Abstract
Purpose
Metastatic disease is a major cause of mortality in colorectal cancer patients. Even after complete resection of isolated liver metastases, recurrence develops in the majority of patients. Therefore, development of strategies to prevent recurrent liver metastases is of major clinical importance. The present prospectively randomised phase III trial investigates the efficiency of active specific immunotherapy (ASI) after liver resection for hepatic metastases of colorectal cancer.
Methods
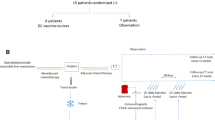
Patients with histologically confirmed liver metastases from colorectal cancer were randomised to the vaccination or control group. After complete resection of liver metastases, patients randomised to the vaccination group received six doses of Newcastle disease virus (NDV) infected autologous tumour cell vaccine (ATV-NDV). The primary end-point was overall survival, secondary end-points were disease-free survival and metastases-free survival.
Results
Fifty-one patients were enrolled in the study with 50 patients available for analysis. The follow-up period was 116.1 ± 23.8 month in the vaccination arm and 112.4 ± 18.5 month in the control group. In the total patient group, no differences in the primary and secondary end-points were detected. Most interestingly, subgroup analysis revealed a significant advantage for vaccinated colon cancer patients with respect to overall survival [hazard ratio: 3.3; 95%, confidence interval (CI): 1.0–10.4; P = 0.042] and metastases-free survival (hazard ratio: 2.7; 95%, CI: 1.0–7.4; P = 0.047) in the intention-to-treat analysis.
Conclusion
Active specific immunotherapy in unselected colorectal cancer patients was not effective for prevention of recurrent metastatic disease. However, in colon cancer patients, ASI with ATV-NDV appears to be beneficial prolonging overall and metastases-free survival.



Similar content being viewed by others
References
Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A, Feuer EJ, Thun MJ (2005) Cancer statistics. CA Cancer J Clin 55:10–30
Ballantyne GH, Quin J (1993) Surgical treatment of liver metastases in patients with colorectal cancer. Cancer 71:4252–4266
Bengmark S, Hafstrom L (1969) The natural history of primary and secondary malignant tumors of the liver, I. The prognosis for patients with hepatic metastases from colonic and rectal carcinoma by laparotomy. Cancer 23:198–202
de Brauw LM, van de Velde CJ, Bouwhuis-Hoogerwerf ML, Zwaveling A (1987) Diagnostic evaluation and survival analysis of colorectal cancer patients with liver metastases. J Surg Oncol 34:81–86
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH (1999) Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 230:309–318
Foster JH (1978) Survival after liver resection for secondary tumors. Am J Surg 135:389–394
Scheele J, Stangl R, Altendorf-Hofmann A (1990) Hepatic metastases from colorectal carcinoma: impact of surgical resection on the natural history. Br J Surg 77:1241–1246
Fernandez-Trigo V, Shamsa F, Sugarbaker PH (1995) Repeat liver resections from colorectal metastasis. Repeat hepatic metastases registry. Surgery 117:296–304
Figueras J, Valls C, Rafecas A, Fabregat J, Ramos E, Jaurrieta E (2001) Resection rate and effect of postoperative chemotherapy on survival after surgery for colorectal liver metastases. Br J Surg 88:980–985
Biasco G, Derenzini E, Grazi G, Ercolani G, Ravaioli M, Pantaleo MA, Brandi G (2006) Treatment of hepatic metastases from colorectal cancer: many doubts, some certainties. Cancer Treat Rev 17:214–228
Lehner B, Schlag P, Liebrich W, Schirrmacher V (1990) Postoperative active specific immunization in curatively resected colorectal cancer patients with a virus-modified autologous tumor cell vaccine. Cancer Immunol Immunother 32:173–178
Haas C, Ertel C, Gerhards R, Schirrmacher V (1998) Introduction of adhesive and costimulatory immune functions into tumor cells by infection with Newcastle disease virus. Int J Oncol 13:1105–1115
Washburn B, Schirrmacher V (2002) Human tumor cell infection by Newcastle disease virus leads to upregulation of HLA and cell adhesion molecules and to induction of interferons, chemokines and finally apoptosis. Int J Oncol 21:85–93
Termeer CC, Schirrmacher V, Brocker EB, Becker JC (2000) Newcastle disease virus infection induces B7-1/B7-2-independent T-cell costimulatory activity in human melanoma cells. Cancer Gene Ther 7:316–323
Ertel C, Millar NS, Emmerson PT, Schirrmacher V, von Hoegen P (1993) Viral hemagglutinin augments peptide-specific cytotoxic T cell responses. Eur J Immunol 23:2592–2596
Bai L, Koopmann J, Fiola C, Fournier P, Schirrmacher V (2002) Dendritic cells pulsed with viral oncolysates potently stimulate autologous T cells from cancer patients. Int J Oncol 21:685–694
Schirrmacher V, Bai L, Umansky V, Yu L, Xing Y, Qian Z (2000) Newcastle disease virus activates macrophages for anti-tumor activity. Int J Oncol 16:363–373
Washburn B, Weigand MA, Grosse-Wilde A, Janke M, Stahl H, Rieser E, Sprick MR, Schirrmacher V, Walczak H (2003) TNF-related apoptosis-inducing ligand mediates tumoricidal activity of human monocytes stimulated by Newcastle disease virus. J Immunol 170:1814–1821
Zeng J, Fournier P, Schirrmacher V (2002) Induction of interferon-alpha and tumor necrosis factor-related apoptosis-inducing ligand in human blood mononuclear cells by hemagglutinin-neuraminidase but not F protein of Newcastle disease virus. Virology 297:19–30
Lokuta MA, Maher J, Noe KH, Pitha PM, Shin ML, Shin HS (1996) Mechanisms of murine RANTES chemokine gene induction by Newcastle disease virus. J Biol Chem 271:13731–13738
Schirrmacher V (2005) Clinical trials of antitumor vaccination with an autologous tumor cell vaccine modified by virus infection: improvement of patient survival based on improved antitumor immune memory. Cancer Immunol Immunother 54:587–598
Schlag P, Manasterski M, Gerneth T, Hohenberger P, Dueck M, Herfarth C, Liebrich W, Schirrmacher V (1992) Active specific immunotherapy with Newcastle-disease-virus-modified autologous tumor cells following resection of liver metastases in colorectal cancer, First evaluation of clinical response of a phase II-trial. Cancer Immunol Immunother 35:325–330
Vermorken JB, Claessen AM, van Tinteren H, Gall HE, Ezinga R, Meijer S, Scheper RJ, Meijer CJ, Bloemena E, Ransom JH, Hanna MG Jr., Pinedo HM (1999) Active specific immunotherapy for stage II and stage III human colon cancer: a randomised trial. Lancet 353:345–350
Hoover HC Jr, Brandhorst JS, Peters LC, Surdyke MG, Takeshita Y, Madariaga J, Muenz LR, Hanna MG Jr (1993) Adjuvant active specific immunotherapy for human colorectal cancer: 6.5-year median follow-up of a phase III prospectively randomized trial. J Clin Oncol 11:390–399
Rimoldi M, Chieppa M, Salucci V, Avogadri F, Sonzogni A, Sampietro GM, Nespoli A, Viale G, Allavena P, Rescigno M (2005) Intestinal immune homeostasis is regulated by the crosstalk between epithelial cells and dendritic cells. Nat Immunol 6:507–514
Johansson C, Kelsall BL (2005) Phenotype and function of intestinal dendritic cells. Semin Immunol 17:284–294
Hawkins N, Norrie M, Cheong K, Mokany E, Ku SL, Meagher A, O’Connor T, Ward R (2002) CpG island methylation in sporadic colorectal cancers and its relationship to microsatellite instability. Gastroenterology 122:1376–1387
Heicappell R, Schirrmacher V, von Hoegen P, Ahlert T, Appelhans B (1986) Prevention of metastatic spread by postoperative immunotherapy with virally modified autologous tumor cells. I. Parameters for optimal therapeutic effects. Int J Cancer 37:569–577
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schulze, T., Kemmner, W., Weitz, J. et al. Efficiency of adjuvant active specific immunization with Newcastle disease virus modified tumor cells in colorectal cancer patients following resection of liver metastases: results of a prospective randomized trial. Cancer Immunol Immunother 58, 61–69 (2009). https://doi.org/10.1007/s00262-008-0526-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-008-0526-1