background nonsteroidal anti inflammatory drug (NSAIDs) in the treatment of early spondyloarthritis (SpA) patients who do not have X-ray structural changes (AS EARLY). ClinicalTrials.gov [online], (2012).
Effect of golimumab in participants with active axial spondyloarthritis (P07642) (GO-AHEAD). ClinicalTrials.gov [online], (2012).
Sieper, J. et al. Efficacy and safety of adalimumab in patients with non-radiographic axial spondyloarthritis—results of the phase 3 study [abstract 2486A]. Arthritis Rheum. 63 (Suppl.), S970–S971 (2011).
Baraliakos, X. et al. Persistent clinical efficacy and safety of infliximab in ankylosing spondylitis after 8 years—early clinical response predicts long-term outcome. Rheumatology (Oxford) 50, 1690–1699 (2011).
Martin-Mola, E. et al. Sustained efficacy and safety, including patient-reported outcomes, with etanercept treatment over 5 years in patients with ankylosing spondylitis. Clin. Exp. Rheumatol. 28, 238–245 (2010).
Sieper, J. et al. Early response to adalimumab predicts long-term remission through 5 years of treatment in patients with ankylosing spondylitis. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2011-200358.
Sieper, J. et al. Persistent reduction of spinal inflammation as assessed by magnetic resonance imaging in patients with ankylosing spondylitis after 2 yrs of treatment with the anti-tumour necrosis factor agent infliximab. Rheumatology (Oxford) 44, 1525–1530 (2005).
Braun, J. et al. Golimumab reduces spinal inflammation in ankylosing spondylitis: MRI results of the randomised, placebo- controlled GO-RAISE study. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2011-200308
Rudwaleit, M., Listing, J., Brandt, J., Braun, J. & Sieper, J. Prediction of a major clinical response (BASDAI 50) to tumour necrosis factor α blockers in ankylosing spondylitis. Ann. Rheum. Dis. 63, 665–670 (2004).
Rudwaleit, M. et al. Effectiveness, safety, and predictors of good clinical response in 1250 patients treated with adalimumab for active ankylosing spondylitis. J. Rheumatol. 36, 801–808 (2009).
Vastesaeger, N. et al. Predicting the outcome of ankylosing spondylitis therapy. Ann. Rheum. Dis. 70, 973–981 (2011).
Rudwaleit, M. et al. MRI in predicting a major clinical response to anti-tumour necrosis factor treatment in ankylosing spondylitis. Ann. Rheum. Dis. 67, 1276–1281 (2008).
de Vries, M. K. et al. Decreased clinical response to infliximab in ankylosing spondylitis is correlated with anti-infliximab formation. Ann. Rheum. Dis. 66, 1252–1254 (2007).
de Vries, M. K. et al. Decreased clinical response to adalimumab in ankylosing spondylitis is associated with antibody formation. Ann. Rheum. Dis. 68, 1787–1788 (2009).
Baert, F. et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn's disease. N. Engl. J. Med. 348, 601–608 (2003).
Breban, M. et al. Maintenance of infliximab treatment in ankylosing spondylitis: results of a one-year randomized controlled trial comparing systematic versus on-demand treatment. Arthritis Rheum. 58, 88–97 (2008).
Li, E. K. et al. Short-term efficacy of combination methotrexate and infliximab in patients with ankylosing spondylitis: a clinical and magnetic resonance imaging correlation. Rheumatology (Oxford) 47, 1358–1363 (2008).
Mulleman, D. et al. Infliximab in ankylosing spondylitis: alone or in combination with methotrexate? A pharmacokinetic comparative study. Arthritis Res. Ther. 13, R82 (2011).
Rudwaleit, M., Van den Bosch, F., Kron, M., Kary, S. & Kupper, H. Effectiveness and safety of adalimumab in patients with ankylosing spondylitis or psoriatic arthritis and history of anti-tumor necrosis factor therapy. Arthritis Res. Ther. 12, R117 (2010).
Lie, E. et al. Effectiveness of switching between TNF inhibitors in ankylosing spondylitis: data from the NOR-DMARD register. Ann. Rheum. Dis. 70, 157–163 (2011).
Haibel, H., Rudwaleit, M., Listing, J. & Sieper, J. Open label trial of anakinra in active ankylosing spondylitis over 24 weeks. Ann. Rheum. Dis. 64, 296–298 (2005).
Song, I. H. et al. Different response to rituximab in tumor necrosis factor blocker-naive patients with active ankylosing spondylitis and in patients in whom tumor necrosis factor blockers have failed: a twenty-four-week clinical trial. Arthritis Rheum. 62, 1290–1297 (2010).
Song, I. H. et al. Treatment of active ankylosing spondylitis with abatacept: an open-label, 24-week pilot study. Ann. Rheum. Dis. 70, 1108–1110 (2011).
Lekpa, F. K. et al. Lack of efficacy of abatacept in axial spondylarthropathies refractory to tumor-necrosis-factor inhibition. Joint Bone Spine 79, 47–50 (2012).
A study of RoActemra/Actemra (tocilizumab) in patients with ankylosing spondylitis who have failed treatment with NSAIDs. ClinicalTrials.gov [online], (2011).
A study of RoActemra/Actemra (tocilizumab) in patients with ankylosing spondylitis who have had an inadequate response to previous TNF antagonist therapy. ClinicalTrials.gov [online], (2011).
Appel, H. et al. Analysis of IL-17+ cells in facet joints of patients with spondyloarthritis suggests that the innate immune pathway might be of greater relevance than the TH17-mediated adaptive immune response. Arthritis Res. Ther. 13, R95 (2011).
Griffiths, C. E. et al. Comparison of ustekinumab and etanercept for moderate-to-severe psoriasis. N. Engl. J. Med. 362, 118–128 (2010).
Hueber, W. et al. Effects of AIN457, a fully human antibody to interleukin-17A, on psoriasis, rheumatoid arthritis, and uveitis. Sci. Transl. Med. 2, 52ra72 (2010).
Baeten, D. et al. The anti-IL-17A monoclonal antibody secukinumab (AIN457) showed good safety and efficacy in the treatment of active ankylosing spondylitis. Ann. Rheum. Dis. 70 (Suppl. 3), 127 (2011).
Ustekinumab for the treatment of patients with active ankylosing spondylitis (TOPAS). ClinicalTrials.gov [online], (2011).
Landewe, R., Dougados, M., Mielants, H., van der Tempel, H. & van der Heijde, D. Physical function in ankylosing spondylitis is independently determined by both disease activity and radiographic damage of the spine. Ann. Rheum. Dis. 68, 863–867 (2009).
Wanders, A. et al. Nonsteroidal antiinflammatory drugs reduce radiographic progression in patients with ankylosing spondylitis: a randomized clinical trial. Arthritis Rheum. 52, 1756–1765 (2005).
Boersma, J. W. Retardation of ossification of the lumbar vertebral column in ankylosing spondylitis by means of phenylbutazone. Scand. J. Rheumatol. 5, 60–64 (1976).
Poddubnyy, D. et al. Effect of non-steroidal anti-inflammatory drugs on radiographic progression in patients with axial spondyloartrhitis: results from the German Spondyloarthritis Inception Cohort. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2011-201252
Dougados, M. et al. ASAS recommendations for collecting, analysing and reporting NSAID intake in clinical trials/epidemiological studies in axial spondyloarthritis. Ann. Rheum. Dis. 70, 249–251 (2011).
Blackwell, K. A., Raisz, L. G. & Pilbeam, C. C. Prostaglandins in bone: bad cop, good cop? Trends Endocrinol. Metab. 21, 294–301 (2010).
van der Heijde, D. et al. Radiographic findings following two years of infliximab therapy in patients with ankylosing spondylitis. Arthritis Rheum. 58, 3063–3070 (2008).
van der Heijde, D. et al. Radiographic progression of ankylosing spondylitis after up to two years of treatment with etanercept. Arthritis Rheum. 58, 1324–1331 (2008).
van der Heijde, D. et al. Assessment of radiographic progression in the spines of patients with ankylosing spondylitis treated with adalimumab for up to 2 years. Arthritis Res. Ther. 11, R127 (2009).
Rudwaleit, M. et al. Defining active sacroiliitis on magnetic resonance imaging (MRI) for classification of axial spondyloarthritis: a consensual approach by the ASAS/OMERACT MRI group. Ann. Rheum. Dis. 68, 1520–1527 (2009).
Song, I. H. et al. Relationship between active inflammatory lesions in the spine and sacroiliac joints and new development of chronic lesions on whole-body MRI in early axial spondyloarthritis: results of the ESTHER trial at week 48. Ann. Rheum. Dis. 70, 1257–1263 (2011).
Chiowchanwisawakit, P., Lambert, R. G., Conner-Spady, B. & Maksymowych, W. P. Focal fat lesions at vertebral corners on magnetic resonance imaging predict the development of new syndesmophytes in ankylosing spondylitis. Arthritis Rheum. 63, 2215–2225 (2011).
Sieper, J., Appel, H., Braun, J. & Rudwaleit, M. Critical appraisal of assessment of structural damage in ankylosing spondylitis: implications for treatment outcomes. Arthritis Rheum. 58, 649–656 (2008).
Appel, H., Rudwaleit, M. & Sieper, J. Relevance of osteoproliferation as an outcome parameter in ankylosing spondylitis. Nat. Clin. Pract. Rheumatol. 4, 578–579 (2008).
Maksymowych, W. P. et al. Inflammatory lesions of the spine on magnetic resonance imaging predict the development of new syndesmophytes in ankylosing spondylitis: evidence of a relationship between inflammation and new bone formation. Arthritis Rheum. 60, 93–102 (2009).
Dougados, M. et al. Sulfasalazine in the treatment of spondylarthropathy. A randomized, multicenter, double-blind, placebo-controlled study. Arthritis Rheum. 38, 618–627 (1995).
Clegg, D. O. et al. Comparison of sulfasalazine and placebo in the treatment of ankylosing spondylitis. A Department of Veterans Affairs cooperative study. Arthritis Rheum. 39, 2004–2012 (1996).
Braun, J. et al. Treatment of active ankylosing spondylitis with infliximab: a randomised controlled multicentre trial. Lancet 359, 1187–1193 (2002).
Rudwaleit, M. et al. Effectiveness of adalimumab in treating patients with ankylosing spondylitis associated with enthesitis and peripheral arthritis. Arthritis Res. Ther. 12, R43 (2010).
Dougados, M. et al. A randomised, multicentre, double-blind, placebo-controlled trial of etanercept in adults with refractory heel enthesitis in spondyloarthritis: the HEEL trial. Ann. Rheum. Dis. 69, 1430–1435 (2010).
Gossec, L. et al. European League Against Rheumatism recommendations for the management of psoriatic arthritis with pharmacological therapies. Ann. Rheum. Dis. 71, 4–12 (2012).
Sieper, J. et al. No benefit of long-term ciprofloxacin treatment in patients with reactive arthritis and undifferentiated oligoarthritis: a three-month, multicenter, double-blind, randomized, placebo-controlled study. Arthritis Rheum. 42, 1386–1396 (1999).
Kvien, T. K. Three month treatment of reactive arthritis with azithromycin: a EULAR double blind, placebo controlled study. Ann. Rheum. Dis. 63, 1113–1119 (2004).
Lauhio, A., Leirisalo-Repo, M., Lahdevirta, J., Saikku, P. & Repo, H. Double-blind, placebo-controlled study of three-month treatment with lymecycline in reactive arthritis, with special reference to Chlamydia arthritis. Arthritis Rheum. 34, 6–14 (1991).
Carter, J. D. et al. Chlamydiae as etiologic agents in chronic undifferentiated spondyloarthritis. Arthritis Rheum. 60, 1311–1316 (2009).
Carter, J. D. et al. Combination antibiotics as a treatment for chronic Chlamydia-induced reactive arthritis: a double-blind, placebo-controlled, prospective trial. Arthritis Rheum. 62, 1298–1307 (2010).
Braun, J., Baraliakos, X., Listing, J. & Sieper, J. Decreased incidence of anterior uveitis in patients with ankylosing spondylitis treated with the anti-tumor necrosis factor agents infliximab and etanercept. Arthritis Rheum. 52, 2447–2451 (2005).
Rudwaleit, M. et al. Adalimumab effectively reduces the rate of anterior uveitis flares in patients with active ankylosing spondylitis: results of a prospective open-label study. Ann. Rheum. Dis. 68, 696–701 (2009).
Sieper, J. et al. Analysis of uveitis rates across all etanercept ankylosing spondylitis clinical trials. Ann. Rheum. Dis. 69, 226–229 (2010).
Wendling, D. et al. New onset of uveitis during anti-tumor necrosis factor treatment for rheumatic diseases. Semin. Arthritis Rheum. 41, 503–510 (2011).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
J. Sieper has worked as a consultant for Abbott, Merck, Pfizer, Roche and UCB, has been a member of a speakers bureau for Abbott, Merck, Pfizer and UCB, and has received grant or research support from Abbott, Bristol-Myers Squibb, Merck, Pfizer and Roche.
Rights and permissions
About this article
Cite this article
Sieper, J. Developments in therapies for spondyloarthritis. Nat Rev Rheumatol 8, 280–287 (2012). https://doi.org/10.1038/nrrheum.2012.40
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2012.40
This article is cited by
-
Spondyloarthritis
Wiener klinische Wochenschrift Education (2017)
-
TNF-α Promoter Polymorphisms Predict the Response to Etanercept More Powerfully than that to Infliximab/Adalimumab in Spondyloarthritis
Scientific Reports (2016)
-
New evidence on the management of spondyloarthritis
Nature Reviews Rheumatology (2016)
-
Nichtradiographische axiale Spondyloarthritis
Manuelle Medizin (2016)
-
Regulation of tumour necrosis factor signalling: live or let die
Nature Reviews Immunology (2015)