Abstract
Summary
There is marked bone loss after spinal cord injury (SCI); however, its pathogenesis and clinical management remain unclear. The increased circulating levels of receptor activator of nuclear factor kB ligand (RANKL) associated with bone loss shortly after SCI and the prevention of bone loss with denosumab treatment suggest a contributory role of RANKL in SCI-induced osteoporosis.
Introduction
Bone turnover and bone loss are markedly increased shortly after SCI. However, the pathogenesis and clinical management of this process remain unclear, especially the role of the osteoprotegerin (OPG)/RANKL system in this disorder. The aim of this study was to analyze serum levels of OPG and RANKL in bone loss associated with recent SCI and the effect of denosumab treatment on these mediators.
Methods
Twenty-three males with recent complete SCI were prospectively included. Serum OPG and RANKL levels, bone turnover markers (PINP, bone ALP, CTX), and bone mineral density (BMD) were assessed at baseline, at 6 months of follow-up, prior to initiating denosumab, and 6 months after treatment. The results were compared with a healthy control group.
Results
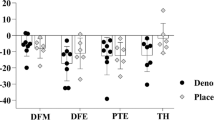
At baseline, SCI patients showed higher RANKL levels vs. controls which were correlated with days-since-SCI and total hip BMD loss at 6 months. OPG levels were similar to controls at baseline. After denosumab treatment, OPG showed no changes, whereas RANKL levels became undetectable in 67% of patients. Patients with undetectable RANKL during treatment showed better response in femoral BMD and bone markers vs. patients with detectable RANKL at 6 months of denosumab. Increased parathormone (PTH) levels during treatment were negatively correlated with RANKL changes.
Conclusions
RANKL levels are increased after SCI and related to BMD loss at the proximal femur, becoming undetectable after denosumab treatment. The effect of denosumab on preventing sublesional bone loss, especially in patients with undetectable levels during treatment, suggests a contributory role of RANKL in this process.




Similar content being viewed by others
Abbreviations
- SCI:
-
Spinal cord injury
- RANKL:
-
Receptor activator of nuclear factor kB ligand
- OPG:
-
Osteoprotegerin
- BMD:
-
Bone mineral density
- 25OHD:
-
25 hydroxyvitamin D
- BMI:
-
Body mass index
- AIS:
-
ASIA Impairment Scale
- PTH:
-
Parathormone
- Bone ALP:
-
Bone alkaline phosphatase
- PINP:
-
Propeptide amino-terminal of type I procollagen
- CTX:
-
Serum carboxy-terminal telopeptide of type I collagen
- SD:
-
Standard deviation
References
Jiang SD, Dai LY, Jiang LS (2006) Osteoporosis after spinal cord injury. Osteoporos Int 17(2):180–192
Giangregorio L, McCartney N (2006) Bone loss and muscle atrophy in spinal cord injury: epidemiology, fracture prediction, and rehabilitation strategies. J Spinal Cord Med 29(5):489–500
Gifre L, Vidal J, Carrasco JL, Filella X, Ruiz-Gaspà S, Muxi A, Portell E, Monegal A, Guañabens N, Peris P (2015) Effect of recent spinal cord injury on wnt signaling antagonists (sclerostin and dkk-1) and their relationship with bone loss. A 12-month prospective study. J Bone Miner Res 30(6):1014–1021
Spatz JM, Wein MN, Gooi JH, Qu Y, Garr JL, Liu S, Barry KJ, Uda Y, Lai F, Dedic C, Balcells-Camps M, Kronenberg HM, Babij P, Pajevic PD (2015) The Wnt inhibitor sclerostin is up-regulated by mechanical unloading in osteocytes in vitro. J Biol Chem 290(27):16744–16758
Qin W, Sun L, Cao J, Peng Y, Collier L, Wu Y, Creasey G, Li J, Qin Y, Jarvis J, Bauman WA, Zaidi M, Cardozo C (2013) The central nervous system (CNS)-independent anti-bone-resorptive activity of muscle contraction and the underlying molecular and cellular signatures. J Biol Chem 288(19):13511–13521
Moriishi T, Fukuyama R, Ito M, Miyazaki T, Maeno T, Kawai Y, Komori H, Komori T (2012) Osteocyte network; a negative regulatory system for bone mass augmented by the induction of Rankl in osteoblasts and Sost in osteocytes at unloading. PLoS One 7(6):e40143
Jiang SD, Jiang LS, Dai LY (2007) Effects of spinal cord injury on osteoblastogenesis, osteoclastogenesis and gene expression profiling in osteoblasts in young rats. Osteoporos Int 18(3):339–349
Qin W, Li X, Peng Y, Harlow LM, Ren Y, Wu Y, Li J, Qin Y, Sun J, Zheng S, Brown T, Feng JQ, Ke HZ, Bauman WA, Cardozo CC (2015) Sclerostin antibody preserves the morphology and structure of osteocytes and blocks the severe skeletal deterioration after motor-complete spinal cord injury in rats. J Bone Miner Res 30(11):1994–2004
Maïmoun L, Couret I, Mariano-Goulart D, Dupuy AM, Micallef JP, Peruchon E, Ohanna F, Cristol JP, Rossi M, Leroux JL (2005) Changes in osteoprotegerin/RANKL system, bone mineral density, and bone biochemicals markers in patients with recent spinal cord injury. Calcif Tissue Int 76(6):404–411
Morse LR, Sudhakar S, Lazzari AA, Tun C, Garshick E, Zafonte R, Battaglino RA (2013) Sclerostin: a candidate biomarker of SCI-induced osteoporosis. Osteoporos Int 24(3):961–968
Morse LR, Nguyen HP, Jain N, Williams S, Tun CG, Battaglino RA, Stashenko P, Garshick E (2008) Age and motor score predict osteoprotegerin level in chronic spinal cord injury. J Musculoskelet Neuronal Interact 8(1):50–57
Miller PD, Pannacciulli N, Brown JP, Czerwinski E, Nedergaard BS, Bolognese MA, Malouf J, Bone HG, Reginster JY, Singer A, Wang C, Wagman RB, Cummings SR (2016) Denosumab or zoledronic acid in postmenopausal women with osteoporosis previously treated with oral bisphosphonates. J Clin Endocrinol Metab 101(8):3163–3170
Papapoulos S, Lippuner K, Roux C, Lin CJ, Kendler DL, Lewiecki EM, Brandi ML, Czerwiński E, Franek E, Lakatos P, Mautalen C, Minisola S, Reginster JY, Jensen S, Daizadeh NS, Wang A, Gavin M, Libanati C, Wagman RB, Bone HG (2015) The effect of 8 or 5 years of denosumab treatment in postmenopausal women with osteoporosis: results from the FREEDOM Extension study. Osteoporos Int 26(12):2773–2783
Gifre L, Vidal J, Carrasco JL, Muxi A, Portell E, Monegal A, Guañabens N, Peris P (2016) Denosumab increases sublesional bone mass in osteoporotic individuals with recent spinal cord injury. Osteoporos Int 27(1):405–410
Kirshblum SC, Burns SP, Biering-Sorensen F, Donovan W, Graves DE, Jha A, Johansen M, Jones L, Krassioukov A, Mulcahey MJ, Schmidt-Read M, Waring W (2011) International standards for neurological classification of spinal cord injury (revised 2011). J Spinal Cord Med 34(6):535–546
(1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO study group. World Health Organ Tech Rep Ser 843:1–129
Tobin J (1958) Estimation of relationships for limited dependent variables. Econometrica 26:24–36
Nie L, Chub H, Liu C, Cole SR, Vexler A, Schisterman EF (2010) Linear regression with an independent variable subject to a detection limit. Epidemiology 21:S17–S24
Rucci N, Rufo A, Alamanou M, Teti A (2007) Modeled microgravity stimulates osteoclastogenesis and bone resorption by increasing osteoblast RANKL/OPG ratio. J Cell Biochem 100(2):464–473
Sun L, Pan J, Peng Y, Wu Y, Li J, Liu X, Qin Y, Bauman WA, Cardozo C, Zaidi M, Qin W (2013) Anabolic steroids reduce spinal cord injury-related bone loss in rats associated with increased Wnt signaling. J Spinal Cord Med 36(6):616–622
Morse L, Teng YD, Pham L, Newton K, Yu D, Liao WL, Kohler T, Müller R, Graves D, Stashenko P, Battaglino R (2008) Spinal cord injury causes rapid osteoclastic resorption and growth plate abnormalities in growing rats (SCI-induced bone loss in growing rats). Osteoporos Int 19(5):645–652
Alvarez L, Peris P, Guañabens N, Vidal S, Ros I, Pons F, Filella X, Monegal A, Muñoz-Gomez J, Ballesta AM (2003) Serum osteoprotegerin and its ligand in Paget’s disease of bone. Arthritis Rheum 48(3):824–828
Anastasilakis AD, Polyzos SA, Gkiomisi A, Bisbinas I, Gerou S, Makras P (2013) Comparative effect of zoledronic acid versus denosumab on serum sclerostin and dickkopf-1 levels of naive postmenopausal women with low bone mass: a randomized, head-to-head clinical trial. J Clin Endocrinol Metab 98(8):3206–3212
Anastasilakis AD, Polyzos SA, Efstathiadou ZA, Savvidis M, Sakellariou GT, Papatheodorou A, Kokkoris P, Makras P (2015) Denosumab in treatment-naïve and pre-treated with zoledronic acid postmenopausal women with low bone mass: effect on bone mineral density and bone turnover markers. Metabolism 64(10):1291–1297
Anastasilakis AD, Polyzos SA, Gkiomisi A, Saridakis ZG, Digkas D, Bisbinas I, Sakellariou GT, Papatheodorou A, Kokkoris P, Makras P (2015) Denosumab versus zoledronic acid in patients previously treated with zoledronic acid. Osteoporos Int 26(10):2521–2527
Makras P, Polyzos SA, Papatheodorou A, Kokkoris P, Chatzifotiadis D, Anastasilakis AD (2013) Parathyroid hormone changes following denosumab treatment in postmenopausal osteoporosis. Clin Endocrinol 79(4):499–503
Ma YL, Cain RL, Halladay DL, Yang X, Zeng Q, Miles RR, Chandrasekhar S, Martin TJ, Onyia JE (2001) Catabolic effects of continuous human PTH (1-38) in vivo is associated with sustained stimulation of RANKL and inhibition of osteoprotegerin and gene-associated bone formation. Endocrinology 142(9):4047–4054
Cirnigliaro CM, Myslinski MJ, La Fountaine MF, Kirshblum SC, Forrest GF, Bauman WA (2017) Bone loss at the distal femur and proximal tibia in persons with spinal cord injury: imaging approaches, risk of fracture, and potential treatment options. Osteoporos Int 28(3):747–765
Acknowledgments
This work was funded by grants from Fundación Española de Investigación Ósea y Metabolismo Mineral (FEIOMM) and Fundació La Marató de TV3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Gifre, L., Ruiz-Gaspà, S., Carrasco, J.L. et al. Effect of recent spinal cord injury on the OPG/RANKL system and its relationship with bone loss and the response to denosumab therapy. Osteoporos Int 28, 2707–2715 (2017). https://doi.org/10.1007/s00198-017-4090-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-017-4090-4