Abstract
Introduction
Idelalisib is a selective inhibitor of the delta isoform of phosphatidylinositol 3-kinase which was approved by the United States Federal Drug Administration in 2014 for the treatment of relapsed chronic lymphocytic leukemia and indolent non-Hodgkin lymphoma. Drug-induced injury of the gastrointestinal tract is a relatively frequent but usually under-recognized disease entity.
Case presentation
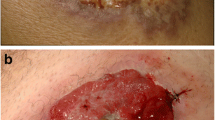
We report the case of a 56-year-old male with a history of relapsed follicular lymphoma status post allogenic bone marrow transplant who developed severe diarrhea with a skin eruption mimicking graft-versus-host disease (GVHD) 6 months after starting idelalisib. He underwent a colonoscopy demonstrating a grossly normal-appearing colon and terminal ileum. Biopsies taken during the procedure revealed mild active ileitis, colitis, and proctitis with frequent epithelial apoptosis, and focal intra-epithelial lymphocytosis. Skin biopsies revealed sub-acute spongiotic dermatitis suggestive of either contact dermatitis or an eczematous drug reaction. Symptoms were attributed to idelalisib given their resolution with withdrawal of the drug in conjunction with the skin and colonic biopsies.
Conclusion
High clinical suspicion and awareness of the histological features of idelalisib-associated colitis is important to distinguish it from potential mimickers such as GVHD and infectious colitis.



Similar content being viewed by others
References
Madanat YF, Smith MR, Almasan A, et al. Idelalisib therapy of indolent B-cell malignancies: chronic lymphocytic leukemia and small lymphocytic or follicular lymphomas. Blood Lymphat Cancer. 2016;6:1–6.
O’Brien SM, Lamanna N, Kipps TJ, et al. A phase 2 study of idelalisib plus rituximab in treatment-naive older patients with chronic lymphocytic leukemia. Blood. 2015;126(25):2686–94.
Kahl BS, Spurgeon SE, Furman RR, et al. A phase 1 study of the PI3Kdelta inhibitor idelalisib in patients with relapsed/refractory mantle cell lymphoma (MCL). Blood. 2014;123(22):3398–405.
Villanacci V, Manenti S, Antonelli E, et al. Non-IBD colitides: clinically useful histopathological clues. Rev Esp Enferm Dig. 2011;103(7):366–72.
Lee FD. Drug-related pathological lesions of the intestinal tract. Histopathology. 1994;25(4):303–8.
Villanacci V, Casella G, Bassotti G. The spectrum of drug-related colitides: important entities, though frequently overlooked. Dig Liver Dis. 2011;43(7):523–8. doi:10.1016/j.dld.2010.12.016.
Coutre SE, Barrientos JC, Brown JR, et al. Management of adverse events associated with idelalisib treatment: expert panel opinion. Leuk Lymphoma. 2015;56(10):2779–86. doi:10.3109/10428194.2015.1022770.
Uno JK, Rao KN, Matsuoka K, et al. Altered macrophage function contributes to colitis in mice defective in the phosphoinositide-3 kinase subunit p110delta. Gastroenterology. 2010;139(5):1642–53. doi:10.1053/j.gastro.2010.07.008.
Weidner AS, Panarelli NC, Geyer JT, et al. Idelalisib-associated colitis: histologic findings in 14 patients. Am J Surg Pathol. 2015;39(12):1661–7. doi:10.1097/PAS.0000000000000522.
Balagoni H, Chaudhari D, Reddy C, et al. Idelalisib: a rare cause of enterocolitis. Ann Gastroenterol. 2016;29(2):233–5. doi:10.20524/aog.2016.0022.
Louie CY, DiMaio MA, Matsukuma KE, et al. Idelalisib-associated enterocolitis: clinicopathologic features and distinction from other enterocolitides. Am J Surg Pathol. 2015;39(12):1653–60. doi:10.1097/PAS.0000000000000525.
Kohler S, Hendrickson MR, Chao NJ, et al. Value of skin biopsies in assessing prognosis and progression of acute graft-versus-host disease. Am J Surg Pathol. 1997;21(9):988–96.
Chao SC, Tsao CJ, Liu CL, et al. Acute cutaneous graft-versus-host disease with ichthyosiform features. Br J Dermatol. 1998;139(3):553–5.
Acknowledgements
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare that they have no conflict of interest.
Human Rights:
All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed Consent:
Informed consent was obtained from all patients for being included in the study.
Rights and permissions
About this article
Cite this article
Hammami, M.B., Al-Taee, A., Meeks, M. et al. Idelalisib-induced colitis and skin eruption mimicking graft-versus-host disease . Clin J Gastroenterol 10, 142–146 (2017). https://doi.org/10.1007/s12328-016-0707-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-016-0707-y