Abstract
Purpose
To compare the responses of intravitreal injections of bevacizumab, ranibizumab, or aflibercept for the treatment of neovascular age-related macular degeneration (nAMD).
Methods
This retrospective study examined 232 eyes of 232 patients who received intravitreal anti-vascular endothelial growth factor (VEGF) injections due to treatment-naïve nAMD. All patients, who were followed-up for at least 1 year, were treated with intravitreal injections monthly until 3 months, and then as needed. We evaluated the effects of intravitreal injections for treatment of nAMD using the central macular thickness (CMT), subretinal fluid (SRF), pigment epithelial detachment (PED) size, and best-corrected visual acuity (BCVA).
Results
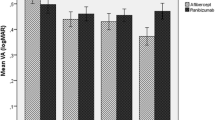
CMT, SRF, PED size, and BCVA (LogMAR) were significantly decreased after treatment with all three anti-VEGF agents. Overall, the bevacizumab, ranibizumab, and aflibercept treatments showed no significant differences in their responses. However, the aflibercept injections decreased PED size more quickly than bevacizumab injections (P = 0.034).
Conclusions
Bevacizumab, ranibizumab, and aflibercept injections are effective treatments for nAMD and have similar responses, although the number of injections of aflibercept was fewer than other anti-VEGF agents. In addition, aflibercept injections may be a better choice than other anti-VEGF agents for cases of severe increases in PED height.




Similar content being viewed by others
References
Andreoli CM, Miller JW (2007) Anti-vascular endothelial growth factor therapy for ocular neovascular disease. Curr Opin Ophthalmol 18(6):502–508. doi:10.1097/ICU.0b013e3282f0ca54
Rosenfeld PJ, Rich RM, Lalwani GA (2006) Ranibizumab: phase III clinical trial results. Ophthalmol Clin N Am 19(3):361–372. doi:10.1016/j.ohc.2006.05.009
Ferrara N, Damico L, Shams N, Lowman H, Kim R (2006) Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina 26(8):859–870. doi:10.1097/01.iae.0000242842.14624.e7
Heier JS, Boyer D, Nguyen QD, Marcus D, Roth DB, Yancopoulos G, Stahl N, Ingerman A, Vitti R, Berliner AJ, Yang K, Brown DM et al (2011) The 1-year results of CLEAR-IT 2, a phase 2 study of vascular endothelial growth factor trap-eye dosed as-needed after 12-week fixed dosing. Ophthalmology 118(6):1098–1106. doi:10.1016/j.ophtha.2011.03.020
Ladewig MS, Ziemssen F, Jaissle G, Helb HM, Scholl HP, Eter N, Bartz-Schmidt KU, Holz FG (2006) Intravitreal bevacizumab for neovascular age-related macular degeneration. Der Ophthalmologe: Zeitschrift der Deutschen Ophthalmologischen Gesellschaft 103(6):463–470. doi:10.1007/s00347-006-1352-5
Ho QT, Kuo CJ (2007) Vascular endothelial growth factor: biology and therapeutic applications. Int J Biochem Cell Biol 39(7–8):1349–1357. doi:10.1016/j.biocel.2007.04.010
Brown DM, Kaiser PK, Michels M, Soubrane G, Heier JS, Kim RY, Sy JP, Schneider S, AS Group (2006) Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 355(14):1432–1444. doi:10.1056/NEJMoa062655
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY, MS Group (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355(14):1419–1431. doi:10.1056/NEJMoa054481
Comparison of Age-related Macular Degeneration Treatments Trials Research G, Martin DF, Maguire MG, Fine SL, Ying GS, Jaffe GJ, Grunwald JE, Toth C, Redford M, Ferris FL, 3rd (2012) Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 119 (7):1388–1398 10.1016/j.ophtha.2012.03.053
Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD, Kirchhof B, Ho A, Ogura Y, Yancopoulos GD, Stahl N, Vitti R, Berliner AJ, Soo Y, Anderesi M, Groetzbach G, Sommerauer B, Sandbrink R, Simader C, Schmidt-Erfurth U, View Groups VS (2012) Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119(12):2537–2548. doi:10.1016/j.ophtha.2012.09.006
Jorstad OK, Faber RT, Moe MC (2015) Initial improvements when converting eyes with treatment-resistant exudative AMD to aflibercept are substantially diminished after increasing treatment intervals from 4 to 8 weeks. Acta Ophthalmol 93(6):e510–e511. doi:10.1111/aos.12681
Bakall B, Folk JC, Boldt HC, Sohn EH, Stone EM, Russell SR, Mahajan VB (2013) Aflibercept therapy for exudative age-related macular degeneration resistant to bevacizumab and ranibizumab. Am J Ophthalmol 156(1):15–22. doi:10.1016/j.ajo.2013.02.017
Arcinue CA, Ma F, Barteselli G, Sharpsten L, Gomez ML, Freeman WR (2015) One-year outcomes of aflibercept in recurrent or persistent neovascular age-related macular degeneration. Am J Ophthalmol 159(3):426–436. doi:10.1016/j.ajo.2014.11.022
Ali F, Chan WC, Stevenson MR, Muldrew KA, Chakravarthy U (2004) Morphometric analysis of angiograms of exudative lesions in age-related macular degeneration. Arch Ophthalmol 122(5):710–715. doi:10.1001/archopht.122.5.710
Pauleikhoff D (2005) Neovascular age-related macular degeneration: natural history and treatment outcomes. Retina 25(8):1065–1084
Kang HM, Kwon HJ, Yi JH, Lee CS, Lee SC (2014) Subfoveal choroidal thickness as a potential predictor of visual outcome and treatment response after intravitreal ranibizumab injections for typical exudative age-related macular degeneration. Am J Ophthalmol 157(5):1013–1021. doi:10.1016/j.ajo.2014.01.019
Amoaku WM, Chakravarthy U, Gale R, Gavin M, Ghanchi F, Gibson J, Harding S, Johnston RL, Kelly SP, Lotery A, Mahmood S, Menon G, Sivaprasad S, Talks J, Tufail A, Yang Y (2015) Defining response to anti-VEGF therapies in neovascular AMD. Eye 29(6):721–731. doi:10.1038/eye.2015.48
Carneiro AM, Mendonca LS, Falcao MS, Fonseca SL, Brandao EM, Falcao-Reis FM (2012) Comparative study of 1 + PRN ranibizumab versus bevacizumab in the clinical setting. Clin Ophthalmol 6:1149–1157. doi:10.2147/OPTH.S33017
Gamulescu MA, Radeck V, Lustinger B, Fink B, Helbig H (2010) Bevacizumab versus ranibizumab in the treatment of exudative age-related macular degeneration. Int Ophthalmol 30(3):261–266. doi:10.1007/s10792-009-9318-7
De Bats F, Grange JD, Cornut PL, Feldman A, Burillon C, Denis P, Kodjikian L (2012) Bevacizumab versus ranibizumab in the treatment of exudative age-related macular degeneration: a retrospective study of 58 patients. J Fr Ophtalmol 35(9):661–666. doi:10.1016/j.jfo.2012.01.015
Kanesa-Thasan A, Grewal DS, Gill MK, Lyon AT, Mirza RG (2015) Quantification of change in pigment epithelial detachment volume and morphology after transition to intravitreal aflibercept in eyes with recalcitrant neovascular amd: 18-month results. Ophthalmic Surge Lasers Imaging Retina 46(6):638–641. doi:10.3928/23258160-20150610-07
Batioglu F, Demirel S, Ozmert E, Abdullayev A, Bilici S (2015) Short-term outcomes of switching anti-VEGF agents in eyes with treatment-resistant wet AMD. BMC ophthalmol 15:40. doi:10.1186/s12886-015-0025-z
Schmid MK, Bachmann LM, Fas L, Kessels AG, Job OM, Thiel MA (2015) Efficacy and adverse events of aflibercept, ranibizumab and bevacizumab in age-related macular degeneration: a trade-off analysis. Br J Ophthalmol 99(2):141–146. doi:10.1136/bjophthalmol-2014-305149
Stefansson E (2009) Physiology of vitreous surgery. Graefe’s archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 247(2):147–163. doi:10.1007/s00417-008-0980-7
Gisladottir S, Loftsson T, Stefansson E (2009) Diffusion characteristics of vitreous humour and saline solution follow the Stokes Einstein equation. Graefe’s archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 247(12):1677–1684. doi:10.1007/s00417-009-1141-3
Christoforidis JB, Xie Z, Jiang A, Wang J, Pratt C, Gemensky-Metzler A, Abdel-Rasoul M, Roy S, Liu Z (2013) Serum levels of intravitreal bevacizumab after vitrectomy, lensectomy and non-surgical controls. Curr Eye Res 38(7):761–766. doi:10.3109/02713683.2013.763988
Acknowledgements
The authors do not have any proprietary interest in the material described in this manuscript and financial support was not received for this study.
Funding
This study was supported by Soonchunhyang University Research Fund. No grants or sponsoring organizations were involved in the work presented in this submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no financial conflicts of interest.
Rights and permissions
About this article
Cite this article
Park, D.H., Sun, H.J. & Lee, S.J. A comparison of responses to intravitreal bevacizumab, ranibizumab, or aflibercept injections for neovascular age-related macular degeneration. Int Ophthalmol 37, 1205–1214 (2017). https://doi.org/10.1007/s10792-016-0391-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-016-0391-4