Abstract
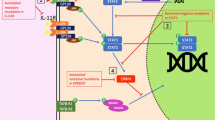
Hyper-IgE syndrome (HIES) is a complex primary immunodeficiency characterized by high serum IgE, chronic eczematoid dermatitis, and recurrent extracellular bacterial infections. Two types of HIES have been reported: type 1 and type 2. Type 1 HIES displays abnormalities in multiple systems, including the skeletal, dental, and immune systems, whereas type 2 shows abnormalities confined to the immune system. We recently identified hypomorphic mutations in the signal transducer and activator of transcription 3 (STAT3) gene in type 1 HIES and a null mutation in the tyrosine kinase 2 (Tyk2) gene, accompanied by susceptibility to intracellular bacteria in type 2 HIES. Analyses of cytokine responses in both types of HIES revealed that severe defects in the signal transduction for multiple cytokines, including interleukin-6 and interleukin-23, are leading to impaired T-helper type 17 function. These findings suggest that HIES is caused by the defects in multiple cytokine signals and that the susceptibility to various infections in HIES is associated with the T-helper type 17 defect.
Similar content being viewed by others
References and Recommended Reading
Minegishi Y, Karasuyama H: Hyperimmunoglobulin E syndrome and tyrosine kinase 2 deficiency. Curr Opin Allergy Clin Immunol 2007, 7:506–509.
Grimbacher B, Holland SM, Puck JM: Hyper-IgE syndromes. Immunol Rev 2005, 203:244–250.
Grimbacher B, Holland SM, Gallin JI, et al.: Hyper-IgE syndrome with recurrent infections—an autosomal dominant multisystem disorder. N Engl J Med 1999, 340:692–702.
Freeman AF, Kleiner DE, Nadiminti H, et al.: Causes of death in hyper-IgE syndrome. J Allergy Clin Immunol 2007, 119:1234–1240.
Renner ED, Puck JM, Holland SM, et al.: Autosomal recessive hyperimmunoglobulin E syndrome: a distinct disease entity. J Pediatr 2004, 144:93–99.
Ihle JN: Cytokine receptor signalling. Nature 1995, 377:591–594.
Liu KD, Gaffen SL, Goldsmith MA: JAK/STAT signaling by cytokine receptors. Curr Opin Immunol 1998, 10:271–278.
O’shea JJ, Gadina M, Schreiber RD: Cytokine signaling in 2002: new surprises in the Jak/Stat pathway. Cell 2002, 109(Suppl):S121–S131.
Muller M, Briscoe J, Laxton C, et al.: The protein tyrosine kinase JAK1 complements defects in interferon-alpha/beta and-gamma signal transduction. Nature 1993, 366:129–135.
Rodig SJ, Meraz MA, White JM, et al.: Disruption of the Jak1 gene demonstrates obligatory and nonredundant roles of the Jaks in cytokine-induced biologic responses. Cell 1998, 93:373–383.
Neubauer H, Cumano A, Muller M, et al.: Jak2 deficiency defines an essential developmental checkpoint in definitive hematopoiesis. Cell 1998, 93:397–409.
Parganas E, Wang D, Stravopodis D, et al.: Jak2 is essential for signaling through a variety of cytokine receptors. Cell 1998, 93:385–395.
Nosaka T, van Deursen JM, Tripp RA, et al.: Defective lymphoid development in mice lacking Jak3. Science 1995, 270:800–802.
Park SY, Saijo K, Takahashi T, et al.: Developmental defects of lymphoid cells in Jak3 kinase-deficient mice. Immunity 1995, 3:771–782.
Macchi P, Villa A, Giliani S, et al.: Mutations of Jak-3 gene in patients with autosomal severe combined immune deficiency (SCID). Nature 1995, 377:65–68.
Russell SM, Tayebi N, Nakajima H, et al.: Mutation of Jak3 in a patient with SCID: essential role of Jak3 in lymphoid development. Science 1995, 270:797–800.
Firmbach-Kraft I, Byers M, Shows T, et al.: Tyk2, prototype of a novel class of non-receptor tyrosine kinase genes. Oncogene 1990, 5:1329–1336.
Velazquez L, Fellous M, Stark GR, Pellegrini S: A protein tyrosine kinase in the interferon alpha/beta signaling pathway. Cell 1992, 70:313–322.
Karaghiosoff M, Neubauer H, Lassnig C, et al.: Partial impairment of cytokine responses in Tyk2-deficient mice. Immunity 2000, 13:549–560.
Shimoda K, Kato K, Aoki K, et al.: Tyk2 plays a restricted role in IFN alpha signaling, although it is required for IL-12-mediated T cell function. Immunity 2000, 13:561–571.
Bacon CM, McVicar DW, Ortaldo JR, et al.: Interleukin 12 (IL-12) induces tyrosine phosphorylation of JAK2 and TYK2: differential use of Janus family tyrosine kinases by IL-2 and IL-12. J Exp Med 1995, 181:399–404.
Stahl N, Boulton TG, Farruggella T, et al.: Association and activation of Jak-Tyk kinases by CNTF-LIF-OSM-IL-6 beta receptor components. Science 1994, 263:92–95.
Minegishi Y, Coustan-Smith E, Wang YH, et al.: Mutations in the human lambda5/14.1 gene result in B cell deficiency and agammaglobulinemia. J Exp Med 1998, 187:71–77.
Minegishi Y, Rohrer J, Coustan-Smith E, et al.: An essential role for BLNK in human B cell development. Science 1999, 286:1954–1957.
Conley ME, Rohrer J, Rapalus L, et al.: Defects in early B-cell development: comparing the consequences of abnormalities in pre-BCR signaling in the human and the mouse. Immunol Rev 2000, 178:75–90.
Akira S, Nishio Y, Inoue M, et al.: Molecular cloning of APRF, a novel IFN-stimulated gene factor 3 p91-related transcription factor involved in the gp130-mediated signaling pathway. Cell 1994, 77:63–71.
Zhong Z, Wen Z, Darnell JE Jr: Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science 1994, 264:95–98.
Wegenka UM, Buschmann J, Lutticken C, et al.: Acute-phase response factor, a nuclear factor binding to acute-phase response elements, is rapidly activated by interleukin-6 at the posttranslational level. Mol Cell Biol 1993, 13:276–288.
Lutticken C, Wegenka UM, Yuan J, et al.: Association of transcription factor APRF and protein kinase Jak1 with the interleukin-6 signal transducer gp130. Science 1994, 263:89–92.
Nakajima K, Yamanaka Y, Nakae K, et al.: A central role for Stat3 in IL-6-induced regulation of growth and differentiation in M1 leukemia cells. EMBO J 1996, 15:3651–3658.
Williams L, Bradley L, Smith A, Foxwell B: Signal transducer and activator of transcription 3 is the dominant mediator of the anti-inflammatory effects of IL-10 in human macrophages. J Immunol 2004, 172:567–576.
Minami M, Inoue M, Wei S, et al.: STAT3 activation is a critical step in gp130-mediated terminal differentiation and growth arrest of a myeloid cell line. Proc Natl Acad Sci U S A 1996, 93:3963–3966.
Takeda K, Noguchi K, Shi W, et al.: Targeted disruption of the mouse Stat3 gene leads to early embryonic lethality. Proc Natl Acad Sci U S A 1997, 94:3801–3804.
Akira S: Roles of STAT3 defined by tissue-specific gene targeting. Oncogene 2000, 19:2607–2611.
Kasprzycka M, Marzec M, Liu X, et al.: Nucleophosmin/anaplastic lymphoma kinase (NPM/ALK) oncoprotein induces the T regulatory cell phenotype by activating STAT3. Proc Natl Acad Sci U S A 2006, 103:9964–9969.
Davis SD, Schaller J, Wedgwood RJ: Job’s syndrome. Recurrent, “cold,” staphylococcal abscesses. Lancet 1966, 1:1013–1015.
Buckley RH, Wray BB, Belmaker EZ: Extreme hyperimmunoglobulinemia E and undue susceptibility to infection. Pediatrics 1972, 49:59–70.
Del Prete G, Tiri A, Maggi E, et al.: Defective in vitro production of gamma-interferon and tumor necrosis factor-alpha by circulating T cells from patients with the hyper-immunoglobulin E syndrome. J Clin Invest 1989, 84:1830–1835.
Borges WG, Augustine NH, Hill HR: Defective interleukin-12/interferon-gamma pathway in patients with hyperimmunoglobulinemia E syndrome. J Pediatr 2000, 136:176–180.
Chehimi J, Elder M, Greene J, et al.: Cytokine and chemokine dysregulation in hyper-IgE syndrome. Clin Immunol 2001, 100:49–56.
Wood PM, Fieschi C, Picard C, et al.: Inherited defects in the interferon-gamma receptor or interleukin-12 signalling pathways are not sufficient to cause allergic disease in children. Eur J Pediatr 2005, 164:741–747.
Minegishi Y, Saito M, Morio T, et al.: Human tyrosine kinase 2 deficiency reveals its requisute roles in multiple cytokine signals involved in innate and acquired immunity. Immunity 2006, 25:745–755.
Casanova JL, Abel L: Genetic dissection of immunity to mycobacteria: the human model. Annu Rev Immunol 2002, 20:581–620.
Minegishi Y, Saito M, Tsuchiya S, et al.: Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature 2007, 448:1058–1062.
Grimbacher B, Schaffer AA, Holland SM, et al.: Genetic linkage of hyper-IgE syndrome to chromosome 4. Am J Hum Genet 1999, 65:735–744.
Holland SM, DeLeo FR, Elloumi HZ, et al.: STAT3 mutations in the hyper-IgE syndrome. N Engl J Med 2007, 357:1608–1619.
Renner ED, Torgerson TR, Rylaarsdam S, et al.: STAT3 mutation in the original patient with Job’s syndrome. N Engl J Med 2007, 357:1667–1668.
Dupuis S, Dargemont C, Fieschi C, et al.: Impairment of mycobacterial but not viral immunity by a germline human STAT1 mutation. Science 2001, 293:300–303.
Itoh S, Udagawa N, Takahashi N, et al.: A critical role for interleukin-6 family-mediated Stat3 activation in osteoblast differentiation and bone formation. Bone 2006, 39:505–512.
Yang XO, Panopoulos AD, Nurieva R, et al.: STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J Biol Chem 2007, 282:9358–9363.
Toy D, Kugler D, Wolfson M, et al.: Cutting edge: interleukin 17 signals through a heteromeric receptor complex. J Immunol 2006, 177:36–39.
Aujla SJ, Chan YR, Zheng M, et al.: IL-22 mediates mucosal host defense against gram-negative bacterial pneumonia. Nat Med 2008, 14:275–281.
Zheng Y, Valdez PA, Danilenko DM, et al.: Interleukin-22 mediates early host defense against attaching and effacing bacterial pathogens. Nat Med 2008, 14:282–289.
Kreymborg K, Etzensperger R, Dumoutier L, et al.: IL-22 is expressed by Th17 cells in an IL-23-dependent fashion, but not required for the development of autoimmune encephalomyelitis. J Immunol 2007, 179:8098–8104.
Wolk K, Kunz S, Witte E, et al.: IL-22 increases the innate immunity of tissues. Immunity 2004, 21:241–254.
Milner JD, Brenchley JM, Laurence A, et al.: Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 2008, 452:773–776.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Minegishi, Y., Karasuyama, H. Genetic origins of hyper-IgE syndrome. Curr Allergy Asthma Rep 8, 386–391 (2008). https://doi.org/10.1007/s11882-008-0075-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11882-008-0075-x