Abstract
Purpose
To assess the validity and reliability of the fatigue subscale of the Functional Assessment of Chronic Illness Therapy (FACIT-F), a 6-item subset from the thrombocytopenia subscale of the Functional Assessment of Cancer Therapy (FACT-Th6) and the Short Form-36 Version 2 (SF-36v2) in 2 clinical trials of the thrombopoietin receptor agonist eltrombopag in chronic immune thrombocytopenia (ITP) patients.
Methods
In the 6-month, RAndomized placebo-controlled ITP Study with Eltrombopag (RAISE; n = 197), the FACIT-F, FACT-Th6, and SF-36v2 were administered at baseline, day 43, weeks 14 and 26, or early withdrawal. In the ongoing open-label extension study, Eltrombopag EXTENDed Dosing Study (EXTEND; n = 154), measures were administered at baseline, at the beginning of each stage, and at permanent discontinuation of study medication.
Results
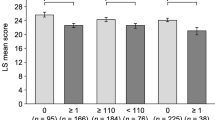
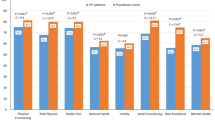
FACIT-F, FACT-Th6, and SF-36v2 demonstrated acceptable internal consistency reliability (i.e., all Cronbach’s alphas >0.70) and test–retest reliability (all intraclass correlation coefficients >0.70). Construct validity was supported by moderate (0.35 < r < 0.50) to strong (r > 0.50) inter-measure correlations for baseline and change scores. A small to medium magnitude of effect was captured by the FACIT-F and FACT-Th6 among patients who experienced sustained platelet responses.
Conclusions
Results provide support for the validity, reliability, and responsiveness of the FACIT-F, FACT-Th6, and SF-36v2 in chronic ITP patients.
Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- EXTEND:
-
Eltrombopag EXTENDed dosing study
- FACIT-F:
-
Fatigue subscale of the Functional Assessment of Chronic Illness Therapy
- FACT-Th:
-
Functional Assessment of Cancer Therapy-Thrombocytopenia
- HRQoL:
-
Health-related quality of life
- ICC:
-
Intraclass correlation coefficient
- ITP:
-
Immune thrombocytopenia
- LOTA:
-
Last on-treatment assessment
- MCS:
-
Mental component summary
- MEI-SF:
-
Motivation and Energy Inventory-Short Form
- MID:
-
Minimally important difference
- PCS:
-
Physical component summary
- RAISE:
-
RAndomized placebo-controlled ITP Study with Eltrombopag
- SD:
-
Standard deviation
- SF-36v2:
-
Medical Outcomes Study Short Form-36 version 2
- WHO:
-
World Health Organization
References
Ballem, P. J., Segal, G. M., Stratton, J. R., Gernsheimer, T., Adamson, J. W., & Slichter, S. J. (1987). Mechanisms of thrombocytopenia in chronic autoimmune thrombocytopenic purpura. Evidence of both impaired platelet production and increased platelet clearance. The Journal of Clinical Investigation, 80, 33–40.
Chang, M., Nakagawa, P. A., Williams, S. A., Schwartz, M. R., Imfeld, K. L., Buzby, J. S., et al. (2003). Immune thrombocytopenic purpura (ITP) plasma and purified ITP monoclonal autoantibodies inhibit megakaryocytopoiesis in vitro. Blood, 102, 887–895.
Cines, D. B., & Blanchette, V. S. (2002). Immune thrombocytopenic purpura. New England Journal of Medicine, 346, 995–1008.
Feudjo-Tepie, M. A., Robinson, N. J., & Bennett, D. (2008). Prevalence of diagnosed chronic immune thrombocytopenic purpura in the us: Analysis of a large us claim database: A rebuttal. Journal of Thrombosis Haemostasis, 6, 711–712, author reply 713.
Mathias, S. D., Bussel, J. B., George, J. N., McMillan, R., Okano, G. J., & Nichol, J. L. (2007). A disease-specific measure of health-related quality of life for use in adults with immune thrombocytopenic purpura: Its development and validation. Health and Quality of Life Outcomes, 5, 11.
Mathias, S. D., Gao, S. K., Miller, K. L., Cella, D., Snyder, C., Turner, R., et al. (2008). Impact of chronic immune thrombocytopenic purpura (ITP) on health-related quality of life: A conceptual model starting with the patient perspective. Health and Quality of Life Outcomes, 6, 13. Available at http://www.hqlo.com/content/16/11/13.
Bussel, J. B., Cheng, G., Saleh, M. N., Psaila, B., Kovaleva, L., Meddeb, B., et al. (2007). Eltrombopag for the treatment of chronic idiopathic thrombocytopenic purpura. New England Journal of Medicine, 357, 2237–2247.
Cella, D., Eton, D. T., Lai, J. S., Peterman, A. H., & Merkel, D. E. (2002). Combining anchor and distribution-based methods to derive minimal clinically important differences on the functional assessment of cancer therapy (FACT) anemia and fatigue scales. Journal of Pain and Symptom Management, 24, 547–561.
Webster, K., Cella, D., & Yost, K. (2003). The functional assessment of chronic illness therapy (FACIT) measurement system: Properties, applications, and interpretation. Health and Quality of Life Outcomes, 1, 79.
Cella, D., Beaumont, J. L., Webster, K. A., Lai, J. S., & Elting, L. (2006). Measuring the concerns of cancer patients with low platelet counts: The functional assessment of cancer therapy–thrombocytopenia (FACT-Th) questionnaire. Supportive Care in Cancer, 14, 1220–1231.
Ware, J. E., Kosinski, M., Bjorner, J. B., Turner-Bowker, D. M., Gandek, B., & Mariush, M. E. (2007). User’s manual for the sf-36v2 health survey (2nd ed.). Lincoln, RI: Quality Metric Incorporated.
Cheng, G., Saleh, M. N., Marcher, C., Vasey, S., Mayer, B., Aivado, M., et al. (2010). Eltrombopag for the long-term management of chronic idiopathic thrombocytopenic purpura: A 6-month, randomized, phase iii study (RAISE). Lancet. Epub ahead of print.
Saleh, M. N., Bussel, J. B., Cheng, G., Meddeb, B., Mayer, B., Bailey, C., et al. (2009). Long-term treatment of chronic immune thrombocytopenic purpura with oral eltrombopag: Results from the extend study. Blood, 114, Abstract 682.
Fehnel, S. E., Bann, C. M., Hogue, S. L., Kwong, W. J., & Mahajan, S. S. (2004). The development and psychometric evaluation of the motivation and energy inventory (MEI). Quality of Life Research, 13, 1321–1336.
Marra, C. A., Woolcott, J. C., Kopec, J. A., Shojania, K., Offer, R., Brazier, J. E., et al. (2005). A comparison of generic, indirect utility measures (the hui2, hui3, sf-6d, and the eq-5d) and disease-specific instruments (the raqol and the haq) in rheumatoid arthritis. Social Science and Medicine, 60, 1571–1582.
Guttman, L. (1954). Some necessary conditions for common-factor analysis. Psychometrika, 19, 149–161.
Kaiser, H. F. (1960). The application of electronic computers to factor analysis. Educational and Psychological Measurement, 20, 141–151.
Kaiser, H. F. (1961). A note on Guttman’s lower bound for the number of common factors. British Journal of Mathematical and Statistical Psychology, 14, 1.
Kline, P. (1993). Handbook of psychological testing. London: Routledge.
Bland, J. M., & Altman, D. G. (1997). Cronbach’s alpha. British Medical Journal, 314, 572.
Shrout, P. E., & Fleiss, J. L. (1979). Intraclass correlations: Uses in addressing rater reliability. Psychological Bulletin, 86, 420–428.
Cohen, J. (1977). Statistical power analysis for the behavioral science (revised ed.) New York: Academic Press.
Moretti, C., Viola, S., Pistorio, A., Magni-Manzoni, S., Ruperto, N., Martini, A., et al. (2005). Relative responsiveness of condition specific and generic health status measures in juvenile idiopathic arthritis. Annals of the Rheumatic Diseases, 64, 257–261.
Liang, K.-Y., & Seger, S. L. (1986). Longitudinal data analysis using generalized linear models. Biometrika, 73, 13–22.
Cella, D., Lai, J. S., Chang, C. H., Peterman, A., & Slavin, M. (2002). Fatigue in cancer patients compared with fatigue in the general united states population. Cancer, 94, 528–538.
Guyatt, G. H., King, D. R., Feeny, D. H., Stubbing, D., & Goldstein, R. S. (1999). Generic and specific measurement of health-related quality of life in a clinical trial of respiratory rehabilitation. Journal of Clinical Epidemiology, 52(3), 187–92. PubMed PMID: 10210235.
McGraw, K. O., & Wong, S. P. (1996). Forming inferences about some intraclass correlation coefficients. Psychological Methods, 1, 30–46.
Acknowledgments
Funding for this study was provided by GlaxoSmithKline. All listed authors meet the criteria for authorship set forth by the International Committee for Medical Journal Editors. The authors wish to acknowledge the following individuals for their contributions and critical review during the development of this manuscript: Lisa Breck, Steve Collins, and AOI Communications, L.P., for editorial assistance, and Kimberly Marino for critical review.
Author information
Authors and Affiliations
Corresponding author
Additional information
Trial registration numbers: EXTEND NCT00351468, RAISE NCT00370331.
Rights and permissions
About this article
Cite this article
Signorovitch, J., Brainsky, A. & Grotzinger, K.M. Validation of the FACIT-fatigue subscale, selected items from FACT-thrombocytopenia, and the SF-36v2 in patients with chronic immune thrombocytopenia. Qual Life Res 20, 1737–1744 (2011). https://doi.org/10.1007/s11136-011-9912-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-011-9912-9