Abstract
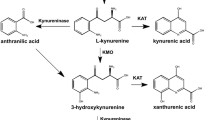
Kynurenine aminotransferases (KATs) catalyze the synthesis of kynurenic acid (KYNA), an endogenous antagonist of N-methyl-d-aspartate and alpha 7-nicotinic acetylcholine receptors. Abnormal KYNA levels in human brains are implicated in the pathophysiology of schizophrenia, Alzheimer’s disease, and other neurological disorders. Four KATs have been reported in mammalian brains, KAT I/glutamine transaminase K/cysteine conjugate beta-lyase 1, KAT II/aminoadipate aminotransferase, KAT III/cysteine conjugate beta-lyase 2, and KAT IV/glutamic-oxaloacetic transaminase 2/mitochondrial aspartate aminotransferase. KAT II has a striking tertiary structure in N-terminal part and forms a new subgroup in fold type I aminotransferases, which has been classified as subgroup Iε. Knowledge regarding KATs is vast and complex; therefore, this review is focused on recent important progress of their gene characterization, physiological and biochemical function, and structural properties. The biochemical differences of four KATs, specific enzyme activity assays, and the structural insights into the mechanism of catalysis and inhibition of these enzymes are discussed.







Similar content being viewed by others
References
Stone TW, Darlington LG (2002) Endogenous kynurenines as targets for drug discovery and development. Nat Rev Drug Discov 1:609–620
Kessler M, Terramani T, Lynch G, Baudry M (1989) A glycine site associated with N-methyl-d-aspartic acid receptors: characterization and identification of a new class of antagonists. J Neurochem 52:1319–1328
Daoudal G, Debanne D (2003) Long-term plasticity of intrinsic excitability: learning rules and mechanisms. Learn Mem 10:456–465
Whitlock JR, Heynen AJ, Shuler MG, Bear MF (2006) Learning induces long-term potentiation in the hippocampus. Science 313:1093–1097
Mark LP, Prost RW, Ulmer JL, Smith MM, Daniels DL, Strottmann JM, Brown WD, Hacein-Bey L (2001) Pictorial review of glutamate excitotoxicity: fundamental concepts for neuroimaging. Am J Neuroradiol 22:1813–1824
Schwarcz R, Guidetti P, Sathyasaikumar KV, Muchowski PJ (2009) Of mice, rats and men: revisiting the quinolinic acid hypothesis of Huntington’s disease. Prog Neurobiol
Vamos E, Pardutz A, Klivenyi P, Toldi J, Vecsei L (2009) The role of kynurenines in disorders of the central nervous system: possibilities for neuroprotection. J Neurol Sci 283:21–27
Heyes MP, Saito K, Crowley JS, Davis LE, Demitrack MA, Der M, Dilling LA, Elia J, Kruesi MJ, Lackner A, Larsen SA, Lee K, Leonard HL, Markey SP, Martin A, Milstein S, Mouradian MM, Pranzatelli MR, Quearry BJ, Salazar A, Smith M, Strauss SE, Sunderland T, Swedo SW, Tourtellotte WW (1992) Quinolinic acid and kynurenine pathway metabolism in inflammatory and non-inflammatory neurological disease. Brain 115(Pt 5):1249–1273
Baran H, Jellinger K, Deecke L (1999) Kynurenine metabolism in Alzheimer’s disease. J Neural Transm 106:165–181
Ogawa T, Matson WR, Beal MF, Myers RH, Bird ED, Milbury P, Saso S (1992) Kynurenine pathway abnormalities in Parkinson’s disease. Neurology 42:1702–1706
Rejdak K, Bartosik-Psujek H, Dobosz B, Kocki T, Grieb P, Giovannoni G, Turski WA, Stelmasiak Z (2002) Decreased level of kynurenic acid in cerebrospinal fluid of relapsing-onset multiple sclerosis patients. Neurosci Lett 331:63–65
Guidetti P, Reddy PH, Tagle DA, Schwarcz R (2000) Early kynurenergic impairment in Huntington’s disease and in a transgenic animal model. Neurosci Lett 283:233–235
Jauch D, Urbanska EM, Guidetti P, Bird ED, Vonsattel JP, Whetsell WO Jr, Schwarcz R (1995) Dysfunction of brain kynurenic acid metabolism in Huntington’s disease: focus on kynurenine aminotransferases. J Neurol Sci 130:39–47
Beal MF, Matson WR, Storey E, Milbury P, Ryan EA, Ogawa T, Bird ED (1992) Kynurenic acid concentrations are reduced in Huntington’s disease cerebral cortex. J Neurol Sci 108:80–87
Baran H, Kepplinger B, Herrera-Marschitz M, Stolze K, Lubec G, Nohl H (2001) Increased kynurenic acid in the brain after neonatal asphyxia. Life Sci 69:1249–1256
Ceresoli-Borroni G, Schwarcz R (2001) Neonatal asphyxia in rats: acute effects on cerebral kynurenine metabolism. Pediatr Res 50:231–235
Medana IM, Day NP, Salahifar-Sabet H, Stocker R, Smythe G, Bwanaisa L, Njobvu A, Kayira K, Turner GD, Taylor TE, Hunt NH (2003) Metabolites of the kynurenine pathway of tryptophan metabolism in the cerebrospinal fluid of Malawian children with malaria. J Infect Dis 188:844–849
Heyes MP, Brew BJ, Saito K, Quearry BJ, Price RW, Lee K, Bhalla RB, Der M, Markey SP (1992) Inter-relationships between quinolinic acid, neuroactive kynurenines, neopterin and beta 2-microglobulin in cerebrospinal fluid and serum of HIV-1-infected patients. J Neuroimmunol 40:71–80
Baran H, Cairns N, Lubec B, Lubec G (1996) Increased kynurenic acid levels and decreased brain kynurenine aminotransferase I in patients with Down syndrome. Life Sci 58:1891–1899
Ilzecka J, Kocki T, Stelmasiak Z, Turski WA (2003) Endogenous protectant kynurenic acid in amyotrophic lateral sclerosis. Acta Neurol Scand 107:412–418
Erhardt S, Blennow K, Nordin C, Skogh E, Lindstrom LH, Engberg G (2001) Kynurenic acid levels are elevated in the cerebrospinal fluid of patients with schizophrenia. Neurosci Lett 313:96–98
Erhardt S, Schwieler L, Nilsson L, Linderholm K, Engberg G (2007) The kynurenic acid hypothesis of schizophrenia. Physiol Behav 92:203–209
Nilsson LK, Linderholm KR, Engberg G, Paulson L, Blennow K, Lindstrom LH, Nordin C, Karanti A, Persson P, Erhardt S (2005) Elevated levels of kynurenic acid in the cerebrospinal fluid of male patients with schizophrenia. Schizophr Res 80:315–322
Wu HQ, Rassoulpour A, Goodman JH, Scharfman HE, Bertram EH, Schwarcz R (2005) Kynurenate and 7-chlorokynurenate formation in chronically epileptic rats. Epilepsia 46:1010–1016
Pereira EF, Hilmas C, Santos MD, Alkondon M, Maelicke A, Albuquerque EX (2002) Unconventional ligands and modulators of nicotinic receptors. J Neurobiol 53:479–500
Hilmas C, Pereira EF, Alkondon M, Rassoulpour A, Schwarcz R, Albuquerque EX (2001) The brain metabolite kynurenic acid inhibits alpha7 nicotinic receptor activity and increases non-alpha7 nicotinic receptor expression: physiopathological implications. J Neurosci 21:7463–7473
Alkondon M, Pereira EF, Yu P, Arruda EZ, Almeida LE, Guidetti P, Fawcett WP, Sapko MT, Randall WR, Schwarcz R, Tagle DA, Albuquerque EX (2004) Targeted deletion of the kynurenine aminotransferase ii gene reveals a critical role of endogenous kynurenic acid in the regulation of synaptic transmission via alpha7 nicotinic receptors in the hippocampus. J Neurosci 24:4635–4648
Stone TW (2007) Kynurenic acid blocks nicotinic synaptic transmission to hippocampal interneurons in young rats. Eur J Neurosci 25:2656–2665
Hogg RC, Raggenbass M, Bertrand D (2003) Nicotinic acetylcholine receptors: from structure to brain function. Rev Physiol Biochem Pharmacol 147:1–46
Gotti C, Clementi F (2004) Neuronal nicotinic receptors: from structure to pathology. Prog Neurobiol 74:363–396
Kalamida D, Poulas K, Avramopoulou V, Fostieri E, Lagoumintzis G, Lazaridis K, Sideri A, Zouridakis M, Tzartos SJ (2007) Muscle and neuronal nicotinic acetylcholine receptors. Structure, function and pathogenicity. FEBS J 274:3799–3845
Albuquerque EX, Pereira EF, Alkondon M, Rogers SW (2009) Mammalian nicotinic acetylcholine receptors: from structure to function. Physiol Rev 89:73–120
Yu P, Di Prospero NA, Sapko MT, Cai T, Chen A, Melendez-Ferro M, Du F, Whetsell WO Jr, Guidetti P, Schwarcz R, Tagle DA (2004) Biochemical and phenotypic abnormalities in kynurenine aminotransferase II-deficient mice. Mol Cell Biol 24:6919–6930
Yu P, Li Z, Zhang L, Tagle DA, Cai T (2006) Characterization of kynurenine aminotransferase III, a novel member of a phylogenetically conserved KAT family. Gene 365:111–118
Carpenedo R, Pittaluga A, Cozzi A, Attucci S, Galli A, Raiteri M, Moroni F (2001) Presynaptic kynurenate-sensitive receptors inhibit glutamate release. Eur J Neurosci 13:2141–2147
Rassoulpour A, Wu HQ, Ferre S, Schwarcz R (2005) Nanomolar concentrations of kynurenic acid reduce extracellular dopamine levels in the striatum. J Neurochem 93:762–765
Grilli M, Raiteri L, Patti L, Parodi M, Robino F, Raiteri M, Marchi M (2006) Modulation of the function of presynaptic alpha7 and non-alpha7 nicotinic receptors by the tryptophan metabolites, 5-hydroxyindole and kynurenate in mouse brain. Br J Pharmacol 149:724–732
Wu HQ, Rassoulpour A, Schwarcz R (2007) Kynurenic acid leads, dopamine follows: a new case of volume transmission in the brain? J Neural Transm 114:33–41
Zmarowski A, Wu HQ, Brooks JM, Potter MC, Pellicciari R, Schwarcz R, Bruno JP (2009) Astrocyte-derived kynurenic acid modulates basal and evoked cortical acetylcholine release. Eur J Neurosci 29:529–538
Sapko MT, Guidetti P, Yu P, Tagle DA, Pellicciari R, Schwarcz R (2006) Endogenous kynurenate controls the vulnerability of striatal neurons to quinolinate: implications for Huntington’s disease. Exp Neurol 197:31–40
Coyle JT (2006) Glial metabolites of tryptophan and excitotoxicity: coming unglued. Exp Neurol 197:4–7
Wang J, Simonavicius N, Wu X, Swaminath G, Reagan J, Tian H, Ling L (2006) Kynurenic acid as a ligand for orphan G protein-coupled receptor GPR35. J Biol Chem 281:22021–22028
Mellor AL, Munn DH (2004) IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat Rev Immunol 4:762–774
Taylor MW, Feng GS (1991) Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J 5:2516–2522
Colombari E, Sato MA, Cravo SL, Bergamaschi CT, Campos RR Jr, Lopes OU (2001) Role of the medulla oblongata in hypertension. Hypertension 38:549–554
Ito S, Komatsu K, Tsukamoto K, Sved AF (2000) Excitatory amino acids in the rostral ventrolateral medulla support blood pressure in spontaneously hypertensive rats. Hypertension 35:413–417
Kwok JB, Kapoor R, Gotoda T, Iwamoto Y, Iizuka Y, Yamada N, Isaacs KE, Kushwaha VV, Church WB, Schofield PR, Kapoor V (2002) A missense mutation in kynurenine aminotransferase-1 in spontaneously hypertensive rats. J Biol Chem 277:35779–35782
Fukui S, Schwarcz R, Rapoport SI, Takada Y, Smith QR (1991) Blood–brain barrier transport of kynurenines: implications for brain synthesis and metabolism. J Neurochem 56:2007–2017
Okuno E, Nakamura M, Schwarcz R (1991) Two kynurenine aminotransferases in human brain. Brain Res 542:307–312
Guidetti P, Okuno E, Schwarcz R (1997) Characterization of rat brain kynurenine aminotransferases I and II. J Neurosci Res 50:457–465
Schwarcz R, Pellicciari R (2002) Manipulation of brain kynurenines: glial targets, neuronal effects, and clinical opportunities. J Pharmacol Exp Ther 303:1–10
Han Q, Li J, Li J (2004) pH dependence, substrate specificity and inhibition of human kynurenine aminotransferase I. Eur J Biochem 271:4804–4814
Guidetti P, Amori L, Sapko MT, Okuno E, Schwarcz R (2007) Mitochondrial aspartate aminotransferase: a third kynurenate-producing enzyme in the mammalian brain. J Neurochem 102:103–111
Han Q, Robinson H, Cai T, Tagle DA, Li J (2009) Biochemical and structural properties of mouse kynurenine aminotransferase III. Mol Cell Biol 29:784–793
Perez-De La Cruz V, Konigsberg M, Santamaria A (2007) Kynurenine pathway and disease: an overview. CNS Neurol Disord Drug Targets 6:398–410
Nemeth H, Toldi J, Vecsei L (2005) Role of kynurenines in the central and peripheral nervous systems. Curr Neurovasc Res 2:249–260
Schwarcz R (2004) The kynurenine pathway of tryptophan degradation as a drug target. Curr Opin Pharmacol 4:12–17
Stone TW, Mackay GM, Forrest CM, Clark CJ, Darlington LG (2003) Tryptophan metabolites and brain disorders. Clin Chem Lab Med 41:852–859
Costantino G (2009) New promises for manipulation of kynurenine pathway in cancer and neurological diseases. Expert Opin Ther Targets 13:247–258
Erhardt S, Olsson SK, Engberg G (2009) Pharmacological manipulation of kynurenic acid: potential in the treatment of psychiatric disorders. CNS Drugs 23:91–101
Nemeth H, Toldi J, Vecsei L (2006) Kynurenines, Parkinson’s disease and other neurodegenerative disorders: preclinical and clinical studies. J Neural Transm 70(Suppl):285–304
Perry S, Harries H, Scholfield C, Lock T, King L, Gibson G, Goldfarb P (1995) Molecular cloning and expression of a cDNA for human kidney cysteine conjugate beta-lyase. FEBS Lett 360:277–280
Goh DL, Patel A, Thomas GH, Salomons GS, Schor DS, Jakobs C, Geraghty MT (2002) Characterization of the human gene encoding alpha-aminoadipate aminotransferase (AADAT). Mol Genet Metab 76:172–180
Martini F, Angelaccio S, Barra D, Pascarella S, Maras B, Doonan S, Bossa F (1985) The primary structure of mitochondrial aspartate aminotransferase from human heart. Biochim Biophys Acta 832:46–51
Papadimitriou JM, van Duijn P (1970) The ultrastructural localization of the isozymes of aspartate aminotransferase in murine tissues. J Cell Biol 47:84–98
Tanaka M, Takeda N, Tohyama M, Matsunaga T (1990) Immunocytochemical localization of mitochondrial and cytosolic aspartate aminotransferase isozymes in the vestibular end-organs of rats. Eur Arch Otorhinolaryngol 247:119–121
Giannattasio S, Marra E, Abruzzese MF, Greco M, Quagliariello E (1991) The in vitro-synthesized precursor and mature mitochondrial aspartate aminotransferase share the same import pathway in isolated mitochondria. Arch Biochem Biophys 290:528–534
Faff-Michalak L, Albrecht J (1991) Aspartate aminotransferase, malate dehydrogenase, and pyruvate carboxylase activities in rat cerebral synaptic and nonsynaptic mitochondria: effects of in vitro treatment with ammonia, hyperammonemia and hepatic encephalopathy. Metab Brain Dis 6:187–197
McKenna MC, Stevenson JH, Huang X, Hopkins IB (2000) Differential distribution of the enzymes glutamate dehydrogenase and aspartate aminotransferase in cortical synaptic mitochondria contributes to metabolic compartmentation in cortical synaptic terminals. Neurochem Int 37:229–241
Cechetto JD, Sadacharan SK, Berk PD, Gupta RS (2002) Immunogold localization of mitochondrial aspartate aminotransferase in mitochondria and on the cell surface in normal rat tissues. Histol Histopathol 17:353–364
Shrawder E, Martinez-Carrion M (1972) Evidence of phenylalanine transaminase activity in the isoenzymes of aspartate transaminase. J Biol Chem 247:2486–2492
Suzuki T, Kishi Y, Totani M, Kagamiyama H, Murachi T (1987) Monoclonal and polyclonal antibodies against porcine mitochondrial aspartate aminotransferase: their inhibition modes and application to enzyme immunoassay. Biotechnol Appl Biochem 9:170–180
Mawal MR, Mukhopadhyay A, Deshmukh DR (1991) Purification and properties of alpha-aminoadipate aminotransferase from rat liver and kidney mitochondria. Prep Biochem 21:151–162
Takeuchi F, Otsuka H, Shibata Y (1983) Purification, characterization and identification of rat liver mitochondrial kynurenine aminotransferase with alpha-aminoadipate aminotransferase. Biochim Biophys Acta 743:323–330
Rossi F, Garavaglia S, Montalbano V, Walsh MA, Rizzi M (2008) Crystal structure of human kynurenine aminotransferase II, a drug target for the treatment of schizophrenia. J Biol Chem 283:3559–3566
Han Q, Robinson H, Li J (2008) Crystal structure of human kynurenine aminotransferase II. J Biol Chem 283:3567–3573
Fang J, Han Q, Li J (2002) Isolation, characterization, and functional expression of kynurenine aminotransferase cDNA from the yellow fever mosquito, Aedes aegypti(1). Insect Biochem Mol Biol 32:943–950
Han Q, Li J (2004) Cysteine and keto acids modulate mosquito kynurenine aminotransferase catalyzed kynurenic acid production. FEBS Lett 577:381–385
Cooper AJ (2004) The role of glutamine transaminase K (GTK) in sulfur and alpha-keto acid metabolism in the brain, and in the possible bioactivation of neurotoxicants. Neurochem Int 44:557–577
Cooper AJ, Meister A (1981) Comparative studies of glutamine transaminases from rat tissues. Comp Biochem Physiol B 69B:137–145
McGoldrick TA, Lock EA, Rodilla V, Hawksworth GM (2003) Renal cysteine conjugate C-S lyase mediated toxicity of halogenated alkenes in primary cultures of human and rat proximal tubular cells. Arch Toxicol 77:365–370
Dekant W, Vamvakas S, Anders MW (1994) Formation and fate of nephrotoxic and cytotoxic glutathione S-conjugates: cysteine conjugate beta-lyase pathway. Adv Pharmacol 27:115–162
Spencer JP, Whiteman M, Jenner P, Halliwell B (2002) 5-S-Cysteinyl-conjugates of catecholamines induce cell damage, extensive DNA base modification and increases in caspase-3 activity in neurons. J Neurochem 81:122–129
Cooper AJ, Pinto JT, Krasnikov BF, Niatsetskaya ZV, Han Q, Li J, Vauzour D, Spencer JP (2008) Substrate specificity of human glutamine transaminase K as an aminotransferase and as a cysteine S-conjugate beta-lyase. Arch Biochem Biophys 474:72–81
Matsuda M, Ogur M (1969) Enzymatic and physiological properties of the yeast glutamate-alpha-ketoadipate transaminase. J Biol Chem 244:5153–5158
Tobes MC, Mason M (1975) l-kynurenine aminotransferase and l-alpha-aminoadipate aminotransferase. I. Evidence for identity. Biochem Biophys Res Commun 62:390–397
Tobes MC, Mason M (1977) Alpha-Aminoadipate aminotransferase and kynurenine aminotransferase. Purification, characterization, and further evidence for identity. J Biol Chem 252:4591–4599
Buchli R, Alberati-Giani D, Malherbe P, Kohler C, Broger C, Cesura AM (1995) Cloning and functional expression of a soluble form of kynurenine/alpha-aminoadipate aminotransferase from rat kidney. J Biol Chem 270:29330–29335
Yu P, Mosbrook DM, Tagle DA (1999) Genomic organization and expression analysis of mouse kynurenine aminotransferase II, a possible factor in the pathophysiology of Huntington’s disease. Mamm Genome 10:845–852
Han Q, Cai T, Tagle DA, Robinson H, Li J (2008) Substrate specificity and structure of human aminoadipate aminotransferase/kynurenine aminotransferase II. Biosci Rep 28:205–215
Miyazaki T, Miyazaki J, Yamane H, Nishiyama M (2004) alpha-Aminoadipate aminotransferase from an extremely thermophilic bacterium, Thermus thermophilus. Microbiology 150:2327–2334
Xu H, Andi B, Qian J, West AH, Cook PF (2006) The alpha-aminoadipate pathway for lysine biosynthesis in fungi. Cell Biochem Biophys 46:43–64
Zabriskie TM, Jackson MD (2000) Lysine biosynthesis and metabolism in fungi. Nat Prod Rep 17:85–97
Matsuda M, Ogur M (1969) Separation and specificity of the yeast glutamate-alpha-ketoadipate transaminase. J Biol Chem 244:3352–3358
Han Q, Fang J, Li J (2001) Kynurenine aminotransferase and glutamine transaminase K of Escherichia coli: identity with aspartate aminotransferase. Biochem J 360:617–623
Cooper AJ, Meister A (1985) Metabolic significance of transamination. In: Christen P, Metzler DE (eds) Transaminases. Wiley, New York, pp 500–580
Moran J, Alavez S, Rivera-Gaxiola M, Valencia A, Hurtado S (1999) Effect of NMDA antagonists on the activity of glutaminase and aspartate aminotransferase in the developing rat cerebellum. Int J Dev Neurosci 17:57–65
Moran J, Rivera-Gaxiola M (1992) Effect of potassium and N-methyl-d-aspartate on the aspartate aminotransferase activity in cultured cerebellar granule cells. J Neurosci Res 33:239–247
Westergaard N, Drejer J, Schousboe A, Sonnewald U (1996) Evaluation of the importance of transamination versus deamination in astrocytic metabolism of [U-13C]glutamate. Glia 17:160–168
Yudkoff M, Daikhin Y, Nissim I, Horyn O, Lazarow A, Luhovyy B, Wehrli S (2005) Response of brain amino acid metabolism to ketosis. Neurochem Int 47:119–128
Schousboe A, Westergaard N, Sonnewald U, Petersen SB, Huang R, Peng L, Hertz L (1993) Glutamate and glutamine metabolism and compartmentation in astrocytes. Dev Neurosci 15:359–366
Hertz L, Drejer J, Schousboe A (1988) Energy metabolism in glutamatergic neurons, GABAergic neurons and astrocytes in primary cultures. Neurochem Res 13:605–610
McKenna MC, Tildon JT, Stevenson JH, Boatright R, Huang S (1993) Regulation of energy metabolism in synaptic terminals and cultured rat brain astrocytes: differences revealed using aminooxyacetate. Dev Neurosci 15:320–329
Sonnewald U, Westergaard N, Schousboe A (1997) Glutamate transport and metabolism in astrocytes. Glia 21:56–63
McKenna MC, Tildon JT, Stevenson JH, Huang X (1996) New insights into the compartmentation of glutamate and glutamine in cultured rat brain astrocytes. Dev Neurosci 18:380–390
Palaiologos G, Hertz L, Schousboe A (1989) Role of aspartate aminotransferase and mitochondrial dicarboxylate transport for release of endogenously and exogenously supplied neurotransmitter in glutamatergic neurons. Neurochem Res 14:359–366
Palaiologos G, Hertz L, Schousboe A (1988) Evidence that aspartate aminotransferase activity and ketodicarboxylate carrier function are essential for biosynthesis of transmitter glutamate. J Neurochem 51:317–320
Fitzpatrick SM, Cooper AJ, Duffy TE (1983) Use of beta-methylene-d, l-aspartate to assess the role of aspartate aminotransferase in cerebral oxidative metabolism. J Neurochem 41:1370–1383
Cheeseman AJ, Clark JB (1988) Influence of the malate–aspartate shuttle on oxidative metabolism in synaptosomes. J Neurochem 50:1559–1565
McKenna MC, Waagepetersen HS, Schousboe A, Sonnewald U (2006) Neuronal and astrocytic shuttle mechanisms for cytosolic–mitochondrial transfer of reducing equivalents: current evidence and pharmacological tools. Biochem Pharmacol 71:399–407
McKenna MC, Hopkins IB, Lindauer SL, Bamford P (2006) Aspartate aminotransferase in synaptic and nonsynaptic mitochondria: differential effect of compounds that influence transient hetero-enzyme complex (metabolon) formation. Neurochem Int 48:629–636
Cooper AJ, Bruschi SA, Iriarte A, Martinez-Carrion M (2002) Mitochondrial aspartate aminotransferase catalyses cysteine S-conjugate beta-lyase reactions. Biochem J 368:253–261
Baran H, Okuno E, Kido R, Schwarcz R (1994) Purification and characterization of kynurenine aminotransferase I from human brain. J Neurochem 62:730–738
Schmidt W, Guidetti P, Okuno E, Schwarcz R (1993) Characterization of human brain kynurenine aminotransferases using [3H]kynurenine as a substrate. Neuroscience 55:177–184
Milart P, Urbanska EM, Turski WA, Paszkowski T, Sikorski R (2001) Kynurenine aminotransferase I activity in human placenta. Placenta 22:259–261
Baran H, Amann G, Lubec B, Lubec G (1997) Kynurenic acid and kynurenine aminotransferase in heart. Pediatr Res 41:404–410
Han Q, Robinson H, Cai T, Tagle DA, Li J (2009) Structural insight into the inhibition of human kynurenine aminotransferase I/glutamine transaminase K. J Med Chem 52:2786–2793
Pellicciari R, Venturoni F, Bellocchi D, Carotti A, Marinozzi M, Macchiarulo A, Amori L, Schwarcz R (2008) Sequence variants in kynurenine aminotransferase II (KAT II) orthologs determine different potencies of the inhibitor S-ESBA. ChemMedChem 3:1199–1202
Pellicciari R, Rizzo RC, Costantino G, Marinozzi M, Amori L, Guidetti P, Wu HQ, Schwarcz R (2006) Modulators of the kynurenine pathway of tryptophan metabolism: synthesis and preliminary biological evaluation of (S)-4-(ethylsulfonyl)benzoylalanine, a potent and selective kynurenine aminotransferase II (KAT II) inhibitor. ChemMedChem 1:528–531
Rossi F, Han Q, Li J, Li J, Rizzi M (2004) Crystal structure of human kynurenine aminotransferase I. J Biol Chem 279:50214–50220
Goto M, Omi R, Miyahara I, Hosono A, Mizuguchi H, Hayashi H, Kagamiyama H, Hirotsu K (2004) Crystal structures of glutamine:phenylpyruvate aminotransferase from Thermus thermophilus HB8: induced fit and substrate recognition. J Biol Chem 279:16518–16525
Han Q, Gao YG, Robinson H, Ding H, Wilson S, Li J (2005) Crystal structures of Aedes aegypti kynurenine aminotransferase. FEBS J 272:2198–2206
Wogulis M, Chew ER, Donohoue PD, Wilson DK (2008) Identification of formyl kynurenine formamidase and kynurenine aminotransferase from Saccharomyces cerevisiae using crystallographic, bioinformatic and biochemical evidence. Biochemistry 47:1608–1621
Tomita T, Miyagawa T, Miyazaki T, Fushinobu S, Kuzuyama T, Nishiyama M (2009) Mechanism for multiple-substrates recognition of alpha-aminoadipate aminotransferase from Thermus thermophilus. Proteins 75:348–359
Chon H, Matsumura H, Koga Y, Takano K, Kanaya S (2005) Crystal structure of a human kynurenine aminotransferase II homologue from Pyrococcus horikoshii OT3 at 2.20 A resolution. Proteins 61:685–688
McPhalen CA, Vincent MG, Jansonius JN (1992) X-ray structure refinement and comparison of three forms of mitochondrial aspartate aminotransferase. J Mol Biol 225:495–517
Rossi F, Schwarcz R, Rizzi M (2008) Curiosity to kill the KAT (kynurenine aminotransferase): structural insights into brain kynurenic acid synthesis. Curr Opin Struct Biol 18:748–755
Eliot AC, Kirsch JF (2004) Pyridoxal phosphate enzymes: mechanistic, structural, and evolutionary considerations. Annu Rev Biochem 73:383–415
Han Q, Gao YG, Robinson H, Li J (2008) Structural insight into the mechanism of substrate specificity of Aedes kynurenine aminotransferase. Biochemistry 47:1622–1630
Okamoto A, Nakai Y, Hayashi H, Hirotsu K, Kagamiyama H (1998) Crystal structures of Paracoccus denitrificans aromatic amino acid aminotransferase: a substrate recognition site constructed by rearrangement of hydrogen bond network. J Mol Biol 280:443–461
Han Q, Robinson H, Gao YG, Vogelaar N, Wilson SR, Rizzi M, Li J (2006) Crystal structures of Aedes aegypti alanine glyoxylate aminotransferase. J Biol Chem 281:37175–37182
Jansonius JN (1998) Structure, evolution and action of vitamin B6-dependent enzymes. Curr Opin Struct Biol 8:759–769
Grishin NV, Phillips MA, Goldsmith EJ (1995) Modeling of the spatial structure of eukaryotic ornithine decarboxylases. Protein Sci 4:1291–1304
Kack H, Sandmark J, Gibson K, Schneider G, Lindqvist Y (1999) Crystal structure of diaminopelargonic acid synthase: evolutionary relationships between pyridoxal-5′-phosphate-dependent enzymes. J Mol Biol 291:857–876
Schneider G, Kack H, Lindqvist Y (2000) The manifold of vitamin B6 dependent enzymes. Structure 8:R1–R6
Mehta PK, Hale TI, Christen P (1993) Aminotransferases: demonstration of homology and division into evolutionary subgroups. Eur J Biochem 214:549–561
DeLano WL (2002) The PyMOL molecular graphics system. Delano Scientific, San Carlos
Jensen RA, Gu W (1996) Evolutionary recruitment of biochemically specialized subdivisions of Family I within the protein superfamily of aminotransferases. J Bacteriol 178:2161–2171
Hohenester E, Jansonius JN (1994) Crystalline mitochondrial aspartate aminotransferase exists in only two conformations. J Mol Biol 236:963–968
Rhee S, Silva MM, Hyde CC, Rogers PH, Metzler CM, Metzler DE, Arnone A (1997) Refinement and comparisons of the crystal structures of pig cytosolic aspartate aminotransferase and its complex with 2-methylaspartate. J Biol Chem 272:17293–17302
Miyahara I, Hirotsu K, Hayashi H, Kagamiyama H (1994) X-ray crystallographic study of pyridoxamine 5′-phosphate-type aspartate aminotransferases from Escherichia coli in three forms. J Biochem (Tokyo) 116:1001–1012
Malashkevich VN, Strokopytov BV, Borisov VV, Dauter Z, Wilson KS, Torchinsky YM (1995) Crystal structure of the closed form of chicken cytosolic aspartate aminotransferase at 1.9 A resolution. J Mol Biol 247:111–124
Okamoto A, Higuchi T, Hirotsu K, Kuramitsu S, Kagamiyama H (1994) X-ray crystallographic study of pyridoxal 5′-phosphate-type aspartate aminotransferases from Escherichia coli in open and closed form. J Biochem (Tokyo) 116:95–107
Jager J, Moser M, Sauder U, Jansonius JN (1994) Crystal structures of Escherichia coli aspartate aminotransferase in two conformations. Comparison of an unliganded open and two liganded closed forms. J Mol Biol 239:285–305
Hirotsu K, Goto M, Okamoto A, Miyahara I (2005) Dual substrate recognition of aminotransferases. Chem Rec 5:160–172
Acknowledgment
This work was supported by a NS063836 grant, an Intramural Research Program of the institutes of NIDCR and NINDS at NIH. We are grateful to Elizabeth Watson (Department of Biochemistry, Virginia Tech) for critically reading this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, Q., Cai, T., Tagle, D.A. et al. Structure, expression, and function of kynurenine aminotransferases in human and rodent brains. Cell. Mol. Life Sci. 67, 353–368 (2010). https://doi.org/10.1007/s00018-009-0166-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-009-0166-4