Abstract
Introduction
The peptide receptor radionuclide therapy (PRRT) with 90Y and 177Lu is a form of molecular targeted therapy for inoperable or disseminated neuroendocrine tumors (NET).
Aim
The aim of the study was to evaluate clinical results and long-term side effects of tandem 90Y /177Lu-DOTATATE therapy in patients with NET. Additionally, we evaluated clinical results with reference to the primary site.
Materials and methods
59 patients with disseminated NET were included in the study prospectively. 3–5 cycles of combined 1:1 90Y/177Lu-DOTATATE (total injected activity 11.1–16.65 GBq) with mixed amino acids for kidney protection were performed.
Results
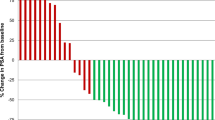
During a median follow-up of 75.8 months, the PFS was 32.2 months, and the OS was 82 months; 25 patients died. Depending on primary tumor’s site, the PFS and the OS for pancreatic NET vs. small bowel, NET vs. large bowel, NET were 30.4 vs. 29.5 vs. 40.3 and 78.9 vs. 58.1 vs. 82.5, respectively. The observed 5-year overall survival was 63%, and a 2-year risk of progression was 39.4%. The following imaging response was observed: CR in 2%, PR in 22%, SD in 65%, and PD in 6% patients. The disease control rate was 89%. The objective response rate was 24%. The PRRT was well tolerated by all patients. One patient (2%) revealed MDS, and one patient (2%) grade 3 nephrotoxicity. No other grade 3 and 4 hematological or renal toxicity was observed.
Conclusions
These results indicated the tandem 90Y/177Lu-DOTATATE therapy for patients with disseminated/inoperable NET as highly effective and safe, considering long-term side effects. In the majority of patients, clinical improvement was observed.





Similar content being viewed by others
References
Kwekkeboom D, Krenning EP, de Jong. Peptide receptor imaging and therapy. J Nucl Med. 2000;41:1704–13.
De Jong M, Bernard HF, Breeman WA, Bernard BF, Krenning EP. Combination of 90Y- and 177Lu-labeled somatostatin analogs is superior for radionuclide therapy compared to 90Y- or 177Lu-labeled analogs only. J Nucl Med. 2002;43:123P–124P.
de Jong M, Breeman WA, Valkema R, Bernard BF, Krenning EP. Combination radionuclide therapy using 177Lu- and 90Y-labeled somatostatin analogs. J Nucl Med. 2005;46:13S–17S.
Kunikowska J, Królicki L, Hubalewska-Dydejczyk A, Mikołajczak R, Sowa-Staszczak A, Pawlak D. Clinical results of radionuclide therapy of neuroendocrine tumours with 90Y-ATATE and tandem 90Y/177Lu-ATATE: which is a better therapy option? Eur J Nucl Med Mol Imaging. 2011;38:1788–97.
Rolleman EJ, Valkema R, de Jong M, Kooij PP, Krenning EP. Safe and effective inhibition of renal uptake of radiolabelled octreotide by a combination of lysine and arginine. Eur J Nucl Med Mol Imaging. 2003;30:9–15.
Bodei L, Mueller-Brand J, Baum RP, Pavel ME, Hörsch D, O’Dorisio MS, et al. The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2013;40:800–16. Erratum in: Eur J Nucl Med Mol Imaging. 2014; 41:584.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81.
Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, et al. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors. N Engl J Med. 2017;376:125–35.
Siegel JA, Stabin MG. Absorbed fractions for electrons and beta particles in spheres of various sizes. J Nucl Med. 1994;35:152–6.
Villard L, Romer A, Marincek N, Brunner P, Koller MT, Schindler C, et al. Cohort study of somatostatin-based radiopeptide therapy with [(90)Y-.A]-TOC versus [(90)Y-.A]-TOC plus [(177)Lu-.A]-TOC in neuroendocrine cancers. J Clin Oncol. 2012;30:1100–6.
Dumont RA, Seiler D, Marincek N, Brunner P, Radojewski P, Rochlitz C, et al. Survival after somatostatin based radiopeptide therapy with (90)Y-.ATOC vs. (90)Y-.ATOC plus (177)Lu-.ATOC in metastasized gastrinoma. Am J Nucl Med Mol Imaging. 2014;5:46–55.
Horsch D, Ezziddin S, Haug A, Gratz KF, Dunkelmann S, Miederer M, et al. Effectiveness and side-effects of peptide receptor radionuclide therapy for neuroendocrine neoplasms in Germany: a multi-institutional registry study with prospective follow-up. Eur J Cancer. 2016;58:41–51.
Radojewski P, Dumont R, Marincek N, Brunner P, Mäcke HR, Müller-Brand J, et al. Towards tailored radiopeptide therapy. Eur J Nucl Med Mol Imaging. 2015;42:1231–7.
Rinke A, Muller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27:4656–63.
Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371:224–33.
Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas C, et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N Engl J Med 2011; 364: 501–513. Erratum. N Engl J Med. 2011;364:1082.
Yao JC, Shah MH, Ito T Bohas CL, Wolin EM, Van Cutsem E, et al. RAD001 in Advanced Neuroendocrine Tumors, Third Trial (RADIANT-3) Study Group. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:514–23.
Kvols LK, Buck M. Chemotherapy of endocrine malignancies: a review. Semin Oncol. 1987;14:343–53.
Ezziddin S, Khalaf F, Vanezi M, Haslerud T, Mayer K, Al Zreiqat A, et al. Outcome of peptide receptor radionuclide therapy with 177Lu-octreotate in advanced grade 1/2 pancreatic neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2014;41:925–33.
Pfeifer AK, Gregersen T, Grønbæk H, Hansen CP, Müller-Brand J, Herskind Bruun K, et al. Peptide receptor radionuclide therapy with Y-.ATOC and (177)Lu-.ATOC in advanced neuroendocrine tumors: results from a Danish cohort treated in Switzerland. Neuroendocrinology. 2011;93:189–96.
Waldherr C, Pless M, Maecke HR, Haldemann A, Mueller-Brand J. The clinical value of [90Y-.A]-D-Phe1-Tyr3-octreotide (90Y-.ATOC) in the treatment of neuroendocrine tumours: a clinical phase II study. Ann Oncol. 2001;12:941–5.
Forrer F, Waldherr C, Maecke HR, Mueller-Brand J. Targeted radionuclide therapy with 90Y-.ATOC in patients with neuroendocrine tumors. Anticancer Res. 2006;26:703–7.
Valkema R, Pauwels S, Kvols LK, Barone R, Jamar F, Bakker WH, et al. Survival and response after peptide receptor radionuclide therapy with [90Y-.A0,Tyr3]octreotide in patients with advanced gastroenteropancreatic neuroendocrine tumors. Semin Nucl Med. 2006;36:147–56.
Paganelli G, Sansovini M, Ambrosetti A, Severi S, Monti M, Scarpi E, et al. 177Lu-Dota-octreotate radionuclide therapy of advanced gastrointestinal neuroendocrine tumors: results from a phase II study. Eur J Nucl Med Mol Imaging. 2014;41:1845–51.
Delpassand ES, Samarghandi A, Zamanian S, Wolin EM, Hamiditabar M, Espenan GD, et al. Peptide receptor radionuclide therapy with 177Lu-.ATATE for patients with somatostatin receptor-expressing neuroendocrine tumors: the first US phase 2 experience. Pancreas. 2014;43:518–25.
Imhof A, Brunner P, Marincek N, Briel M, Schindler C, Rasch H, et al. Response, survival, and long-term toxicity after therapy with the radiolabeled somatostatin analogue [90Y-.A]-TOC in metastasized neuroendocrine cancers. J Clin Oncol. 2011;29:2416–23.
Bodei L, Kidd M, Paganelli G, Grana CM, Drozdov I, Cremonesi M, et al. Long-term tolerability of PRRT in 807 patients with neuroendocrine tumours: the value and limitations of clinical factors. Eur J Nucl Med Mol Imaging. 2015;42:5–19.
Kwekkeboom DJ, de Herder WW, Kam BL, Eijck CH, van Essen M, Kooij PP, et al. Treatment with the radiolabeled somatostatin analog [177Lu-.A0 ,Tyr3]octreotate: toxicity, efficacy, and survival. J Clin Oncol. 2008;26:2124–30.
Kos-Kudła B, Blicharz-Dorniak J, Handkiewicz-Junak D Jarząb B, Jarząb M, Kunikowska J, et al. Diagnostic and therapeutic guidelines for gastro-entero-pancreatic neuroendocrine neoplasms (recommended by the Polish Network of Neuroendocrine Tumours). Endokrynol Pol. 2013;64:418–43.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kunikowska, J., Pawlak, D., Bąk, M.I. et al. Long-term results and tolerability of tandem peptide receptor radionuclide therapy with 90Y/177Lu-DOTATATE in neuroendocrine tumors with respect to the primary location: a 10-year study. Ann Nucl Med 31, 347–356 (2017). https://doi.org/10.1007/s12149-017-1163-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-017-1163-6