Abstract
Summary
We investigated the interaction between periostin SNPs and the SNPs of the genes assumed to modulate serum periostin levels and bone microstructure in a cohort of postmenopausal women. We identified an interaction between LRP5 SNP rs648438 and periostin SNP rs9547970 on serum periostin levels and on radial cortical porosity.
Purpose
The purpose of this study is to investigate the interaction between periostin gene polymorphisms (SNPs) and other genes potentially responsible for modulating serum periostin levels and bone microstructure in a cohort of postmenopausal women.
Methods
In 648 postmenopausal women from the Geneva Retirees Cohort, we analyzed 6 periostin SNPs and another 149 SNPs in 14 genes, namely BMP2, CTNNB1, ESR1, ESR2, LRP5, LRP6, PTH, SPTBN1, SOST, TGFb1, TNFRSF11A, TNFSF11, TNFRSF11B and WNT16. Volumetric BMD and bone microstructure were measured by high-resolution peripheral quantitative computed tomography at the distal radius and tibia.
Results
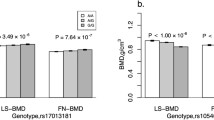
Serum periostin levels were associated with radial cortical porosity, including after adjustment for age, BMI, and years since menopause (p = 0.036). Sixteen SNPs in the ESR1, LRP5, TNFRSF11A, SOST, SPTBN1, TNFRSF11B and TNFSF11 genes were associated with serum periostin levels (p range 0.03–0.001) whereas 26 SNPs in 9 genes were associated with cortical porosity at the radius and/or at the tibia. WNT 16 was the gene with the highest number of SNPs associated with both trabecular and cortical microstructure. The periostin SNP rs9547970 was also associated with cortical porosity (p = 0.04). In particular, SNPs in LRP5, ESR1 and near the TNFRSF11A gene were associated with both cortical porosity and serum periostin levels. Eventually, we identified an interaction between LRP5 SNP rs648438 and periostin SNP rs9547970 on serum periostin levels (interaction p = 0.01) and on radial cortical porosity (interaction p = 0.005).
Conclusion
These results suggest that periostin expression is genetically modulated, particularly by polymorphisms in the Wnt pathway, and is thereby implicated in the genetic variation of bone microstructure.


Similar content being viewed by others
References
Boudin E, Fijalkowski I, Hendrickx G, Van HW (2016) Genetic control of bone mass. Mol Cell Endocrinol 432:3–13
Nagy H, Sornay-Rendu E, Boutroy S, Vilayphiou N, Szulc P, Chapurlat R (2013) Impaired trabecular and cortical microarchitecture in daughters of women with osteoporotic fracture: the MODAM study. Osteoporos Int 24:1881–1889
Bjørnerem Å, Bui M, Wang X, Ghasem-Zadeh A, Hopper JL, Zebaze R, Seeman E (2015) Genetic and environmental variances of bone microarchitecture and bone remodeling markers: a twin study. J Bone Miner Res 30:519–527
Boutroy S, Khosla S, Sornay-Rendu E, Zanchetta MB, McMahon DJ, Zhang CA, Chapurlat RD, Zanchetta J, Stein EM, Bogado C, Majumdar S, Burghardt AJ, Shane E (2016) Microarchitecture and peripheral BMD are impaired in postmenopausal Caucasian women with fracture independently of total hip T-score—an international multicenter study. J Bone Miner Res 31(6):1158–1166
Ralston SH, Crombrugghe B (2006) Genetic regulation of bone mass and susceptibility to osteoporosis. Genes Dev 20:2492–2506
Sims AM, Shephard N, Carter K, Doan T, Dowling A, Duncan EL, Eisman J, Jones G, Nicholson G, Prince R, Seeman E, Thomas G, Wass JA, Brown MA (2008) Genetic analyses in a sample of individuals with high or low BMD shows association with multiple Wnt pathway genes. J Bone Miner Res 23(4):499–506
Mencej-Bedrac S, Prezelj J, Kocjan T, Komadina R, Marc J (2009) Analysis of association of LRP5, LRP6, SOST, DKK1, and CTNNB1 genes with bone mineral density in a Slovenian population. Calcif Tissue Int 85(6):501–506
Koller DL, Zheng HF, Karasik D et al (2013) Meta-analysis of genome-wide studies identifiesWNT16 and ESR1 SNPs associated with bone mineral density in premenopausal women. J Bone Miner Res 28:547–558
Tu P, Duan P, Zhang RS, DB X, Wang Y, HP W, Liu YH, Si L (2015) Polymorphisms in genes in the RANKL/RANK/OPG pathway are associated with bone mineral density at different skeletal sites in post-menopausal women. Osteoporos Int 26(1):179–185
Tural S, Alayli G, Kara N, Tander B, Bilgici A, Kuru O (2013) Association between osteoporosis and polymorphisms of the IL-10 and TGF-beta genes in Turkish postmenopausal women. Hum Immunol 74(9):1179–1183
Bonnet N, Biver E, Durosier C, Chevalley T, Rizzoli R, Ferrari S (2015) Additive genetic effects on circulating periostin contribute to the heritability of bone microstructure. J Clin Endocrinol Metab 100(7):E1014–E1021
Rousseau JC, Sornay-Rendu E, Bertholon C, Chapurlat R, Garnero P (2014) Serum periostin is associated with fracture risk in postmenopausal women: a 7-year prospective analysis of the OFELY study. J Clin Endocrinol Metab 99(7):2533–2539
Xiao SM, Gao Y, Cheung CL, Bow CH, Lau KS, Sham PC, Tan KC, Kung AW (2012) Association of CDX1 binding site of periostin gene with bone mineral density and vertebral fracture risk. Osteoporos Int 23(7):1877–1887
Bonnet N, Garnero P, Ferrari S (2016) Periostin action in bone. Mol Cell Endocrinol 432:75–82
Bonnet N, Conway SJ, Ferrari SL (2012) Regulation of beta catenin signaling and parathyroid hormone anabolic effects in bone by the matricellular protein periostin. Proc Natl Acad Sci U S A 109(37):15048–15053
Hasegawa D, Wada N, Maeda H, Yoshida S, Mitarai H, Tomokiyo A, Monnouchi S, Hamano S, Yuda A, Akamine A (2015) Wnt5a induces collagen production by human periodontal ligament cells through TGFβ1-mediated upregulation of periostin expression. J Cell Physiol 230(11):2647–2660
Biver E, Durosier C, Chevalley T, Herrmann FR, Ferrari S, Rizzoli R (2015) Prior ankle fractures in postmenopausal women are associated with low areal bone mineral density and bone microstructure alterations. Osteoporos Int 26(8):2147–2155
Durosier C, van Lierop A, Ferrari S, Chevalley T, Papapoulos S, Rizzoli R (2013) Association of circulating sclerostin with bone mineral mass, microstructure, and turnover biochemical markers in healthy elderly men and women. J Clin Endocrinol Metab 98(9):3873–3883
Durosier-Izart C, Biver E, Merminod F, van Rietbergen B, Chevalley T, Herrmann FR, Ferrari SL, Rizzoli R (2017) Peripheral skeleton bone strength is positively correlated with total and dairy protein intakes in healthy postmenopausal women. Am J Clin Nutr 105(2):513–525
Nishiyama KK, Macdonald HM, Buie HR, Hanley DA, Boyd SK (2010) Postmenopausal women with osteopenia have higher cortical porosity and thinner cortices at the distal radius and tibia than women with normal aBMD: an in vivo HR-pQCT study. J Bone Miner Res 25:882–890
Cleves MA (2005) Exploratory analysis of single nucleotide polymorphism (SNP) for quantitative traits. Stata J 2:141–153
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Stat Methodol 57:289–300
VanderWeele TJ, Knol MJ (2014) A tutorial on interaction. Epidemiol Methods 3:3–72
Movérare-Skrtic S, Henning P, Liu X et al (2014) Osteoblast-derived WNT16 represses osteoclastogenesis and prevents cortical bone fragility fractures. Nat Med 20:1279–1288
Zheng HF, Tobias JH, Duncan E et al (2012) WNT16 influences bone mineral density, cortical bone thickness, bone strength, and osteoporotic fracture risk. PLoS Genet 8:e1002745
Zupan J, Mencej-Bedrac S, Jurković-Mlakar S, Prezelj J, Marc J (2010) Gene-gene interactions in RANK/RANKL/OPG system influence bone mineral density in postmenopausal women. J Steroid Biochem Mol Biol 118(1–2):102–106
Luo L, Xia W, Nie M, Sun Y, Jiang Y, Zhao J, He S, Xu L (2014) Association of ESR1 and C6orf97 gene polymorphism with osteoporosis in postmenopausal women. Mol Biol Rep 41(5):3235–3243
VanMeurs JB, Trikalinos TA, Ralston SH et al (2008) Large-scale analysis of association between LRP5 and LRP6 variants and osteoporosis. JAMA 299(11):1277–1290
Paternoster L, Lorentzon M, Lehtimäki T, Eriksson J, Kähönen M, Raitakari O, Laaksonen M, Sievänen H, Viikari J, Lyytikäinen LP, Mellström D, Karlsson M, Ljunggren O, Grundberg E, Kemp JP, Sayers A, Nethander M, Evans DM, Vandenput L, Tobias JH, Ohlsson C (2013) Genetic determinants of trabecular and cortical volumetric bone mineral densities and bone microstructure. PLoS Genet 9(2):e1003247
Mamalis A, Markopoulou C, Lagou A, Vrotsos I (2011) Oestrogen regulates proliferation, osteoblastic differentiation, collagen synthesis and periostin gene expression in human periodontal ligament cells through oestrogen receptor beta. Arch Oral Biol 56(5):446–455
Yi J, Cai Y, Yao Z, Lin J (2013) Genetic analysis of the relationship between bone mineral density and low-density lipoprotein receptor-related protein 5 gene polymorphisms. PLoS One 8(12):e85052
Ferrari SL, Deutsch S, Choudhury U, Chevalley T, Bonjour JP, Dermitzakis ET, Rizzoli R, Antonarakis SE (2004) Polymorphisms in the low-density lipoprotein receptor-related protein 5 (LRP5) gene are associated with variation in vertebral bone mass, vertebral bone size, and stature in whites. Am J Hum Genet 74(5):866–875
Kobayashi Y, Uehara S, Udagawa N, Takahashi N (2016) Regulation of bone metabolism by Wnt signals. J Biochem 159(4):387–392
Lauretani F, Cepollaro C, Bandinelli S, Cherubini A, Gozzini A, Masi L, Falchetti A, Del Monte F, Carbonell-Sala S, Marini F, Tanini A, Corsi AM, Ceda GP, Brandi ML, Ferrucci L (2010) LRP5 gene polymorphism and cortical bone. Aging Clin Exp Res 22(4):281–288
Bonnet N, Biver E, Chevalley T, Rizzoli R, Garnero P, Ferrari SL. (2017) Serum levels of a cathepsin-K generated periostin fragment predict incident low-trauma fractures in postmenopausal women independently of BMD and FRAX. J Bone Miner Res [Epub ahead of print]
Acknowledgements
This study was supported by the Swiss National Foundation grant to SLF, the Geneva University Hospitals and Faculty of Medicine Clinical Research Center and the Foundation for Research on Aging AETAS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Pepe, J., Bonnet, N., Herrmann, F.R. et al. Interaction between LRP5 and periostin gene polymorphisms on serum periostin levels and cortical bone microstructure. Osteoporos Int 29, 339–346 (2018). https://doi.org/10.1007/s00198-017-4272-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-017-4272-0