Abstract
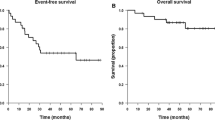
Meningiomas represent 18–20% of all intracranial tumors and have a 10-year recurrence rate of 20–50%, despite aggressive surgery and irradiation. In addition, many tumors are not amenable to surgery due to their deep location or proximity to delicate structures. Chemotherapy is being explored as another potential treatment option for unresectable or refractory meningiomas. Hydroxyurea is an agent that inhibits ribonucleotide reductase and can induce apoptosis in meningioma cell cultures and animal models. We have placed 17 patients with unresectable or residual meningioma on hydroxyurea chemotherapy (20 mg/kg/d orally). The mean age of our cohort was 57.2 years; 13 patients were female. Eleven patients had actively growing tumors or neurological progression at the onset of chemotherapy. Sixteen patients were evaluable for response. Fourteen of the 16 patients (88%) responded with stable disease ranging from 20 to 144+ weeks (median 80 weeks; 10 patients still accruing time). Three of the responders progressed after 20, 36, and 56 weeks, respectively. Two patients had progressive disease after 10 weeks. Toxicity was hematologic in most patients; leukopenia was most common. Nine patients (53%) required dosage reductions (250–500 mg/d) secondary to hematologic toxicity. Hydroxyurea appears to have modest activity against meningiomas and should be considered in patients with unresectable tumors or large residual tumors following surgical resection.
Similar content being viewed by others
References
Rohringer M, Sutherland GR, Louw DF, Sima AAF: Incidence and clinicopathological features of meningioma. J Neurosurg 71: 665-672, 1989
Longstreth WT, Dennis LK, McGuire VM, Drangsholt MT, Koepsell TD: Epidemiology of intracranial meningioma. Cancer 72: 639-648, 1993
Bruner JM, Tien RD, Enterline DS: Tumors of the meninges and related tissues. In: Russell & Rubinstein's Pathology of Tumors of the Nervous System. 6th edn, Bigner DD, McLendon RE, Bruner JM (eds). Arnold Press, London, 1998, pp 67-139
De Monte F: Current management of meningiomas. Oncol 9: 83-96, 1995
Akeyson EW, McCutcheon IE: Management of benign and aggressive intracranial meningiomas. Oncol 10: 747-756, 1996
Mirimanoff RO, Dosoretz DE, Linggood RM, Ojemann RG, Martuza RL: Meningioma: analysis of recurrence and progression following neurosurgical resection. J Neurosurg 62: 18-24, 1985
Miralbell R, Linggood RM, de al Monte S, Convery K, Munzenrider JE, Mirimanoff RO: The role of radiotherapy in the treatment of subtotally resected benign meningiomas. J Neuro-Oncol 13: 157-164, 1992
Dzuik TW, Woo S, Butler EB, Thornby J, Grossman R, Dennis WS, Lu H, Carpenter LS, Chiu JK: Malignant meningioma: an indication for initial aggressive surgery and adjuvant radiotherapy. J Neuro-Oncol 37: 177-188, 1998
Kondziolka D, Levy EI, Niranjan A, Flickinger JC, Lunsford LD: Long-term outcomes after meningioma radiosurgery: physician and patient outcomes. J Neurosurg 91: 44-50, 1999
Grunberg SM, Weiss MH, Spitz IM, Ahmadi J, Sadun A, Russell CA, Lucci L, Stevenson LL: Treatment of unresectable meningiomas with the antiprogesterone agent mifepristone. J Neurosurg 74: 861-866, 1991
Lamberts SWJ, Tanghe HLJ, Avezaata CJJ, Braakman R, Wijngaarde R, Koper JW, de Jong FH: Mifepristone (RU 486) treatment of meningiomas. J Neurol Neurosurg Psych 55: 486-490, 1992
Newton HB, Turowski RC, Stroup TJ, McCoy LK: Clinical presentation, diagnosis, and pharmacotherapy of patients with primary brain tumors. Ann Pharmacother 33: 816-832, 1999
Schrell UMH, Rittig MG, Anders M, Kiesewetter F, Marschalek R, Koch UH, Fahlbusch R: Hydroxyurea for treatment of unresectable and recurrent meningiomas. I. Inhibition of primary human meningioma cells in culture and in meningioma transplants by induction of the apoptotic pathway. J Neurosurg 86: 845-852, 1997
Schrell UMH, Rittig MG, Anders M, Koch UH, Marschalek R, Kiesewetter F, Fahlbusch R: Hydroxyurea for treatment of unresectable and recurrent meningiomas. II. Decrease in the size of meningiomas in patients treated with hydroxyurea. J Neurosurg 86: 840-844, 1997
National Cancer Institute: Common Toxicity Criteria. Bethesda, MD, NCI, 1998
Macdonald DR, Cascino TL, Schold SC, Cairncross JG: Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8: 1277-1280, 1990
Martin DS, Stolfi RL, Colofiore JR. Perspective: the chemotherapeutic relevance of apoptosis and a proposed biochemical cascade for chemotherapeutically induced apoptosis. Cancer Investig 15: 372-381, 1997
Stewart BW: Mechanisms of apoptosis: integration of genetic, biochemical, and cellular indicators. J Natl Cancer Inst 86: 1286-1296, 1994
Mason WP, Abrey L, Hariharan S. Hydroxyurea for treatment of unresectable and recurrent meningiomas: preliminary report of 16 patients (abstract). Neuro-Oncol 1: S39, 1999
Hryniuk WM: Average relative dose intensity and the impact on design of clinical trials. Semin Oncol 14: 65-74, 1987
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Newton, H.B., Slivka, M.A. & Stevens, C. Hydroxyurea Chemotherapy for Unresectable or Residual Meningioma. J Neurooncol 49, 165–170 (2000). https://doi.org/10.1023/A:1026770624783
Issue Date:
DOI: https://doi.org/10.1023/A:1026770624783