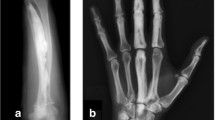
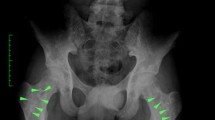
Abstract
Purpose of Review
Melorheostosis is a rare sclerosing bone dysplasia that affects both cortical bone and adjacent soft tissue structures in a sclerotomal distribution. In this review, we describe the natural history, radiological features, proposed pathogenesis, and management options for this debilitating condition.
Recent Findings
Since its first description in 1922, about 400 cases of melorheostosis have been reported, either as single reports or in small case series. Melorheostosis affects the appendicular skeleton more commonly than the axial skeleton and usually presents with lower limb deformity. Diagnosis is based on a combination of clinical and radiological features that help differentiate this condition from other sclerosing bone dysplasias. LEM domain-containing protein 3 (LEMD3) gene mutations have been demonstrated in several familial cases, but these have been more strongly correlated with other hereditary dysplasias, such as osteopoikilosis, and are not thought to be the causative gene for melorheostosis. The exact etiology of classic sporadically occurring melorheostosis remains unknown, with possible causes being somatic LEMD3 mutations, somatic mutations in the bone morphogenetic protein/transforming growth factor-beta pathway, mutations in multiple genes, or other non-genetic causes. Management in recent years has involved nitrogen-containing bisphosphonates in addition to traditional orthopedic surgical approaches and physical therapy.
Summary
Melorheostosis may present as mixed or atypical osseous involvement in addition to the classically described “dripping candle wax” appearance of hyperostosis. Some patients may have overlap with osteopoikilosis or Buschke–Ollendorff syndrome. In the future, better characterization of genetic and developmental factors predisposing to melorheostosis may lead to the development of targeted therapy for this condition, as well as for more commonly encountered skeletal abnormalities.







Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Greenspan A. Sclerosing bone dysplasias—a target-site approach. Skeletal Radiol. 1991;20:561–83
Léri A, Joanny J. Une affection non décrite des os: hyperostose “en coulée” sur toute la longueur d’un membre ou “mélorhéostose”. Bull Mem Soc Med Hosp Paris. 1922;46:1141–5
Murray RO, McCredie J. Melorheostosis and the sclerotomes: a radiological correlation. Skeletal Radiol. 1979;4:57–71
Wynne-Davies R, Gormley J. The prevalence of skeletal dysplasias. An estimate of their minimum frequency and the number of patients requiring orthopaedic care. J Bone Joint Surg Br. 1985;67:133–7
Greenspan A, Azouz EM. Bone dysplasia series melorheostosis: review and update. Can Assoc Radiol J. 1999;50:324–30
Kessler HB, Recht MP, Dalinka MK. Vascular anomalies in association with osteodystrophies—a spectrum. Skeletal Radiol. 1983;10:95–101
Morris JM, Samilson RL, Corley CL. Melorheostosis. Review of the literature and report of an interesting case with a nineteen-year follow-up. J Bone Joint Surg Am. 1963;45:1191–206
Rhys R, Davies AM, Mangham DC, Grimer RJ. Sclerotome distribution of melorheostosis and multicentric fibromatosis. Skeletal Radiol. 1998;27:633–6
Woolridge B, Stone NC, Denic N. Melorheostosis isolated to the calcaneus: a case report and review of the literature. Foot Ankle Int. 2005;26:660–3
• Smith GC, Pingree MJ, Freeman LA, Matsumoto JM, Howe BM, Kannas SN, et al. Melorheostosis: A retrospective clinical analysis of 24 patients at the Mayo Clinic. PM&R. 2016;30:30813–9. This is the largest available case series reporting that the most common radiological finding in melorheostosis patients is a mixed pattern consisting of different forms of hyperostosis.
Ethunandan M, Khosla N, Tilley E, Webb A. Melorheostosis involving the craniofacial skeleton. J Craniofac Surg. 2004;15:1062–5
Williams JW, Monaghan D, Barrington NA. Cranio-facial melorheostosis: case report and review of the literature. Br J Radiol. 1991;64:60–2
• Ihde LL, Forrester DM, Gottsegen CJ, Masih S, Patel DB, Vachon LA, et al. Sclerosing bone dysplasias: review and differentiation from other causes of osteosclerosis. RadioGraphics. 2011;31:1865–82. This review describes the various sclerosing bone dysplasias, differentiating between hereditary and non-hereditary conditions, and discusses each condition briefly.
• Freyschmidt J. Melorheostosis: a review of 23 cases. Eur Radiol. 2001;11:474–9. This article describes patterns of melorheostosis other than the classically described “dripping candle wax” pattern.
Suresh S, Muthukumar T, Saifuddin A. Classical and unusual imaging appearances of melorheostosis. Clin Radiol. 2010;65:593–600
Davis DC, Syklawer R, Cole RL. Melorheostosis on three-phase bone scintigraphy. Case report. Clin Nucl Med. 1992;17:561–4
Mahoney J, Achong DM. Demonstration of increased bone metabolism in melorheostosis by multiphase bone scanning. Clin Nucl Med. 1991;16:847–8
Hassan A, Khalid M, Khawar S. Detection of melorheostosis in a young lady with upper limb pain on Three Phase Bone Scintigram/SPECT-CT. Clin Cases Miner Bone Metab. 2016;13:48–50
Hellemans J, Debeer P, Wright M, Janecke A, Kjaer KW, Verdonk PC, et al. Germline LEMD3 mutations are rare in sporadic patients with isolated melorheostosis. Hum Mut. 2006;27:290
Mumm S, Wenkert D, Zhang X, McAlister WH, Mier RJ, Whyte MP. Deactivating germline mutations in LEMD3 cause osteopoikilosis and Buschke–Ollendorff syndrome, but not sporadic melorheostosis. J Bone Miner Res. 2007;22:243–50
Vanhoenacker FM, De Beuckeleer LH, Van Hul W, Balemans W, Tan GJ, Hill SC, et al. Sclerosing bone dysplasias: genetic and radioclinical features. Eur Radiol. 2000;10:1423–33
Hellemans J, Preobrazhenska O, Willaert A, Debeer P, Verdonk PC, Costa T, et al. Loss-of-function mutations in LEMD3 result in osteopoikilosis, Buschke–Ollendorff syndrome and melorheostosis. Nat Genet. 2004;36:1213–8
Campbell CJ, Papademetriou T, Bonfiglio M. Melorheostosis. A report of the clinical, roentgenographic, and pathological findings in fourteen cases. J Bone Joint Surg Am. 1968;50:1281–304.
Roger D, Bonnetblanc JM, Leroux-Robert C. Melorheostosis with associated minimal change nephrotic syndrome, mesenteric fibromatosis and capillary haemangiomas. Dermatology. 1994;188:166–8
Kim JE, Kim EH, Han EH, Park RW, Park IH, Jun SH, et al. A TGF-beta-inducible cell adhesion molecule, betaig-h3, is downregulated in melorheostosis and involved in osteogenesis. J Cell Biochem. 2000;77:169–78
Whyte MP, Griffith M, Trani L, Mumm S, Gottesman GS, McAlister WH, et al. Melorheostosis: Exome sequencing of an associated dermatosis implicates postzygotic mosaicism of mutated KRAS. Bone. 2017;101:145–55
Jha S, Papadakis G, Kim L, Malayeri A, Cowen EW, Lehky T, et al. Melorheostosis: clinical experience of 23 cases. Endocrine Society 2017. Orlando, FL. 2017. Abstract
Hoshi K, Amizuka N, Kurokawa T, Nakamura K, Shiro R, Ozawa H. Histopathological characterization of melorheostosis. Orthopedics. 2001;24:273–7
Baer SC, Ayala AG, Ro JY, Yasko AW, Raymond AK, Edeiken J. Case report 843. Malignant fibrous histiocytoma of the femur arising in melorheostosis. Skeletal Radiol. 1994;23:310–4
Murphy M, Kearns S, Cavanagh M, O’Connell D, Hurson B. Occurrence of osteosarcoma in a melorheostotic femur. Ir Med J. 2003;96:55–6
Saxena A, Neelakantan A, Jampana R, Sangra M. Melorheostosis causing lumbar radiculopathy: a case report and a review of the literature. Spine J. 2013;13:27
Donáth J, Poór G, Kiss C, Fornet B, Genant H. Atypical form of active melorheostosis and its treatment with bisphosphonate. Skeletal Radiol. 2002;31:709–13.
Slimani S, Nezzar A, Makhloufi H. Successful treatment of pain in melorheostosis with zoledronate, with improvement on bone scintigraphy. BMJ Case Rep. 2013; 2013:pii: bcr2013009820
• Theriault RL. Zoledronic acid (Zometa) use in bone disease. Expert Rev Anticancer Ther. 2003;3:157–66. This case demonstrated radiological and clinical improvement following intravenous zoledronic acid.
Wood J, Bonjean K, Ruetz S, Bellahcène A, Devy L, Foidart JM, et al. Novel antiangiogenic effects of the bisphosphonate compound zoledronic acid. J Pharmacol Exp Ther. 2002;302:1055–61
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Bart Clarke and Anupam Kotwal declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Rare Bone Disease
Rights and permissions
About this article
Cite this article
Kotwal, A., Clarke, B.L. Melorheostosis: a Rare Sclerosing Bone Dysplasia. Curr Osteoporos Rep 15, 335–342 (2017). https://doi.org/10.1007/s11914-017-0375-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-017-0375-y