Abstract
Rationale
Preliminary evidence suggests that cannabidiol (CBD) may be effective in the treatment of neurodegenerative disorders; however, CBD has never been evaluated for the treatment of cognitive impairments associated with schizophrenia (CIAS).
Objective
This study compared the cognitive, symptomatic, and side effects of CBD versus placebo in a clinical trial.
Methods
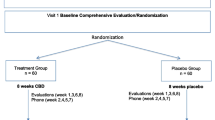
This study was a 6-week, randomized, placebo-controlled, parallel group, fixed-dose study of oral CBD (600 mg/day) or placebo augmentation in 36 stable antipsychotic-treated patients diagnosed with chronic schizophrenia. All subjects completed the MATRICS Consensus Cognitive Battery (MCCB) at baseline and at end of 6 weeks of treatment. Psychotic symptoms were assessed using the Positive and Negative Syndrome Scale (PANSS) at baseline and biweekly.
Results
There was no main effect of time or drug on MCCB Composite score, but a significant drug × time effect was observed (p = 0.02). Post hoc analyses revealed that only placebo-treated subjects improved over time (p = 0.03). There was a significant decrease in PANSS Total scores over time (p < 0. 0001) but there was no significant drug × time interaction (p = 0.18). Side effects were similar between CBD and placebo, with the one exception being sedation, which was more prevalent in the CBD group.
Conclusions
At the dose studied, CBD augmentation was not associated with an improvement in MCCB or PANSS scores in stable antipsychotic-treated outpatients with schizophrenia. Overall, CBD was well tolerated with no worsening of mood, suicidality, or movement side effects.
Trial registration



Similar content being viewed by others
References
Barnes TR (1989) A rating scale for drug-induced akathisia. Br J Psychiatry 154:672–676
Bhattacharyya S, Morrison PD, Fusar-Poli P, Martin-Santos R, Borgwardt S, Winton-Brown T, Nosarti C, O'Carroll CM, Seal M, Allen P, Mehta MA, Stone JM, Tunstall N, Giampietro V, Kapur S, Murray RM, Zuardi AW, Crippa JA, Atakan Z, McGuire PK (2010) Opposite effects of delta-9-tetrahydrocannabinol and cannabidiol on human brain function and psychopathology. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 35:764–774
Boggs DL, Carlson J, Cortes-Briones J, Krystal JH, Cyril D’Souza D (2014) Going up in smoke? A review of nAChRs-based treatment strategies for improving cognition in schizophrenia. Curr Pharm Des 20:5077–5092
Bradley SR, Lameh J, Ohrmund L, Son T, Bajpai A, Nguyen D, Friberg M, Burstein ES, Spalding TA, Ott TR, Schiffer HH, Tabatabaei A, McFarland K, Davis RE, Bonhaus DW (2010) AC-260584, an orally bioavailable M(1) muscarinic receptor allosteric agonist, improves cognitive performance in an animal model. Neuropharmacology 58:365–373
Brandt J (1991) The Hopkins Verbal Learning Test: development of a new memory test with six equivalent forms. Clin Neuropsychol 5:125–142
Buchanan RW, Freedman R, Javitt DC, Abi-Dargham A, Lieberman JA (2007) Recent advances in the development of novel pharmacological agents for the treatment of cognitive impairments in schizophrenia. Schizophr Bull 33:1120–1130
D’Souza MS, Markou A (2012) Schizophrenia and tobacco smoking comorbidity: nAChR agonists in the treatment of schizophrenia-associated cognitive deficits. Neuropharmacology 62:1564–1573
D’Souza DC, Perry E, MacDougall L, Ammerman Y, Cooper T, Wu YT, Braley G, Gueorguieva R, Krystal JH (2004) The psychotomimetic effects of intravenous delta-9-tetrahydrocannabinol in healthy individuals: implications for psychosis. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 29:1558–1572
D’Souza DC, Abi-Saab WM, Madonick S, Forselius-Bielen K, Doersch A, Braley G, Gueorguieva R, Cooper TB, Krystal JH (2005) Delta-9-tetrahydrocannabinol effects in schizophrenia: implications for cognition, psychosis, and addiction. Biol Psychiatry 57:594–608
D’Souza DC, Radhakrishnan R, Perry E, Bhakta S, Singh NM, Yadav R, Abi-Saab D, Pittman B, Chaturvedi SK, Sharma MP, Bell M, Andrade C (2013) Feasibility, safety, and efficacy of the combination of D-serine and computerized cognitive retraining in schizophrenia: an international collaborative pilot study. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 38:492–503
Devane WA, Dysarz FA 3rd, Johnson MR, Melvin LS, Howlett AC (1988) Determination and characterization of a cannabinoid receptor in rat brain. Mol Pharmacol 34:605–613
Eggan SM, Lewis DA (2007) Immunocytochemical distribution of the cannabinoid CB1 receptor in the primate neocortex: a regional and laminar analysis. Cereb Cortex 17:175–191
ElSohly MA, Radwan MM, Gul W, Chandra S, Galal A (2017) Phytochemistry of Cannabis sativa L Phytocannabinoids. In: Kinghorn AD, Falk H, Gibbons S, Kobayashi J (eds) Progress in the chemistry of organic natural products, vol. 103. Phytocannabinoids: unraveling the complex chemistry and pharmacology of Cannabis sativa. Springer International Publishing, New York, pp 1–36
Fagherazzi EV, Garcia VA, Maurmann N, Bervanger T, Halmenschlager LH, Busato SB, Hallak JE, Zuardi AW, Crippa JA, Schroder N (2012) Memory-rescuing effects of cannabidiol in an animal model of cognitive impairment relevant to neurodegenerative disorders. Psychopharmacology 219:1133–1140
Gomes FV, Llorente R, Del Bel EA, Viveros MP, Lopez-Gallardo M, Guimaraes FS (2015) Decreased glial reactivity could be involved in the antipsychotic-like effect of cannabidiol. Schizophr Res 164:155–163
Guy W (1976) Abnormal involuntary movement scale (AIMS). ECDEU Assess Manual Psychopharmacol 338:534–537
Hallak JE, Machado-de-Sousa JP, Crippa JA, Sanches RF, Trzesniak C, Chaves C, Bernardo SA, Regalo SC, Zuardi AW (2010) Performance of schizophrenic patients in the Stroop Color Word Test and electrodermal responsiveness after acute administration of cannabidiol (CBD). Rev Bras Psiquiatr 32:56–61
Harvey PD, Bowie CR (2012) Cognitive enhancement in schizophrenia: pharmacological and cognitive remediation approaches. Psychiatr Clin North Am 35:683–698
Heinrichs RW, Zakzanis KK (1998) Neurocognitive deficit in schizophrenia: a quantitative review of the evidence. Neuropsychology 12:426–445
Hughes C, Kumari V, Soni W, Das M, Binneman B, Drozd S, O’Neil S, Mathew V, Sharma T (2003) Longitudinal study of symptoms and cognitive function in chronic schizophrenia. Schizophr Res 59:137–146
Ibeas Bih C, Chen T, Nunn AV, Bazelot M, Dallas M, Whalley BJ (2015) Molecular targets of cannabidiol in neurological disorders. Neurotherapeutics 12:699–730
Iuvone T, Esposito G, De Filippis D, Scuderi C, Steardo L (2009) Cannabidiol: a promising drug for neurodegenerative disorders? CNS Neurosci Ther 15:65–75
Izzo AA, Borrelli F, Capasso R, Di Marzo V, Mechoulam R (2009) Non-psychotropic plant cannabinoids: new therapeutic opportunities from an ancient herb. Trends Pharmacol Sci 30:515–527
Kay SR, Flszbein A, Opfer LA (1987) The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 13:261
Keefe RS, Eesley CE, Poe MP (2005) Defining a cognitive function decrement in schizophrenia. Biol Psychiatry 57:688–691
Keefe RS, Bilder RM, Harvey PD, Davis SM, Palmer BW, Gold JM, Meltzer HY, Green MF, del Miller D, Canive JM, Adler LW, Manschreck TC, Swartz M, Rosenheck R, Perkins DO, Walker TM, Stroup TS, McEvoy JP, Lieberman JA (2006) Baseline neurocognitive deficits in the CATIE schizophrenia trial. Neuropsychopharmacology 31:2033–2046
Koethe D, Giuffrida A, Schreiber D, Hellmich M, Schultze-Lutter F, Ruhrmann S, Klosterkotter J, Piomelli D, Leweke FM (2009) Anandamide elevation in cerebrospinal fluid in initial prodromal states of psychosis. Br J Psychiatry J Ment Sci 194:371–372
Leweke FM, Giuffrida A, Wurster U, Emrich HM, Piomelli D (1999) Elevated endogenous cannabinoids in schizophrenia. Neuroreport 10:1665–1669
Leweke FM, Piomelli D, Pahlisch F, Muhl D, Gerth CW, Hoyer C, Klosterkotter J, Hellmich M, Koethe D (2012) Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry 2:e94
Leweke FM, Hellmich M, Pahlisch F, Kranaster L, Koethe D (2014) Modulation of the endocannabiniod system as a potential new target in the treatment of schizophrenia. Schizophr Res 153:S47
Lingjaerde O, Ahlfors U, Bech P, Dencker S, Elgen K (1987) The UKU side effect rating scale: a new comprehensive rating scale for psychotropic drugs and a cross-sectional study of side effects in neuroleptic-treated patients. Acta Psychiatr Scand 76:1–100
Magen I, Avraham Y, Ackerman Z, Vorobiev L, Mechoulam R, Berry EM (2010) Cannabidiol ameliorates cognitive and motor impairments in bile-duct ligated mice via 5-HT1A receptor activation. Br J Pharmacol 159:950–957
McGuire P, Robson P, Cubala WJ, Vasile D, Morrison PD, Barron R, Taylor A, Wright S (2017) Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a Multicenter Randomized Controlled Trial. Am J Psychiatry 175(3):225–231
Mechoulam R, Parker LA (2013) The endocannabinoid system and the brain. Annu Rev Psychol 64:21–47
Moreira FA, Guimaraes FS (2005) Cannabidiol inhibits the hyperlocomotion induced by psychotomimetic drugs in mice. Eur J Pharmacol 512:199–205
Morgan CJ, Schafer G, Freeman TP, Curran HV (2010) Impact of cannabidiol on the acute memory and psychotomimetic effects of smoked cannabis: naturalistic study: naturalistic study [corrected]. Br J Psychiatry J Ment Sci 197:285–290
Murphy M, Mills S, Winstone J, Leishman E, Wager-Miller J, Bradshaw H, Mackie K (2017) Chronic adolescent Δ9-tetrahydrocannabinol treatment of male mice leads to long-term cognitive and behavioral dysfunction, which are prevented by concurrent cannabidiol treatment. Cannabis Cannabinoid Res 2:235–246
Nuechterlein KH, Green MF, Kern RS, Baade LE, Barch DM, Cohen JD, Essock S, Fenton WS, Frese FJ 3rd, Gold JM, Goldberg T, Heaton RK, Keefe RS, Kraemer H, Mesholam-Gately R, Seidman LJ, Stover E, Weinberger DR, Young AS, Zalcman S, Marder SR (2008) The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am J Psychiatry 165:203–213
O’Donnell CJ, Rogers BN, Bronk BS, Bryce DK, Coe JW, Cook KK, Duplantier AJ, Evrard E, Hajos M, Hoffmann WE, Hurst RS, Maklad N, Mather RJ, McLean S, Nedza FM, O’Neill BT, Peng L, Qian W, Rottas MM, Sands SB, Schmidt AW, Shrikhande AV, Spracklin DK, Wong DF, Zhang A, Zhang L (2010) Discovery of 4-(5-methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabicyclo[3.2.2]nonane (CP-810,123), a novel alpha 7 nicotinic acetylcholine receptor agonist for the treatment of cognitive disorders in schizophrenia: synthesis, SAR development, and in vivo efficacy in cognition models. J Med Chem 53:1222–1237
Radek RJ, Kohlhaas KL, Rueter LE, Mohler EG (2010) Treating the cognitive deficits of schizophrenia with alpha4beta2 neuronal nicotinic receptor agonists. Curr Pharm Des 16:309–322
Ranganathan M, D’Souza DC (2006) The acute effects of cannabinoids on memory in humans: a review. Psychopharmacology 188:425–444
Riedel G, Davies SN (2005) Cannabinoid function in learning, memory and plasticity. Handb Exp Pharmacol 168:445–477
Riley EM, McGovern D, Mockler D, Doku VC, OCeallaigh S, Fannon DG, Tennakoon L, Santamaria M, Soni W, Morris RG, Sharma T (2000) Neuropsychological functioning in first-episode psychosis—evidence of specific deficits. Schizophr Res 43:47–55
Rund BR, Melle I, Friis S, Larsen TK, Midboe LJ, Opjordsmoen S, Simonsen E, Vaglum P, McGlashan T (2004) Neurocognitive dysfunction in first-episode psychosis: correlates with symptoms, premorbid adjustment, and duration of untreated psychosis. Am J Psychiatry 161:466–472
Schatz AR, Lee M, Condie RB, Pulaski JT, Kaminski NE (1997) Cannabinoid receptors CB1 and CB2: a characterization of expression and adenylate cyclase modulation within the immune system. Toxicol Appl Pharmacol 142:278–287
Scuderi C, Filippis DD, Iuvone T, Blasio A, Steardo A, Esposito G (2009) Cannabidiol in medicine: a review of its therapeutic potential in CNS disorders. Phytother Res: PTR 23:597–602
Simpson G, Angus J (1970) A rating scale for extrapyramidal side effects. Acta Psychiatr Scand 45:11–19
Wechsler D (1955) Manual: Wechsler adult intelligence scale. Psychological Corp, New York
Yucel M, Bora E, Lubman DI, Solowij N, Brewer WJ, Cotton SM, Conus P, Takagi MJ, Fornito A, Wood SJ, McGorry PD, Pantelis C (2012) The impact of cannabis use on cognitive functioning in patients with schizophrenia: a meta-analysis of existing findings and new data in a first-episode sample. Schizophr Bull 38:316–330
Zuardi AW, Guimaraes FS, Moreira AC (1993) Effect of cannabidiol on plasma prolactin, growth hormone and cortisol in human volunteers. Braz J Med Biol Res 26:213–217
Zuardi AW, Morais SL, Guimaraes FS, Mechoulam R (1995) Antipsychotic effect of cannabidiol. J Clin Psychiatry 56:485–486
Zuardi AW, Hallak JE, Dursun SM, Morais SL, Sanches RF, Musty RE, Crippa JA (2006) Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol 20:683–686
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Support granted by the Stanely Medical Research Institute.
Mohini Ranganathan has in the past 3 years or currently received research grant support administered through Yale University School of Medicine from Insys Therapeutics and Pfizer Inc. Deepak Cyril D’Souza has in the past 3 years or currently received research grant support administered through Yale University School of Medicine from Pfizer Inc. Pfizer, Inc. had a role in the design and conduct of the study: collection, management, analysis, and interpretation of the data; preparation, review, and approval of the manuscript; and the decision to submit the manuscript for publication. Andrew Davies is a founder and full time employee of STI Pharmaceuticals Ltd (UK). Douglas Boggs, Aarti Gupta, John Cahill, Brian Pittman, Ashley Schnakenberg Martin, Halle Thurnauer, Swapnil Gupta, and Toral Surti report no financial relationships with commercial interests.
Electronic supplementary material
Supplemental Table 1
(DOCX 68 kb)
Rights and permissions
About this article
Cite this article
Boggs, D.L., Surti, T., Gupta, A. et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacology 235, 1923–1932 (2018). https://doi.org/10.1007/s00213-018-4885-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4885-9