Background
Sleeve gastrectomy (SG) has been used for the surgical treatment of morbid obesity, as a first step or as a definitive treatment.The objective of this pilot study was to establish an animal model for SG in Wistar rats.
Methods
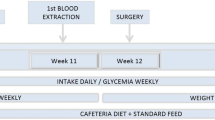
12 male Wistar rats were operated on. 8 of them underwent a SG, with a hand-sewn invaginating suture, and 4 underwent a laparotomy and stomach manipulation (control group). Sterilized materials, prophylactic antibiotics and nutritional supplementation were used. The animals’ weight was checked on the operation day and on a weekly basis, for 7 weeks.
Results
The experimental SG model was possible due to careful procedures in the pre- and postoperative period. The average weight loss in the sleeve gastrectomy group was 49.6 g (43 to 60 g) until the 4th week, whereas in the control group average weight loss was 15 g (10 to 25 g) but the weight loss was achieved in the 1st week. After the 7th week, there was weight gain in both groups. However, in the SG group, this gain was significantly lower than in the control group.
Conclusion
It has been possible to obtain a SG experimental model. The effects of this SG appear to be beneficial in weight loss. Biochemical and molecular mechanisms may also be investigated based on this model.
Similar content being viewed by others
References
Thompson D, Wolf AM. The medical-care cost burden of obesity. Obes Rev 2001; 2: 189–97.
Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. The evidence report. National Institutes of Health. NIH Publication. No 98-4083, September 1998.
Deitel M. Overweight and obesity worldwide now estimated to involve 1.7 billion people (Editorial). Obes Surg 2003; 13: 329–30.
Buchwald H, Buchwald JN. Evolution of operative procedures for the management of morbid obesity 1950–2000. Obes Surg 2002; 12: 705–17.
de Jong JR, van Ramshorst B, Timmer R et al. Effect of laparoscopic gastric banding on esophageal motility. Obes Surg 2006;16: 52–8.
Chevalier JM, Zinzindohoúe F, Douard R et al. Complications after laparoscopic adjustable gastric banding for morbid obesity: experience with 1,000 patients over 7 years. Obes Surg 2004; 14: 407–14.
Peterli R, Danodini A, Peters T et al. Re-operations following laparoscopic adjustable gastric banding. Obes Surg 2002; 12: 851–6.
Marano Jr. JB. Endoscopy after Roux-en-Y gastric bypass: A community hospital experience. Obes Surg 2005; 15: 342–5.
Schirmer B, Erenoglu C, Anna Miller RN. Flexible endoscopy in the management of patients undergoing Roux-en-Y gastric bypass. Obes Surg 2002; 12: 634–8.
Nguyen NT, Longoria M, Chalifoux BS et al. Gastrointestinal hemorrhage after laparoscopic gastric bypass. Obes Surg 2004; 14: 1308–12.
Mehran A, Szomstein S, Zundel N et al. Management of acute bleeding after laparoscopic Roux-en-Y gastric bypass. Obes Surg 2003; 13: 842–7.
Stellato TA, Crouse C, Hallowell PT. Bariatric surgery: creating new challenges for the endoscopist. Gastrointest Endosc 2003; 57: 86–94.
Adami GF, Cordera E, Marinari G et al. Plasma ghrelin concentration in the short-term following biliopancreatic diversion. Obes Surg 2003;13: 889–92.
Fruhbeck G, Rotellar F; Hernãndez-Lizoain JL et al. Fasting plasma ghrelin concentration 6 months after gastric bypass are not determined by weight loss or changes in insulinemia. Obes Surg 2004; 14:1208–15.
Fruhbeck G, Diez-Caballero A, Gil MJ et al. The decrease in plasma ghrelin concentrations following bariatric surgery depends on the functional integrity of the fundus. Obes Surg 2004; 14: 606–12.
Couce ME, Cottam D, Esplen J et al. Is ghrelin the culprit for weight loss after gastric bypass surgery? A negative answer. Obes Surg 2006; 16: 870–8.
Regan JP, Inabnet B, Gagner M et al. Early experience with two-stage laparoscopic Roux-en-Y gastric bypass as an alternative in the super-super obese patient. Obes Surg 2003; 13: 861–4.
Almogy G, Crookes PF, Anthone GJ. Longitudinal gastrectomy as a treatment for the high-risk superobese patient. Obes Surg 2004; 14: 492–7.
Mognol P, Chosidow D, Marmuse JP. Laparoscopic sleeve gastrectomy as an initial bariatric operation for high-risk patients: initial results in 10 patients. Obes Surg 2005; 15: 1030–3.
Catherine JM, Cohen R, Cocktail I et al. Traitment de la super super obésité morbide par la gastrectomie longitudinale. Presse Med 2006; 33: 383–7.
Baltasar A, Serra C, Pérez N et al. Laparoscopic sleeve gastrectomy: a multi-purpose operation. Obes Surg 2005; 15: 1124–8.
Cohen R, Uzzan B, Bihan H et al. Ghrelin levels and sleeve gastrectomy in super-super-obesity (Letter). Obes Surg 2005; 15: 1501–2.
Han SM, Kim WW, Hyun J. Results of laparoscopic sleeve gastrectomy (LSG) at 1 year in morbidly obese Korean patients. Obes Surg 2005; 15:1469–75.
Milone L, Strong V, Gagner M. Laparoscopic sleeve gastrectomy is superior to endoscopic intragastric balloon as a first stage procedure for super-obese patients (BMI>50). Obes Surg 2005; 5: 612–7.
Xu Y, Ohinata K, Meguid MM et al. Gastric bypass model in the obese rat to study metabolic mechanisms of weight loss. J Surg Res 2002; 107: 56–63.
Monteiro MP, Monteiro JD, Águas AP et al. A rat model of restrictive bariatric surgery with gastric banding. Obes Surg 2006; 16: 48–51.
Fusco PEB, Poggetti RS, Younes RN et al. Evaluation of gastric greater curvature invagination for weight loss in rats. Obes Surg 2006; 16: 172–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Castelan F°, J.d.B., Bettiol, J., d’Acampora, A.J. et al. Sleeve Gastrectomy Model in Wistar Rats. OBES SURG 17, 957–961 (2007). https://doi.org/10.1007/s11695-007-9150-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-007-9150-y