Abstract
Background
Simethicone is an adjunct frequently used during bowel preparation before colonoscopy and currently there is no consensus on whether it should be recommended in standard bowel preparation. We performed a systematic review and meta-analysis to determine the effect simethicone has on bowel cleanliness, adenoma detection rate (ADR), and tolerability.
Methods
We searched the literature for studies that compared colon cleansing of patients that received standard bowel preparation alone and in combination with simethicone prior to colonoscopy. The primary outcomes were colon cleanliness, ADR, and tolerability.
Results
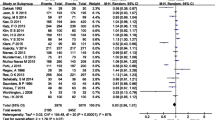
Sixteen randomized controlled trials with 5630 patients were included in meta-analysis. Overall, polyethylene glycol (PEG) with simethicone improves colon cleansing compared with PEG alone (odds ratio [OR] 1.48, CI 1.11 to 1.97, P = 0.008). This improvement was seen for single dosing (OR 1.83, CI 1.20 to 2.79, P = 0.005) but not for split dosing (OR 1.32, CI 0.72 to 2.43, P = 0.38). Overall, simethicone had no effect on ADR (OR 1.22, CI 0.81 to 1.83, P = 0.33), but in patients receiving single dosing, simethicone significantly increased ADR (OR 1.96, CI 1.22 to 3.16, P = 0.005). The rates of nausea (OR 0.96, CI 0.75 to 1.24, P = 0.75), vomiting (OR 1.00, CI 0.69 to 1.44, P = 0.99), and abdominal pain (OR 0.69, CI 0.40 to 1.18, P = 0.17) were not significantly different between PEG and PEG + simethicone cohorts. For abdominal bloating, the PEG cohort had greater odds of experiencing bloating than the PEG + simethicone cohort (OR 2.33, CI 1.70 to 3.20, P < 0.00001).
Conclusions
Simethicone improves colon cleanliness and ADR; however, this improvement is not seen in patients receiving split-dose PEG. Furthermore, simethicone decreases abdominal bloating but has no effect on nausea, vomiting, and abdominal pain. Simethicone may be a useful bowel preparation adjunct in patients unable to receive split-dose PEG.







Similar content being viewed by others
References
Saltzman JR, Cash BD, Pasha SF, Early DS, Raman Muthusamy V, Khashab MA, Chathadi KV, Fanelli RD, Chandrasekhara V, Lightdale JR, Fonkalsrud L, Shergill AK, Hwang JH, Decker GA, Jue TL, Sharaf R, Fisher DA, Evans JA, Foley K, Shaukat A, Eloubeidi MA, Faulx AL, Wang A, Acosta RD (2015) Bowel preparation before colonoscopy. Gastrointest Endosc 81:781–794. https://doi.org/10.1016/j.gie.2014.09.048
Winawer SJ (2007) Colorectal cancer screening. Best Pract Res Clin Gastroenterol 21:1031–1048. https://doi.org/10.1016/j.bpg.2007.09.004
Rex D, Petrini J, Baron T, Chak A, Cohen J, Deal S (2006) Quality indicators for colonoscopy. Am J Gastroenterol 101:1200–1208. https://doi.org/10.1111/j.1572-0241.2006.00673.x
Park S, Lim YJ (2014) Adjuncts to colonic cleansing before colonoscopy. World J Gastroenterol 20:2735–2740. https://doi.org/10.3748/wjg.v20.i11.2735
Brečević L, Bošan-Kilibarda I, Strajnar F (1994) Mechanism of antifoaming action of simethicone. J Appl Toxicol 14:207–211
Benmassaoud A, Parent J (2018) Canadian association of gastroenterology position statement on the impact of simethicone on endoscope reprocessing. J Can Assoc Gastroenterol 1:40–42
Wu L, Cao Y, Liao C, Huang J, Gao F (2011) Systematic review and meta-analysis of randomized controlled trials of Simethicone for gastrointestinal endoscopic visibility. Scand J Gastroenterol. https://doi.org/10.3109/00365521.2010.525714
Ofstead CL, Wetzler HP, Johnson EA, Heymann OL, Maust TJ, Shaw MJ (2016) Simethicone residue remains inside gastrointestinal endoscopes despite reprocessing. Am J Infect Control 44:1237–1240. https://doi.org/10.1016/j.ajic.2016.05.016
Higgins JPT, Green S (2011) Cochrane handbook for systematic reviews of interventions. Wiley, New York
Wieszczy P, Regula J, Kaminski MF (2017) Adenoma detection rate and risk of colorectal cancer. Best Pract Res Clin Gastroenterol 31:441–446. https://doi.org/10.1016/j.bpg.2017.07.002
Manes G, Fontana P, De Nucci G, Radaelli F, Hassan C, Ardizzone S (2015) Colon cleansing for colonoscopy in patients with ulcerative colitis: efficacy and acceptability of a 2-L PEG plus bisacodyl versus 4-L PEG. Inflamm Bowel Dis 21:2137–2144. https://doi.org/10.1097/MIB.0000000000000463
Collaboration C (2014) Review manager (Version 5.3) [computer software]. Denmark Nord Cochrane Cent, Copenhagen
Bai Y, Fang J, Zhao SB, Wang D, Li YQ, Shi RH, Sun ZQ, Sun MJ, Ji F, Si JM, Li ZS (2018) Impact of preprocedure simethicone on adenoma detection rate during colonoscopy: a multicenter, endoscopist-blinded randomized controlled trial. Endoscopy 50:128–136. https://doi.org/10.1055/s-0043-119213
Cesaro P, Hassan C, Spada C, Petruzziello L, Vitale G, Costamagna G (2013) A new low-volume isosmotic polyethylene glycol solution plus bisacodyl versus split-dose 4L polyethylene glycol for bowel cleansing prior to colonoscopy: a randomised controlled trial. Dig Liver Dis 45:23–27. https://doi.org/10.1016/j.dld.2012.07.011
Matro R, Tupchong K, Daskalakis C, Gordon V, Katz L, Kastenberg D (2012) The effect on colon visualization during colonoscopy of the addition of simethicone to polyethylene glycol–electrolyte solution: a randomized single-blind study. Clin Transl Gastroenterol 3:e26–e28. https://doi.org/10.1038/ctg.2012.16
Pontone S, Angelini R, Standoli M, Patrizi G, Culasso F, Pontone P, Redler A (2011) Low-volume plus ascorbic acid vs high-volume plus simethicone bowel preparation before colonoscopy. World J Gastroenterol 17:4689–4695. https://doi.org/10.3748/wjg.v17.i42.4689
Yoo IK, Jeen YT, Kang SH, Lee JH, Kim SH, Lee JM, Choi HS, Kim ES, Keum B, Chun HJ, Lee HS, Kim CD (2016) Improving of bowel cleansing effect for polyethylene glycol with ascorbic acid using simethicone: a randomized controlled trial. Medicine (U S). https://doi.org/10.1097/MD.0000000000004163
Zhang S, Zheng D, Wang J, Wu J, Lei P, Luo Q, Wang L, Zhang B, Wang H, Cui Y, Chen M (2018) Simethicone improves bowel cleansing with low-volume polyethylene glycol: a multicenter randomized trial. Endoscopy 50:412–422. https://doi.org/10.1055/s-0043-121337
Zorzi M, Valiante F, Germanà B, Baldassarre G, Coria B, Rinaldi M, Heras Salvat H, Carta A, Bortoluzzi F, Cervellin E, Polo ML, Bulighin G, Azzurro M, Di Piramo D, Turrin A, Monica F (2016) Comparison between different colon cleansing products for screening colonoscopyA noninferiority trial in population-based screening programs in Italy. Endoscopy 48:223–231. https://doi.org/10.1055/s-0035-1569574
Valiante F, Bellumat A, De Bona M, De Boni M (2013) Bisacodyl plus split 2-L polyethylene glycol-citrate-simethicone improves quality of bowel preparation before screening colonoscopy. World J Gastroenterol 19:5493–5499. https://doi.org/10.3748/wjg.v19.i33.5493
Gentile M, De Rosa M, Cestaro G, Forestieri P (2013) 2 L PEG plus ascorbic acid versus 4 L PEG plus simethicon for colonoscopy preparation: a randomized single-blind clinical trial. Surg Laparosc Endosc Percutan Tech 23:276–280. https://doi.org/10.1097/SLE.0b013e31828e389d
Lazzaroni M, Petrillo M, Desideri S, Porro GB (1993) Efficacy and tolerability of polyethylene glycol-electrolyte lavage solution with and without simethicone in the preparation of patients with inflammatory bowel disease for colonoscopy. Aliment Pharmacol Ther 7:655–659. https://doi.org/10.1111/j.1365-2036.1993.tb00148.x
Parente F, Vailati C, Bargiggia S, Manes G, Fontana P, Masci E, Arena M, Spinzi G, Baccarin A, Mazzoleni G, Testoni PA (2015) 2-Litre polyethylene glycol-citrate-simethicone plus bisacodyl versus 4-litre polyethylene glycol as preparation for colonoscopy in chronic constipation. Dig Liver Dis 47:857–863. https://doi.org/10.1016/j.dld.2015.06.008
Repici A, Cestari R, Annese V, Biscaglia G, Vitetta E, Minelli L, Trallori G, Orselli S, Andriulli A, Hassan C (2012) Randomised clinical trial: low-volume bowel preparation for colonoscopy: a comparison between two different PEG-based formulations. Aliment Pharmacol Ther 36:717–724. https://doi.org/10.1111/apt.12026
Spada C, Cesaro P, Bazzoli F, Saracco GM, Cipolletta L, Buri L, Crosta C, Petruzziello L, Ceroni L, Fuccio L, Giordanino C, Elia C, Rotondano G, Bianco MA, Simeth C, Consalvo D, De Roberto G, Fiori G, Campanale M, Costamagna G (2017) Evaluation of Clensia®, a new low-volume PEG bowel preparation in colonoscopy: multicentre randomized controlled trial versus 4L PEG. Dig Liver Dis 49:651–656. https://doi.org/10.1016/j.dld.2017.01.167
McNally R, Maydonovitch CL, Wong KH (1989) The effect of simethicone on colonic visibility after night-prior colonic lavage. J Clin Gastroenterol 11:650–652. https://doi.org/10.1097/00004836-198912000-00010
Shaver WA, Storms P, Peterson WL (1988) Improvement of oral colonic lavage with supplemental simethicone. Dig Dis Sci 33:185–188. https://doi.org/10.1007/BF01535731
McDonald GB, O’Leary R, Stratton C (1978) Pre-endoscopic use of oral simethicone. Gastrointest Endosc 24:283
Kilgore TW, Abdinoor AA, Szary NM, Schowengerdt SW, Yust JB, Choudhary A, Matteson ML, Puli SR, Marshall JB, Bechtold ML (2011) Bowel preparation with split-dose polyethylene glycol before colonoscopy: a meta-analysis of randomized controlled trials. Gastrointest Endosc 73:1240–1245. https://doi.org/10.1016/J.GIE.2011.02.007
Martel M, Barkun AN, Menard C, Restellini S, Kherad O, Vanasse A (2015) Split-dose preparations are superior to day-before bowel cleansing regimens: a meta-analysis. Gastroenterology 149:79–88. https://doi.org/10.1053/J.GASTRO.2015.04.004
Bucci C, Rotondano G, Hassan C, Rea M, Bianco MA, Cipolletta L, Ciacci C, Marmo R (2014) Optimal bowel cleansing for colonoscopy: split the dose! A series of meta-analyses of controlled studies. Gastrointest Endosc 80:566–576. https://doi.org/10.1016/J.GIE.2014.05.320
Kovaleva J, Peters FTM, van der Mei Mei HC, Degener JE (2013) Transmission of infection by flexible gastrointestinal endoscopy and bronchoscopy. Clin Microbiol Rev 26:230–253. https://doi.org/10.1128/CMR.00085-12
Gastroenterol CO (2016) What level of bowel prep quality requires early repeat colonoscopy: systematic review and meta-analysis of the impact of preparation quality on adenoma detection rate Brian. Am J Gastrointerol 31:81–88. https://doi.org/10.1038/ajg.2014.232.What
Altintaş E, Üçbilek E, Sezgin O, Sayici Y (2008) Alverine citrate plus simethicone reduces cecal intubation time in colonoscopy: a randomized study. Turk J Gastroenterol 19:174–179
Tongprasert S, Sobhonslidsuk A, Rattanasiri S (2009) Improving quality of colonoscopy by adding simethicone to sodium phosphate bowel preparation. World J Gastroenterol 15:3032–3037. https://doi.org/10.3748/wjg.15.3032
Funding
The study was funded $6000 by the Department of Surgery Summer Studentship from the Department of Surgery, University of Alberta, Edmonton, Alberta, Canada.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Dr. Sultanian serves as a consultant for Boston Scientific. Muhammad Moolla and Drs. Dang, Shaw, Dang, Tian, and Karmali have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moolla, M., Dang, J.T., Shaw, A. et al. Simethicone decreases bloating and improves bowel preparation effectiveness: a systematic review and meta-analysis. Surg Endosc 33, 3899–3909 (2019). https://doi.org/10.1007/s00464-019-07066-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-019-07066-5