Abstract
Objective
Invasive candidiasis (IC) outcomes in intensive care units (ICUs) could be improved by the early administration of antifungals. The Candida Score (CS) prediction rule has been proposed for the selection of patients who could develop IC. Procalcitonin (PCT) levels allow prompt identification of sepsis, but their behavior in the setting of IC is unclear. We hypothesize that PCT could be helpful in the early diagnosis of IC in patients with Candida sp. colonization.
Design
Prospective observational study.
Setting
Thirty-six ICUs in Spain, Portugal and France.
Patients
Every non-neutropenic critically ill patient hospitalized for more than 7 days without concurrent bacterial infection. The CS was calculated weekly. Serums were collected concomitantly.
Measurements and results
Two hundred twenty PCT levels were measured in 136 patients [neither colonized nor infected (NCNI): n = 73; multifocal colonization (MF): n = 43; MF + IC: n = 20]. Baseline PCT levels were significantly higher in the MF + IC group than in other groups (p = 0.001). In patients with MF, the highest CS value calculated during the patient’s stay was the sole independent predictor of IC. The receiver-operating curve analysis showed that the diagnosis values of PCT and CS were comparable (AUROCC = 0.713, and 0.727, respectively). Moreover, PCT increased the positive predictive value of CS from 44.7 to 59.3%.
Conclusions
After 7 days of hospitalization, PCT levels in patients with MF who go on to develop IC are higher than in others. Serum PCT could also improve the predictive value of CS. PCT together with CS could therefore be considered for the assessment of IC risk.
Similar content being viewed by others
Introduction
Invasive candidiasis (IC) has become a leading cause of sepsis in critically ill patients [1, 2]. Screening for fungal colonization has been proposed as a method for early identification of patients with the highest risk of developing IC [3–5]. Thus, although common in this setting, multifocal colonization is often managed by implementing antifungal therapy whose expected benefits are the prevention of such infections as well as the improvement of their outcome [6–10]. Additional markers are therefore needed. Some clinical scores have been proposed for this purpose [11, 12]. Among them, the Candida Score (CS) could be more reliable than colonization alone.
Procalcitonin (PCT) elevation is strongly associated with systemic bacterial infections [13]. It is unknown to what extent measuring PCT levels could contribute to the early diagnosis of IC [14].
We therefore address this issue in critically ill patients included in a large prospective, observational, multicenter cohort study designed to validate the CS [15].
Materials and methods
Every patient over the age of 18 years old who was admitted for at least 7 days to 36 medical–surgical ICUs were eligible. The study was approved by the Institutional Review Board of the participating centers.
The exclusion criteria used were as follows: (1) age under 18 years; (2) neutropenia, defined as a neutrophil count ≤500/mm3 for more than 3 weeks; (3) life expectancy of less than a week; (4) pregnant women and nursing mothers; (5) fungal infections other than those caused by Candida spp; (6) patients that had Candida spp isolation from a sterile site or were treated with antifungal drugs during the first 7 days of ICU; (7) refusal to give informed consent.
In all patients, surveillance samples for the detection of fungal growth were cultured. The diagnosis of bacterial infection was then considered at the discretion of the attending physician and recorded accordingly.
Definitions have been published elsewhere [15]. Briefly, patients were considered as colonized (i.e., multifocal colonization [MF]) if cultures from at least two non-contiguous body sites grew with Candida species. Invasive candidiasis was considered in colonized patients: (1) if at least one blood culture was positive for Candida species in a patient with consistent clinical symptoms; (2) if the role of Candida was established in patients with peritonitis; (3) if chorioretinitis was diagnosed through fundoscopy in the presence of consistent clinical symptoms. Otherwise, the included patients were considered as being neither colonized nor infected (NCNI).
The rounded CSs for a cutoff value of 3 were as follows: TPN × 1; plus surgery × 1; plus MF × 1, and plus severe sepsis × 2 [11]. For each patient the CS value kept for analysis was its maximum value at or before an IC episode if appropriate.
Values are expressed as mean ± SD unless otherwise stated. Continuous variables were compared with the Mann–Whitney U test or the Kruskall–Wallis test, depending on the number of categories. Categorical variables were compared using the χ 2 test.
A p value <0.05 was considered as statistically significant for all analyses. STATA software was used for all analyses (STATA Statistical Package, College Station, TX).
Results
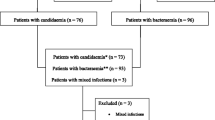
A total of 1,107 patients were included in the CAVA study. Serum samples were obtained from 240 of them, and 136 of these patients without bacterial infection throughout their ICU stay were selected for PCT analysis: 73 were neither colonized nor infected (NCNI), 43 were colonized (MF), and 20 were colonized and developed invasive candidiasis (MF + IC) (Table 1). Patients from the IC group were more likely to be surgical than those from the MF and the NCNI group.
At the time of inclusion, patients from the IC group were more likely to present with septic shock than their non-infected counterparts. In addition, SOFA scores were higher in the IC group.
The highest CSs were found in patients with multifocal fungal colonization (Table 2). Similarly, colonized patients who developed IC had higher CS than those who did not.
PCT levels at the time of inclusion were significantly higher in colonized patients who went on to develop IC than in those who did not (Fig. 1). In addition, PCT was higher in the latter patients than in those with neither infection nor colonization. Moreover, PCT levels were found to be within the “normal” range of values in most of the cases. Indeed, PCT obtained at the onset of infection remained lower than 1.5 ng/ml in 17 out of the 20 patients with IC.
Then, we searched for potent confounding variables that could account for the abovementioned differences in PCT elevation on day 7. Surgery on admission, a high SOFA score, the presence of septic shock and a high CS were also found to be associated with the risk of IC by univariate analysis (Tables 1, 2). All of these relevant variables were therefore entered into a multivariate analysis model. As a result, only CS remained independently associated with the occurrence of IC (Table 3).
We aimed thereafter to assess the predictive value of both PCT-D7 and CS regarding the risk of IC by constructing the corresponding ROC curves (Fig. 2 in ESM). These two variables were found to be comparable (Table 4). The optimal cutoff values of PCT-D7 were also extracted from the curves. The use of a PCT-D7 ≥0.3 ng/ml and a CS ≥3 points yielded 80.0% sensitivity and 74.4% specificity. As a result, the positive predictive value of the CS was significantly improved from 44.7 to 59.3% while the negative predictive value remained high (88.9%).
Discussion
We show herein that the weekly calculation of the CS in combination with serum PCT levels in critically ill patients might be helpful in differentiating between those who go on to develop IC and those who do not.
The present multicenter study has shown that a CS of less than 3 points could accurately and safely identify patients who will not benefit from early antifungal therapy [15].
In selected patients included in the present study (i.e., multifocal Candida spp colonization without concurrent bacterial infection), we demonstrate that PCT on day 7 following ICU admission was significantly higher in patients who go on to develop IC than in those who do not. As a result, PCT could improve the diagnosis value of the CS with a cutoff of 0.3 ng/ml by allowing a slight increase of its positive predictive value.
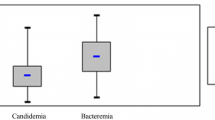
The behavior of serum PCT is unclear in the setting of IC. We have previously reported that PCT elevation was lower in critically ill patients with candidemia compared with those with bacteremia, regardless of the disease severity, suggesting that the host inflammatory response assessed by PCT measurement could be different according to the causative microorganism [16, 17]. The present results are in accordance with such findings since PCT elevation at the onset of IC remains within a low range of values in most of the cases, although septic shock was a frequent condition in these patients.
In addition, our findings suggest that fungal colonization per se could trigger an inflammatory response, the magnitude of which would be related to the risk of developing IC. The degree of fungal invasion could be assessed therefore through PCT measurements in colonized patients.
Accordingly, the interest of combining a clinical score with a biomarker has already been suggested in the setting of ventilator-associated pneumonia, another common but difficult to diagnose nosocomial infection [18].
Several limitations have to be mentioned. First of all, the small size of our sample might have hidden some differences between groups because of a lack of statistical power. Accordingly, we could not exclude the possibility that the greater severity of the disease in patients from the IC group accounted for the higher PCT levels. PCT should therefore be used as a surrogate for the assessment of disease severity. Second, our results were obtained in selected patients, namely those without bacterial sepsis, in order to avoid other causes of a PCT increase. Hence, our subset of patients is peculiar and somewhat different from the whole cohort population [15]. Accordingly, the pre-test probability of IC is higher, and the diagnosis value of the combination of CS and PCT might have been overestimated. Our findings should therefore be very cautiously translated into clinical practice given the high prevalence of bacterial sepsis in the ICU setting. Third, we cannot exclude the possibility that patients with actual bacterial sepsis, although not proven, were improperly selected. It has been shown that no microbial agent was isolated in around 70% of the patients with septic shock [19]. As a result, PCT values obtained on day 7 in patients who developed IC could have been overestimated.
Measurement of serum PCT in addition to calculation of the CS in critically ill patients exhibiting multifocal Candida spp colonization without bacterial infection might be useful for the risk assessment of further IC. Early antifungal therapy might be considered in patients with a CS ≥3 points combined with a PCT ≥0.3 ng/ml. Further studies are needed to assess prospectively the relevance of such findings.
References
Marchetti O, Bille J, Fluckiger U, Eggimann P, Ruef C, Garbino J, Calandra T, Glauser MP, Tauber MG, Pittet D (2004) Epidemiology of candidemia in Swiss tertiary care hospitals: secular trends, 1991–2000. Clin Infect Dis 38:311–320
Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB (2004) Nosocomial bloodstream infections in us hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 39:309–317
Eggimann P, Garbino J, Pittet D (2003) Epidemiology of candida species infections in critically ill non-immunosuppressed patients. Lancet Infect Dis 3:685–702
Pittet D, Monod M, Suter PM, Frenk E, Auckenthaler R (1994) Candida colonization and subsequent infections in critically ill surgical patients. Ann Surg 220:751–758
Gauzit R, Cohen Y, Dupont H, Hennequin C, Montravers P, Timsit JF, Veber B, Chevalier E, Blin P, Palestro B (2003) Infections by candida sp. In intensive care. Survey of French practices. Presse Med 32:440–449
Piarroux R, Grenouillet F, Balvay P, Tran V, Blasco G, Millon L, Boillot A (2004) Assessment of preemptive treatment to prevent severe candidiasis in critically ill surgical patients. Crit Care Med 32:2443–2449
Rangel-Frausto MS, Wiblin T, Blumberg HM, Saiman L, Patterson J, Rinaldi M, Pfaller M, Edwards JE Jr, Jarvis W, Dawson J, Wenzel RP (1999) National epidemiology of mycoses survey (NEMIS): variations in rates of bloodstream infections due to candida species in seven surgical intensive care units and six neonatal intensive care units. Clin Infect Dis 29:253–258
Charles PE, Dalle F, Aube H, Doise JM, Quenot JP, Aho LS, Chavanet P, Blettery B (2005) Candida spp. colonization significance in critically ill medical patients: a prospective study. Intensive Care Med 31:393–400
Charles PE (2006) Multifocal candida species colonization as a trigger for early antifungal therapy in critically ill patients: what about other risk factors for fungal infection? Crit Care Med 34:913–914
Nolla-Salas J, Sitges-Serra A, Leon-Gil C, Martinez-Gonzalez J, Leon-Regidor MA, Ibanez-Lucia P, Torres-Rodriguez JM (1997) Candidemia in non-neutropenic critically ill patients: analysis of prognostic factors and assessment of systemic antifungal therapy. Study group of fungal infection in the ICU. Intensive Care Med 23:23–30
Leon C, Ruiz-Santana S, Saavedra P, Almirante B, Nolla-Salas J, Alvarez-Lerma F, Garnacho-Montero J, Leon MA (2006) A bedside scoring system (“Candida score”) for early antifungal treatment in nonneutropenic critically ill patients with candida colonization. Crit Care Med 34:730–737
Ostrosky-Zeichner L, Sable C, Sobel J, Alexander BD, Donowitz G, Kan V, Kauffman CA, Kett D, Larsen RA, Morrison V, Nucci M, Pappas PG, Bradley ME, Major S, Zimmer L, Wallace D, Dismukes WE, Rex JH (2007) Multicenter retrospective development and validation of a clinical prediction rule for nosocomial invasive candidiasis in the intensive care setting. Eur J Clin Microbiol Infect Dis 26:271–276
Christ-Crain M, Muller B (2005) Procalcitonin in bacterial infections—hype, hope, more or less? Swiss Med Wkly 135:451–460
Christofilopoulou S, Charvalos E, Petrikkos G (2002) Could procalcitonin be a predictive biological marker in systemic fungal infections? Study of 14 cases. Eur J Intern Med 13:493–495
Leon C, Ruiz-Santana S, Saavedra P, Galvan B, Blanco A, Castro C, Balasini C, Utande-Vazquez A, Gonzales de Molina FJ, Blaso-Naval-Proto MA, Lopez MJ, Charles PE, Martin E, Hernadez-Viera MA (2009) Usefulness of the “Candida score” for discriminating between Candida colonization and invasive candidiasis in non-neutropenic critically ill patients: a prospective multicenter study. Crit Care Med 37:1624–1633
Charles PE, Dalle F, Aho S, Quenot JP, Doise JM, Aube H, Olsson NO, Blettery B (2006) Serum procalcitonin measurement contribution to the early diagnosis of candidemia in critically ill patients. Intensive Care Med 32:1577–1583
Charles PE, Ladoire S, Aho LS, Quenot JP, Pechinot A, Doise JM, Prin S, Olsson NO, Blettery B (2008) Serum procalcitonin elevation in the critically ill patients at the onset of bacteremia caused by either gram negative or gram positive bacteria. BMC Infect Dis 8:38
Ramirez P, Garcia MA, Ferrer M, Aznar J, Valencia M, Sahuquillo JM, Menendez R, Asenjo MA, Torres A (2008) Sequential measurements of procalcitonin levels in diagnosing ventilator-associated pneumonia. Eur Respir J 31:356–362
Annane D, Aegerter P, Jars-Guincestre MC, Guidet B (2003) Current epidemiology of septic shock: the cub-rea network. Am J Respir Crit Care Med 168:165–172
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the CAVA study group.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Charles, P.E., Castro, C., Ruiz-Santana, S. et al. Serum procalcitonin levels in critically ill patients colonized with Candida spp: new clues for the early recognition of invasive candidiasis?. Intensive Care Med 35, 2146–2150 (2009). https://doi.org/10.1007/s00134-009-1623-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-009-1623-0