Abstract
Aims/hypothesis
More than 90% of Chinese familial early-onset type 2 diabetes mellitus is genetically unexplained. To investigate the molecular aetiology, we identified and characterised whether mutations in the KCNJ11 gene are responsible for these families.
Methods
KCNJ11 mutations were screened for 96 familial early-onset type 2 diabetic probands and their families. Functional significance of the identified mutations was confirmed by physiological analysis, molecular modelling and population survey.
Results
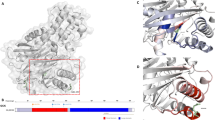
Three novel KCNJ11 mutations, R27H, R192H and S116F117del, were identified in three families with early-onset type 2 diabetes mellitus. Mutated KCNJ11 with R27H or R192H markedly reduced ATP sensitivity (E23K>R27H>C42R>R192H>R201H), but no ATP-sensitive potassium channel currents were detected in the loss-of-function S116F117del channel in vitro. Molecular modelling indicated that R192H had a larger effect on the channel ATP-binding pocket than R27H, which may qualitatively explain why the ATP sensitivity of the R192H mutation is seven times less than R27H. The shape of the S116F117del channel may be compressed, which may explain why the mutated channel had no currents. Discontinuation of insulin and implementation of sulfonylureas for R27H or R192H carriers and continuation/switch to insulin therapy for S116F117del carriers resulted in good glycaemic control.
Conclusions/interpretation
Our results suggest that genetic diagnosis for the KCNJ11 mutations in familial early-onset type 2 diabetes mellitus may help in understanding the molecular aetiology and in providing more personalised treatment for these specific forms of diabetes in Chinese and other Asian patients.




Similar content being viewed by others
Abbreviations
- HI:
-
Hyperinsulinism
- IA-2:
-
Tyrosine phosphatase-like protein
- IGT:
-
Impaired glucose tolerance
- KATP :
-
ATP-sensitive potassium channel
- MODY:
-
Maturity-onset diabetes of the young
- PNDM:
-
Permanent neonatal diabetes
- SNP:
-
Single nucleotide polymorphism
- TM:
-
Transmembrane
References
Bonnefond A, Froguel P, Vaxillaire M (2010) The emerging genetics of type 2 diabetes. Trends Mol Med 16:407–416
American Diabetes Association (2013) Diagnosis and classification of diabetes mellitus. Diabetes Care 36(Suppl 1):S67–S74
Fajans SS, Bell GI (2011) MODY: history, genetics, pathophysiology, and clinical decision making. Diabetes Care 34:1878–1884
Doria A, Yang Y, Malecki M et al (1999) Phenotypic characteristics of early-onset autosomal-dominant type 2 diabetes unlinked to known maturity-onset diabetes of the young (MODY) genes. Diabetes Care 22:253–261
Gloyn AL, Pearson ER, Antcliff JF et al (2004) Activating mutations in the gene encoding the ATP-sensitive potassium-channel subunit Kir6.2 and permanent neonatal diabetes. N Engl J Med 350:1838–1849
Yorifuji T, Nagashima K, Kurokawa K et al (2005) The C42R mutation in the Kir6.2 (KCNJ11) gene as a cause of transient neonatal diabetes, childhood diabetes, or later-onset, apparently type 2 diabetes mellitus. J Clin Endocrinol Metab 90:3174–3178
Bonnefond A, Philippe J, Durand E et al (2012) Whole-exome sequencing and high throughput genotyping identified KCNJ11 as the thirteenth MODY gene. PLoS One 7:e37423
Inagaki N, Gonoi T, Clement JP 4th et al (1995) Reconstitution of IKATP: an inward rectifier subunit plus the sulfonylurea receptor. Science 270:1166–1170
Tucker SJ, Gribble FM, Proks P et al (1998) Molecular determinants of KATP channel inhibition by ATP. EMBO J 17:3290–3296
Girard CA, Wunderlich FT, Shimomura K et al (2009) Expression of an activating mutation in the gene encoding the KATP channel subunit Kir6.2 in mouse pancreatic beta cells recapitulates neonatal diabetes. J Clin Invest 119:80–90
Proks P, Reimann F, Green N, Gribble F, Ashcroft F (2002) Sulfonylurea stimulation of insulin secretion. Diabetes 51(Suppl 3):S368–S376
Gloyn AL, Weedon MN, Owen KR et al (2003) Large-scale association studies of variants in genes encoding the pancreatic beta-cell KATP channel subunits Kir6.2 (KCNJ11) and SUR1 (ABCC8) confirm that the KCNJ11 E23K variant is associated with type 2 diabetes. Diabetes 52:568–572
Seino S, Iwanaga T, Nagashima K, Miki T (2000) Diverse roles of KATP channels learned from Kir6. 2 genetically engineered mice. Diabetes 49:311–318
Miki T, Tashiro F, Iwanaga T et al (1997) Abnormalities of pancreatic islets by targeted expression of a dominant-negative KATP channel. Proc Natl Acad Sci U S A 94:11969–11973
Fukui M, Nakano K, Obayashi H et al (1997) High prevalence of mitochondrial diabetes mellitus in Japanese patients with major risk factors. Metabolism 46:793–795
American Diabetes Association (2010) Diagnosis and classification of diabetes mellitus. Diabetes Care 33(Suppl 1):S62–S69
Yamagata K, Oda N, Kaisaki PJ et al (1996) Mutations in the hepatocyte nuclear factor-1alpha gene in maturity-onset diabetes of the young (MODY3). Nature 384:455–458
Yamagata K, Furuta H, Oda N et al (1996) Mutations in the hepatocyte nuclear factor-4alpha gene in maturity-onset diabetes of the young (MODY1). Nature 384:458–460
Froguel P, Zouali H, Vionnet N et al (1993) Familial hyperglycemia due to mutations in glucokinase: definition of a subtype of diabetes mellitus. N Engl J Med 328:697–702
Chèvre JC, Hani EH, Stoffers DA, Habener JF, Froguel P (1998) Insulin promoter factor 1 gene is not a major cause of maturity-onset diabetes of the young in French Caucasians. Diabetes 47:843–844
Beards F, Frayling T, Bulman M et al (1998) Mutations in hepatocyte nuclear factor 1beta are not a common cause of maturity-onset diabetes of the young in the U.K. Diabetes 47:1152–1154
Malecki MT, Jhala US, Antonellis A et al (1999) Mutations in NEUROD1 are associated with the development of type 2 diabetes mellitus. Nat Genet 23:323–328
The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (2003) Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (position statement). Diabetes Care 26(Suppl 1):S5–S20
Pegan S, Arrabit C, Zhou W et al (2005) Cytoplasmic domain structures of Kir2.1 and Kir3.1 show sites for modulating gating and rectification. Nat Neurosci 8:279–287
Antcliff JF, Haider S, Proks P, Sansom MS, Ashcroft FM (2005) Functional analysis of a structural model of the ATP-binding site of the KATP channel Kir6.2 subunit. EMBO J 24:229–239
Nagano N, Urakami T, Mine Y et al (2012) Diabetes caused by Kir6.2 mutation: successful treatment with oral glibenclamide switched from continuous subcutaneous insulin infusion in the early phase of the disease. Pediatr Int 54:277–279
Pearson ER, Flechtner I, Njølstad PR et al (2006) Switching from insulin to oral sulfonylureas in patients with diabetes due to Kir6.2 mutations. N Engl J Med 355:467–477
Bonellie S, Chalmers J, Gray R, Greer I, Jarvis S, Williams C (2008) Centile charts for birthweight for gestational age for Scottish singleton births. BMC Pregnancy Childbirth 8:5
Ng MC, Lee SC, Ko GT et al (2001) Familial early-onset type 2 diabetes in Chinese patients: obesity and genetics have more significant roles than autoimmunity. Diabetes Care 24:663–671
Xu JY, Dan QH, Chan V et al (2005) Genetic and clinical characteristics of maturity-onset diabetes of the young in Chinese patients. Eur J Hum Genet 13:422–427
Liu L, Furuta H, Minami A et al (2007) A novel mutation, Ser159Pro in the NeuroD1/BETA2 gene contributes to the development of diabetes in a Chinese potential MODY family. Mol Cell Biochem 303:115–120
Villareal DT, Koster JC, Robertson H et al (2009) Kir6.2 variant E23K increases ATP-sensitive K+ channel activity and is associated with impaired insulin release and enhanced insulin sensitivity in adults with normal glucose tolerance. Diabetes 58:1869–1878
Schwanstecher C, Meyer U, Schwanstecher M (2002) Kir6.2 polymorphism predisposes to type 2 diabetes by inducing overactivity of pancreatic beta-cell ATP-sensitive K+ channels. Diabetes 51:875–879
Klupa T, Edghill EL, Nazim J et al (2005) The identification of a R201H mutation in KCNJ11, which encodes Kir6.2, and successful transfer to sustained-release sulphonylurea therapy in a subject with neonatal diabetes: evidence for heterogeneity of beta cell function among carriers of the R201H mutation. Diabetologia 8:1029–1031
Girard CA, Shimomura K, Proks P et al (2006) Functional analysis of six Kir6.2 (KCNJ11) mutations causing neonatal diabetes. Pflugers Arch 453:323–332
Flanagan SE, Patch AM, Mackay DJ et al (2007) Mutations in ATP-sensitive K+ channel genes cause transient neonatal diabetes and permanent diabetes in childhood or adulthood. Diabetes 56:1930–1937
Dunne MJ, Cosgrove KE, Shepherd RM, Aynsley-Green A, Lindley KJ (2004) Hyperinsulinism in infancy: from basic science to clinical disease. Physiol Rev 84:239–275
Loechner KJ, Akrouh A, Kurata HT et al (2011) Congenital hyperinsulinism and glucose hypersensitivity in homozygous and heterozygous carriers of Kir6.2 (KCNJ11) mutation V290M mutation: K(ATP) channel inactivation mechanism and clinical management. Diabetes 60:209–217
Huopio H, Otonkoski T, Vauhkonen I, Reimann F, Ashcroft FM, Laakso M (2003) A new subtype of autosomal dominant diabetes attributable to a mutation in the gene for sulfonylurea receptor 1. Lancet 361:301–307
Nichols CG, Koster JC, Remedi MS (2007) β-cell hyperexcitability: from hyperinsulinism to diabetes. Diabetes Obes Metab 9(Suppl 2):81–88
Suchi M, MacMullen CM, Thornton PS et al (2006) Molecular and immunohistochemical analyses of the focal form of congenital hyperinsulinism. Mod Pathol 19:122–129
Oyama K, Minami K, Ishizaki K, Fuse M, Miki T, Seino S (2006) Spontaneous recovery from hyperglycemia by regeneration of pancreatic beta-cells in Kir6.2G132S transgenic mice. Diabetes 55:1930–1938
Hwang JS, Shin CH, Yang SW, Jung SY, Huh N (2006) Genetic and clinical characteristics of Korean maturity-onset diabetes of the young (MODY) patients. Diabetes Res Clin Pract 74:75–81
Plengvidhya N, Boonyasrisawat W, Chongjaroen N et al (2009) Mutations of maturity-onset diabetes of the young (MODY) genes in Thais with early-onset type 2 diabetes mellitus. Clin Endocrinol (Oxf) 70:847–853
Acknowledgements
We thank Susumu Seino and Tadao Shibasaki (Division of Cellular and Molecular Medicine, Department of Physiology and Cell Biology, Kobe University Graduate School of Medicine, Japan), Nobuya Inagaki (Department of Diabetes and Clinical Nutrition, Kyoto University, Japan) and Long Yu (State Key Laboratory of Genetic Engineering, School of Life Sciences, Fudan University, China) for their cooperation. We also thank Weiping Jia, Xiaojing Ma, Ming Lu, Can Li, Weijing Zhao, Rong Zhang, Jun Yin, Yuqian Bao and Jing Xu (Shanghai Diabetes Institute, Department of Endocrinology & Metabolism, Shanghai Jiaotong University Affiliated Sixth People’s Hospital, Shanghai, China) for their technical support and cooperation.
Funding
This research was supported by grants from the Project of National Natural Science Foundation of China (nos. 81270876, 30771022 and 30971384) and the Shanghai Scientific & Technical Committee Foundation (nos. 10XD1403400 and 06ZR14051). YL is supported by NIH grant SC1DK087655.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
All authors contributed to the conception and design, or analysis and interpretation of data, drafted the article or revised it critically for important intellectual content, and approved the final version to be published.
Author information
Authors and Affiliations
Corresponding author
Additional information
Limei Liu, Kazuaki Nagashima, Takao Yasuda, Yanjun Liu, Hai-rong Hu and Guang He contributed equally to this study.
Rights and permissions
About this article
Cite this article
Liu, L., Nagashima, K., Yasuda, T. et al. Mutations in KCNJ11 are associated with the development of autosomal dominant, early-onset type 2 diabetes. Diabetologia 56, 2609–2618 (2013). https://doi.org/10.1007/s00125-013-3031-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-013-3031-9