Abstract
Background
Intravitreal injections of ranibizumab are the standard of care for neovascular age-related macular degeneration (AMD). In clinical trials, comparable efficacy has been shown for either monthly injections or as needed injections upon monthly controls. Unlike in trial settings, treatment in clinical routine is often delayed by complex approval procedures of health insurance and limited short-term surgical capacities.
Methods
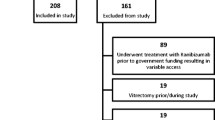
Eighty-nine patients with neovascular AMD were followed for 12 months. Early treatment diabetic retinopathy study (ETDRS) visual acuity (VA), Radner reading VA and spectral domain optical coherence tomography were performed monthly, with additional fluorescein angiography if needed. After an initial loading phase of three consecutive monthly intravitreal injections with ranibizumab, re-injections were performed when recurrent activity of choroidal neovascularization (CNV) was detected.
Results
After an initial increase to a value of +5.0 ± 11.87 ETDRS letters from baseline, VA constantly decreased over 12 months to a value of −0.66 ± 16.82 ETDRS letters below baseline. Central retinal thickness (CRT) decreased from a value of 438.1 ± 191.4 μm at baseline to a value of 289.9 ± 138.6 μm after initial therapy and stabilized at a value of 322.4 ± 199.5 μm. Loss of VA during latency between indication to treat and treatment was significantly greater than re-gain of VA after re-initiation of therapy (−2.2 ± 5.0 versus 0.4 ± 7.4 letters; p = 0.046).
Conclusions
Latency between indication to treat and treatment is responsible for irreversible VA deterioration. A successful PRN treatment regimen for neovascular AMD requires immediate access to therapy after indication.




Similar content being viewed by others
References
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355:1419–1431
Regillo CD, Brown DM, Abraham P, Yue H, Ianchulev T, Schneider S, Shams N (2008) Randomized, double-masked, sham-controlled trial of ranibizumab for neovascular age-related macular degeneration: PIER Study year 1. Am J Ophthalmol 145:239–248
Martin DF, Maguire MG, Ying GS, Grunwald JE, Fine SL, Jaffe GJ (2011) Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med 364:1897–1908
Muether PS, Hermann MM, Koch K, Fauser S (2011) Delay between medical indication to anti-VEGF treatment in age-related macular degeneration can result in a loss of visual acuity. Graefes Arch Clin Exp Ophthalmol 249:633–637
Bashshur ZF, Haddad ZA, Schakal AR, Jaafar RF, Saad A, Noureddin BN (2009) Intravitreal bevacizumab for treatment of neovascular age-related macular degeneration: the second year of a prospective study. Am J Ophthalmol 148:59–65
Lalwani GA, Rosenfeld PJ, Fung AE, Dubovy SR, Michels S, Feuer W, Davis JL, Flynn HW Jr, Esquiabro M (2009) A variable-dosing regimen with intravitreal ranibizumab for neovascular age-related macular degeneration: year 2 of the PrONTO Study. Am J Ophthalmol 148:43–58
Keane PA, Liakopoulos S, Chang KT, Wang M, Dustin L, Walsh AC, Sadda SR (2008) Relationship between optical coherence tomography retinal parameters and visual acuity in neovascular age-related macular degeneration. Ophthalmology 115:2206–2214
Heimes B, Lommatzsch A, Zeimer M, Gutfleisch M, Spital G, Dietzel M, Pauleikhoff D (2011) Long-term visual course after anti-VEGF therapy for exudative AMD in clinical practice evaluation of the German reinjection scheme. Graefes Arch Clin Exp Ophthalmol 249:639–644. doi:10.1007/s00417-010-1524-5
Gerding H, Loukopoulos V, Riese J, Hefner L, Timmermann M (2011) Results of flexible ranibizumab treatment in age-related macular degeneration and search for parameters with impact on outcome. Graefes Arch Clin Exp Ophthalmol 249:653–662
Acknowledgments
This study was supported by the Koeln Fortune Program, Faculty of Medicine, University of Cologne. The authors thank Susanne Nelles for database maintenance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Koeln Fortune Program, Faculty of Medicine, University of Cologne
Financial relation: Bernd Kirchhof and Sascha Fauser have a consultant and advisory board relationship with Novartis Pharma, Nuernberg, Germany outside the submitted work.
Registered at ClinicalTrials.gov identifier: NCT01213667.
Authors have full control of all primary data and agree to allow Graefes Archive for Clinical and Experimental Ophthalmology to review the data on request.
Registered at ClinicalTrials.gov identifier: NCT01213667.
Rights and permissions
About this article
Cite this article
Muether, P.S., Hoerster, R., Hermann, M.M. et al. Long-term effects of ranibizumab treatment delay in neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 251, 453–458 (2013). https://doi.org/10.1007/s00417-012-2038-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-012-2038-0