Abstract
Background
Sodium hyaluronate (SH) is used in patients with dry eye. We evaluated the efficacy and safety of SH and carboxymethylcellulose (CMC) in the treatment of dry eye syndrome with superficial keratitis.
Methods
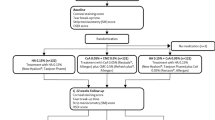
A total of 22 patients with moderate dry eye and superficial keratitis were enrolled in a prospective, randomised, masked-observer, parallel-group, single-centre study. Patients were randomly assigned to a 0.18% SH or 1% CMC solution for a 2-month period. In addition to the commonly assessed parameters in patients with dry eye (among others symptoms and corneal staining with fluorescein), flow cytometry analysis of CD44, HLA DR expressions in impression cytology was investigated as a potential efficacy parameter.
Results
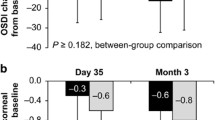
Both treatments improved the symptoms and ocular surface and were well tolerated. SH significantly (p<0.05) decreased CD44 values compared with CMC. Comfort was significantly (P<0.05) better in the SH group than that in the CMC group throughout the study. Recovery in keratitis (type, extent and depth) and symptoms were faster in the SH group than in the CMC group. Blurred vision was reported by patients in the CMC group only.
Conclusions
SH was well tolerated and tended to show a faster efficacy than did the CMC-based formulation in patients with moderate dry eye and superficial keratitis. SH could therefore advantageously be prescribed from the early stages of dry eye disease. This study also showed that flow cytometry in impression cytology specimens is a reliable tool for exploring the ocular surface at the epithelial level and that CD44, in addition to HLA DR, could be an interesting endpoint for future trials in dry eye syndrome with products based on SH.




Similar content being viewed by others
References
Aziz KE, Wakefield D (1995) In vivo and in vitro expression of adhesion molecules by peripheral blood lymphocytes from patients with primary Sjogren’s syndrome: culture-associated enhancement of LECAM-1 and CD44. Rheumatol Int 15:69–74
Baudouin C (2001) The pathology of dry eye. Surv Ophthalmol 45:S211–S220
Baudouin C, Haouat N, Brignole F, Bayle J, Gastaud P (1992) Immunopathological findings in conjunctival cells using immunofluorescence staining of impression cytology specimens. Br J Ophthalmol 76:545–549
Baudouin C, Brignole F, Becquet F, Pisella PJ, Goguel A (1997) Flow cytometry in impression cytology specimens. Invest Ophthalmol Vis Sci 38:1458–1464
Bernatchez SF, Camber O, Tabatabay C, Gurny R (1993) Use of hyaluronic acid in ocular therapy. In: Edman P (ed) Biopharmaceutics of ocular drug delivery, CRC, Boca Raton, pp 105–120
Bernatchez SF, Tabatabay C, Gurny R (1993) Sodium hyaluronate 0.25% used as a vehicle increases the bioavailability of topically administered gentamicin. Graefes Arch Clin Exp Ophthalmol 231:157–171
Brignole F, De Saint-Jean M, Goldschild M, Becquet F, Goguel A, Baudouin C (1998) Expression of Fas–Fas ligand antigens and apoptotic marker AP02.7 by the human conjunctival epithelium, positive correlation with Class II HLA DR expression in inflammatory ocular surface disorders. Exp Eye Res 67:687–697
Cerulli L, Trematerra M, Zapelloni A, D’Ambrosi E, Corsi A (1993) Clinical evaluation of L02A eye drop versus control eye drop in patients suffering from lacrymal hyposecretion. Ann Ottalmol Clin Ocul 119:49–55
Condon PI, McEwen CG, Wright M, Mackintosh G, Prescott RJ, McDonald C (1999) Double-blind, randomised, placebo-controlled, crossover, multicentre study to determine the efficacy of a 0.1% (w/v) sodium hyaluronate solution (Fermavisc) in the treatment of dry eye syndrome. Br J Ophthalmol 83:1121–1124
Fukuda M (1991) Lysosomal membrane glycoproteins. Structure, biosynthesis, and intracellular trafficking. J Biol Chem 266:21327–21330
Gomes JAP, Amankwah R, Powell-Richards A, Dua HS (2004) Sodium hyaluronate (hyaluronic acid) promotes migration of human corneal epithelial cells in vitro. Br J Ophthalmol 88:821–825
Gurny R, Ryser JE, Tabatabay C, Martenet M, Edman P, Camber O (1990) Precorneal residence time in humans of sodium hyaluronate as measured by gamma scintigraphy. Graefes Arch Clin Exp Ophthalmol 228:510–512
Hamano T, Horimoto K, Lee M, Komemushi S (1996) Sodium hyaluronate eye drops enhance tear film stability. Jpn J Ophthalmol 40:62–65
Iester A, Brezzo MV, Rolando M (1993) Double-masked clinical trial of an unpreserved hyaluronic acid based eye drop in dry eye syndromes. Ann Ottalmol Clin Ocul 119:17–31
Lemp MA (1995) Report of the National Eye Institute/Industry Workshop on clinical trials in dry eyes. CLAO J 21:221–232
Levesque MC, Mackin DA, Fleming JA, St Clair EW (2000) Serum levels of soluble CD44 in primary Sjogren’s syndrome. J Rheumatol 27:1444–1449
Limberg MB, Mc Caa C, Kissling GE, Kaufman HE (1987) Topical application of hyaluronic acid and chondroitin sulfate in the treatment of dry eyes. Am J Ophthalmol 103:194–197
McMonnies CW (1986) Key questions in a dry eye history. J Am Optom Assoc 57:512–517
McMonnies CW, Ho A (1987) Patient history in screening for dry eye conditions. J Am Optom Assoc 58:296–301
McMonnies CW, Ho A (1987) Responses to a dry eye questionnaire from a normal population. J Am Optom Assoc 58:588–591
Mengher L, Pandher K, Bron AJ, Davey CC (1986) Effect of sodium hyaluronate (0.1%) on break-up time (NIBUT) in patients with dry eyes. Br J Ophthalmol 70:442–447
Meyer K (1958) Chemical structure of hyaluronic acid. Fed Proc 17:1075–1077
Miyazaki T, Miyauchi S, Nakamura T, Takeshita S, Horie K (1996) The effect of sodium hyaluronate on the growth of rabbit corneal epithelial cells in vitro. J Ocul Pharmacol Ther 12:409–415
Nagy H, Goda K, Fenyvesi F, Bacso Z, Szilasi M, Kappelmayer J, Lustyik G, Cianfriglia M, Szabo G Jr (2004) Distinct groups of multidrug resistance modulating agents are distinguished by competition of P-glycoprotein-specific antibodies. Biochem Biophys Res Commun 315:942–949
Nakamura M, Hikida M, Nakano T (1992) Concentration and molecular weight dependency of rabbit corneal epithelial wound healing on hyaluronan. Curr Eye Res 11:981–986
Nakamura M, Nakano T, Mibu H, Hikida M (1995) Hyaluronan stimulates epithelial wound healing in the rabbit cornea. Folia Ophthalmol Jpn 46:1256–1260
Orsoni JG, Chiari M, Guazi A, De Carli M, Guidolin D (1988) Efficacy of hyaluronic acid solution in the treatment of sicca syndrome. Ophtalmologie 2:355–357
Pisella PJ, Debbasch C, Hamard P, Creuzot-Garcher C, Rat P, Brignole F, Baudouin C (2004) Conjunctival proinflammatory and proapoptotic effects of latanoprost and preserved and unpreserved timolol: an ex vivo and in vitro study. Invest Ophthalmol Vis Sci 45:1360–1368
Sand BB, Marner K, Norn MS (1989) Sodium hyaluronate in the treatment of keratoconjunctivitis sicca. A double masked clinical trial. Acta Ophthalmol 67:181–183
Scrivanti M, Taiti L, Mencucci R., Bardi L, Salvi G (1996) Syndrome sec oculaire: utilisation d’un nouveau substitut lacrymal (LO2A). Ophtalmologie 10:24–27
Shimmura S, Ono M, Shinozaki K, Toda I, Takamura E, Mashima Y, Tsubota K (1995) Sodium hyaluronate eye drops in the treatment of dry eyes. Br J Ophthalmol 79:1007–1011
Skubitz KM, Campbell KD, Skubitz AP (2000) CD63 associates with CD11/CD18 in large detergent-resistant complexes after translocation to the cell surface in human neutrophils. FEBS Lett 469:52–56
Taiti L, Scrivanti M, Mencucci R, Bardi L, Salvi G (1996) A double-masked crossover comparison of a new artificial tear preparation containing hyaluronic acid (L02A) and a reference artificial tear preparation containing hydroxypropyl-methylcellulose in dry-eye syndrome treatment. Ann Ottalmol Clin Ocul 119:33–46
Tiffany JM (1994) Viscoelastic properties of human tears and polymer solutions. Adv Exp Med Biol 350:267–270
Zhu SN, Nolle B, Duncker G (1997) Expression of adhesion molecule CD44 on human corneas. Br J Ophthalmol 81:80–84
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brignole, F., Pisella, PJ., Dupas, B. et al. Efficacy and safety of 0.18% sodium hyaluronate in patients with moderate dry eye syndrome and superficial keratitis. Graefe's Arch Clin Exp Ophthalmol 243, 531–538 (2005). https://doi.org/10.1007/s00417-004-1040-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-004-1040-6