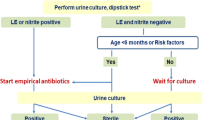
Abstract
This review highlights recent findings regarding complicated urinary tract infections (UTIs). We review recently published randomized controlled trials, Cochrane reviews, and meta-analyses. Topics covered include new findings on the genetics of acute pyelonephritis, methods to prevent catheter-associated UTI, duration of treatment for catheter-associated UTI, and reimbursement changes for Medicare patients who develop nosocomial UTI in US hospitals. Future studies on complicated UTI are needed to better define this complex disease and its management and to confirm the clinical significance of different prevention strategies.
Similar content being viewed by others
References and Recommended Reading
Hooton TM, Stamm WE: Diagnosis and treatment of uncomplicated urinary tract infection. Infect Dis Clin North Am 1997, 11:551–581.
Hang L, Frendeus B, Godaly G, Svanborg C: Interleukin-8 receptor knockout mice have subepithelial neutrophil entrapment and renal scarring following acute pyelonephritis. J Infect Dis 2000, 182:1738–1748.
Frendeus B, Godalay G, Hang L, et al.: Interleukin-8 receptor deficiency confers susceptibility to acute pyelonephritis and may have a human counterpart. J Exp Med 2000, 192:881–890.
Hopkins WJ, Uehling DT, Wargowski DS: Evaluation of a familial predisposition to recurrent urinary tract infections in women. Am J Med Genet 1999, 83:422–424.
Lundstedt AC, McCarthy S, Gustafsson CU, et al.: A genetic basis of susceptibility to acute pyelonephritis. PloS ONE 2007, 2:e825.
Lundstendt AC, Leijonhufvud I, Ragnarsdottir B, et al.: Inherited susceptibility to acute pyelonephritis: a family study of urinary tract infection. J Infect Dis 2007, 195:1227–1234.
Saint S, Kaufman SR, Rogers MA, et al.: Condom versus indwelling urinary catheters: a randomized trial. J Am Geriatr Soc 2006, 54:1055–1061.
Jacobsen SM, Stickler DJ, Mobley HL, Shirtliff ME: Complicated catheter-associated urinary tract infections due to Escherichia coli and Proteus mirabilis. Clin Microb Rev 2008, 21:26–59.
Laehdevirta J, Tallgren KG, Kuhlbaeck B: System of closed, sterile urinary drainage with indwelling catheter. Duodecim 1963, 79:978–984.
Cornia PB, Amory JK, Fraser S, et al.: Computer based order entry decreases duration of indwelling urinary tract infections in hospitalized patients. Am J Med 2003, 114:404–407.
Apisarnthanarak A, Thongphubeth K, Sirinvaravong S, et al.: Effectiveness of multifaceted hospital wide quality improvement programs featuring an intervention to remove unnecessary urinary catheters at the tertiary care center in Thailand. Infect Control Hosp Epidemiol 2007, 28:791–798.
Polliack T, Bluvshtein V, Philo O, et al.: Clinical and economical consequences of volume-or time-dependent intermittent catheterization in patients with spinal cord lesions and neuropathic bladder. Spinal Cord 2005, 43:615–619.
Johnson JR. Kuskowski MA, Wilt TJ: Systematic review: antimicrobial urinary catheters to prevent catheter associated urinary tract infection in hospitalized patients. Ann Intern Med 2006, 144:116–126.
Stensballe J, Tvede M, Looms D, et al.: Infection risk with nitrofurazone-impregnated urinary catheters in trauma patients. Ann Intern Med 2007, 147:285–293.
Trautner BW, Hull RA, Thornby JI, Darouiche RO: Coating urinary catheters with an avirulent strain of Escherichia coli as a means to establish asymptomatic colonization. Infect Control Hosp Epidemiol 2007, 28:92–94.
Niel-Weise BS, Van der Broek PJ: Urinary catheter policies for long-term bladder drainage. Cochrane Database Syst Rev 2005, 25(1):CD004201.
Salomon J, Denys P, Merle C, et al.: Prevention of urinary tract infection in spinal cord-injured patients: safety and efficacy of a weekly oral cyclic antibiotic (WOCA) program with a 2 year follow-up-an observational prospective study. J Antimicrob Chemother 2006: 57:784–788.
Stickler DJ, Jones SM, Adusei GO, et al.: A clinical assessment of the performance of a sensor to detect crystalline biofilm formation on indwelling bladder catheters. BJU Int 2006, 98:1244–1249.
Hazan Z, Zumeris J, Jacob H, et al.: Effective prevention of microbial biofilm formation on medical devices by low-energy surface acoustic waves. Antimicrob Agents Chemother 2006, 50:4144–4152.
Burton E, Gawande PV, Yakandawala K, et al.: Antibiofilm activity of GlmU enzyme inhibitors against catheter-associated uropathogens. Antimicrob Agents Chemother 2006, 50:1835–1840.
Peterson J, Kaul S, Khashab M, et al.: A double-blind, randomized comparison of levofloxacin 750 mg once-daily for five days with ciprofloxacin 400/500 mg twice-daily for 10 days for the treatment of complicated urinary tract infections and acute pyelonephritis. Urology 2008, 71:17–22.
Dow G, Rao P, Harding G, et al.: A prospective, randomized trial of 3 or 14 days of ciprofloxacin treatment for acute urinary tract infection in patients with spinal cord injury. Clin Infect Dis 2004, 39:658–664.
Pohl A: Modes of administration of antibiotics for symptomatic severe urinary tract infections. Cochrane Database Syst Rev 2007, 4:CD003237.
Wald H, Kramer A: Nonpayment for harms resulting from medical care. Catheter-associated urinary tract infections. JAMA 2007, 298:2782–2784.
Saint S, Kowalski C, Kaufman S, et al.: Preventing hospital-acquired urinary tract infections in the United States: a national study. Clin Infect Dis 2008, 46:243–250.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lichtenberger, P., Hooton, T.M. Complicated urinary tract infections. Curr Infect Dis Rep 10, 499–504 (2008). https://doi.org/10.1007/s11908-008-0081-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11908-008-0081-0